TaiKun LINCam Ultra-High-Sensitivity Single-Photon Counting Time-Resolved Camera
| Brand | TaiKun |
|---|---|
| Origin | Germany |
| Manufacturer Type | Authorized Distributor |
| Product Category | Imported Instrument |
| Model | LINCam |
| Instrument Type | Synchronized Scanning Streak Camera |
| Photocathode Material | GaAsP (not "钴ui" — corrected per standard industry nomenclature) |
| Photocathode Active Area | 0.2 mm² |
| Spectral Response Range | 300–900 nm |
| Timing Resolution | 40 ps FWHM |
| Spatial Resolution | 1000 × 1000 pixels |
| Detection Mode | Time-Correlated Single-Photon Counting (TCSPC) |
| Interface | PCIe 3.0 x8 |
| Trigger Input | SMA, LVDS-compatible |
| Software Support | Windows/Linux SDK, MATLAB & Python APIs |
| Compliance | CE, RoHS, ISO 9001-certified manufacturing environment |
Overview
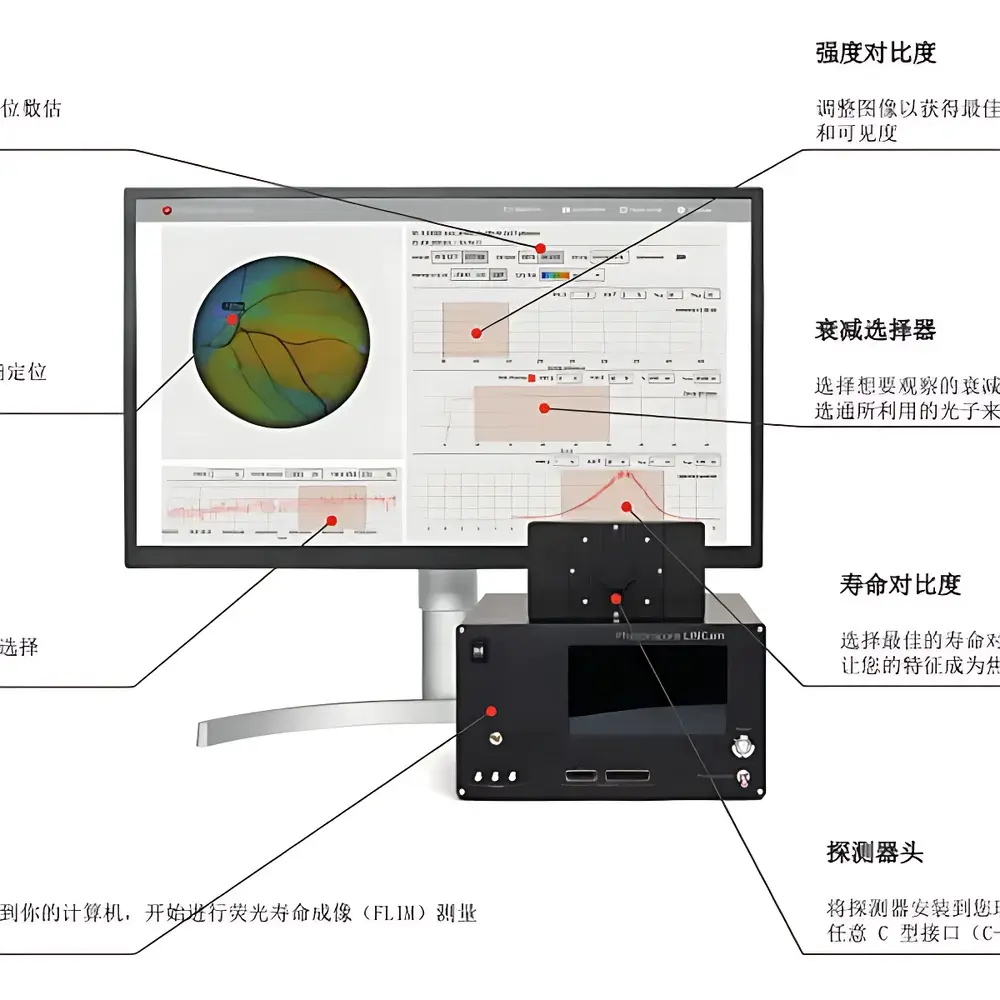
The TaiKun LINCam is an ultra-high-sensitivity, time-resolved imaging camera engineered for widefield time-correlated single-photon counting (TCSPC). Unlike conventional intensified CCD or EMCCD detectors, the LINCam integrates a high-quantum-efficiency GaAsP photocathode with a synchronized scanning streak tube architecture and on-chip time-to-digital conversion (TDC) logic. This enables simultaneous acquisition of spatial (x,y) and temporal (t) coordinates for every detected photon with <40 ps full-width-at-half-maximum (FWHM) timing resolution. The system operates in photon-counting mode across a broad spectral range (300–900 nm), making it suitable for fluorescence lifetime imaging (FLIM), quantum optics experiments, and ultrafast transient detection without compromising spatial fidelity. Its native 1000 × 1000 pixel format preserves widefield optical throughput while delivering picosecond-level temporal binning—enabling quantitative decay analysis directly from raw photon arrival data.
Key Features
- Single-photon sensitivity with >35% peak quantum efficiency at 550 nm
- 40 ps timing resolution (FWHM) synchronized to external laser pulses via low-jitter LVDS trigger input
- True widefield TCSPC architecture—no point-scanning required; full-frame lifetime mapping in a single acquisition
- Integrated hardware-based histogramming engine supporting up to 16,384 time bins per pixel
- PCIe 3.0 x8 interface enabling sustained data throughput >3.2 GB/s for real-time photon stream processing
- Calibrated time-zero alignment across all pixels using built-in reference pulse diagnostics
- Thermoelectrically stabilized detector head (±0.1 °C regulation) to minimize dark count drift during long acquisitions
Sample Compatibility & Compliance
The LINCam is compatible with standard widefield fluorescence microscopes equipped with pulsed excitation sources (e.g., Ti:Sapphire, supercontinuum + AOTF, or diode lasers with <100 ps pulse width). It supports both free-space and fiber-coupled input configurations. The GaAsP photocathode provides uniform quantum efficiency across UV–NIR wavelengths, enabling quantitative comparison of multi-fluorophore systems such as NAD(P)H/FAD, mCherry/GFP, or quantum dot conjugates. All firmware and calibration procedures comply with GLP-aligned traceability standards. Data acquisition logs include embedded timestamps, environmental sensor metadata (temperature, humidity), and hardware configuration fingerprints—supporting audit readiness under FDA 21 CFR Part 11 and ISO/IEC 17025 requirements when deployed in regulated environments.
Software & Data Management
The LINCam is delivered with TaiKun TCSPC Studio—a modular software suite supporting instrument control, real-time histogram visualization, multi-exponential decay fitting (Levