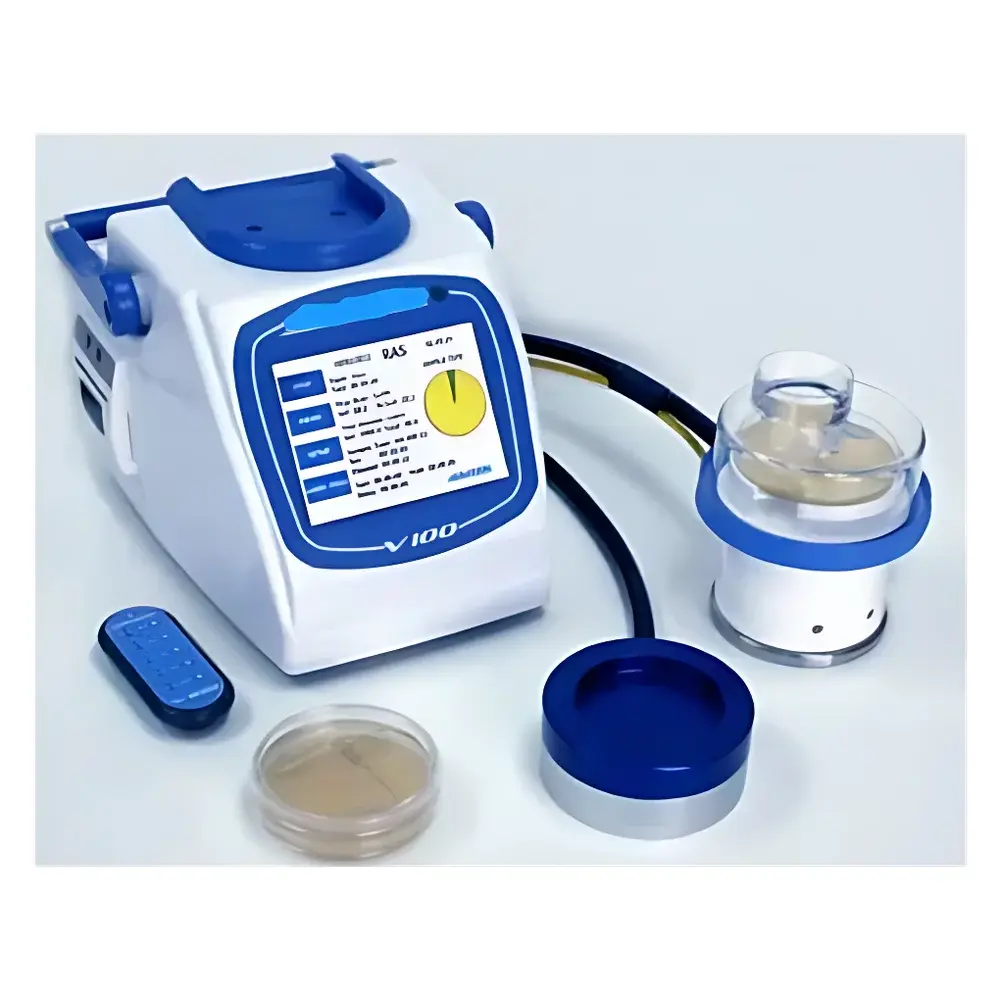
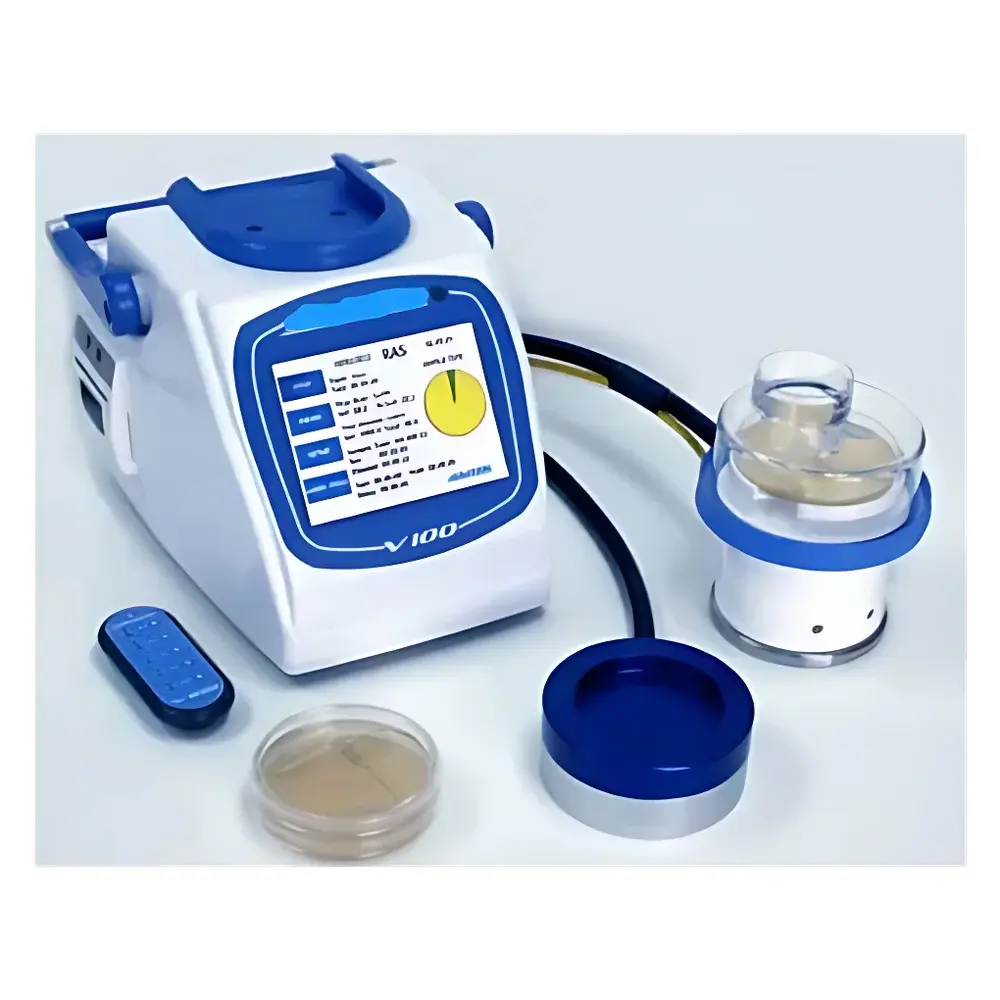
Starwood V100 Compressed Air Microbial Air Sampler
| Brand | Starwood |
|---|---|
| Origin | Beijing, China |
| Manufacturer Type | OEM/ODM Manufacturer |
| Country of Origin | China |
| Model | V100 |
| Pricing | Available Upon Request |
| Flow Rates | 28.3 L/min, 50 L/min, 100 L/min |
| Display | Color LCD with Touch Interface |
| Data Storage | >500 Sampling Records |
| Connectivity | Ethernet & USB |
| Weight | 12 lbs (5.4 kg) |
| Dimensions | 10″ L × 8″ W × 7″ H (254 × 203 × 178 mm) |
| Power Supply | 100–240 V AC, 50/60 Hz |
| Exhaust Filter | 0.2 µm Sterile Vent Filter |
| Housing & Sampling Heads | 316 Stainless Steel + Microban®-Impregnated Kydex® |
| Sampling Methods | Andersen-style Impaction & Rotary Slit™ Impaction |
| Sampling Head Options | R2S (Rotary Slit Standard), RCG (Compressed Gas), RAS (Remote Access Sampling) |
| Remote Control | IR-enabled (Start/Stop/Pause/Resume) |
| Calibration | On-screen Test Plate Verification |
| Sampling Duration per Cycle | 0–120 min |
| Programmable Parameters | Delay Time, Start Time, Sample Duration, Number of Cycles, Inter-Cycle Pause |
| Print Output | Thermal Label + Direct Thermal Paper |
| Sampling Capacity | >300 Agar Plates per Full Memory Cycle |
Overview
The Starwood V100 Compressed Air Microbial Air Sampler is an ISO 14698-1–compliant active air sampler engineered for quantitative recovery of viable airborne microorganisms from compressed air systems and controlled environments. It operates on the principle of inertial impaction—where aerosolized particles are accelerated through precision-engineered orifices and deposited onto nutrient agar surfaces at velocities calibrated to match local airflow conditions. This isothermal, isokinetic sampling design ensures representative microbial capture without desiccation or shear-induced viability loss. Unlike passive settle plates, the V100 delivers traceable, reproducible colony-forming unit (CFU) data required for GMP-regulated environments—including pharmaceutical cleanrooms, medical gas distribution networks, and aseptic manufacturing suites—where compressed air is classified as a critical process fluid per ISO 8573-7 and EU Annex 1.
Key Features
- Triple-mode sampling architecture supporting standard impactor heads (R2S), high-pressure compressed gas adapters (RCG), and remote-access configurations (RAS)—all interoperable with a single V100 host unit
- Programmable multi-stage sampling protocols: configurable delay, start time, cycle count, inter-cycle pause, and per-cycle duration (0–120 min)
- Three selectable volumetric flow rates—28.3 L/min (equivalent to 1 ft³/min), 50 L/min, and 100 L/min—each independently validated via on-screen test plate calibration
- Microban®-infused Kydex® sampling heads and 316 stainless steel structural components minimize surface bioburden carryover between samples
- Integrated thermal printer supports dual-output media: peel-and-stick sample labels and continuous thermal paper—both auto-annotated with timestamp, flow rate, duration, and head type
- IR remote control eliminates operator proximity during sampling initiation/termination, reducing contamination risk in Grade A/B zones
- Adjustable agar height compensation mechanism maintains consistent impaction geometry across variable agar thicknesses (e.g., ±1.5 mm tolerance)
- Embedded audit trail functionality logs all user actions, parameter changes, and system events—aligned with FDA 21 CFR Part 11 data integrity expectations
Sample Compatibility & Compliance
The V100 meets the performance criteria outlined in ISO 14698-1:2003 (Biocontamination control — General principles and methods) and supports validation per USP <1116>, EU GMP Annex 1 (2022), and ISO 8573-7:2019 (Compressed air — Part 7: Viability testing of microbiological contaminants). Its rotary slit (R2S) and Andersen-style impactor heads demonstrate ≥95% collection efficiency for particles ≥0.1 µm under laminar flow conditions, verified using polystyrene latex (PSL) microspheres and Bacillus atrophaeus spore challenges. All wetted materials comply with USP Class VI biocompatibility standards. The 0.2 µm sterile exhaust filter prevents cross-contamination between sampling events and satisfies containment requirements for BSL-2 environments.
Software & Data Management
Data acquisition and reporting are managed via embedded firmware supporting export-ready CSV generation over USB or Ethernet. Each stored record (n > 500) includes full metadata: date/time stamp, selected flow rate, actual measured flow (via integrated mass flow sensor), sampling duration, head ID, agar lot number (manually entered), and operator ID. The system supports GLP/GMP-aligned electronic signatures when paired with validated third-party LIMS platforms. Firmware updates are delivered via signed binary packages; version history and checksum verification are logged automatically. No cloud dependency—data remains resident on-device unless explicitly exported.
Applications
The V100 is deployed across regulated and research-intensive settings where compressed air quality directly impacts product sterility or personnel safety. Primary use cases include: environmental monitoring of ISO Class 5–8 cleanrooms; routine surveillance of instrument air, nitrogen, and oxygen lines in pharmaceutical fill-finish suites; validation of sterile barrier integrity in isolators and RABS; microbial load assessment in hospital central air supply systems; and bioburden characterization in food-grade pneumatic conveying lines. Its portability (12 lb / 5.4 kg) and battery-free operation enable field-deployed audits of off-site compressor stations or mobile GMP facilities.
FAQ
Does the V100 comply with ISO 8573-7 for compressed air microbiological testing?
Yes—the V100’s calibrated flow control, isokinetic sampling head design, and validated recovery efficiency meet the technical requirements of ISO 8573-7:2019 for viable particle enumeration in compressed air.
Can the same V100 unit be used for both ambient room air and high-pressure compressed gas lines?
Yes—by swapping to the RCG (Compressed Gas) sampling head, the system accommodates inlet pressures up to 10 bar(g) while maintaining laminar impaction dynamics and agar viability.
Is firmware validation documentation available for GMP audits?
Yes—Starwood provides IQ/OQ protocol templates, traceable calibration certificates for mass flow sensors, and a complete firmware validation report covering all functional and data integrity requirements.
How is calibration traceability maintained across multiple sampling heads?
Each head is individually serialized and mapped to its certified flow correction factor; the host unit applies real-time compensation during sampling based on selected head ID.
What regulatory standards does the data logging satisfy?
The embedded audit trail captures user identity, timestamp, parameter values, and system status—meeting ALCOA+ principles and supporting compliance with FDA 21 CFR Part 11, EU Annex 11, and ISO/IEC 17025 data management clauses.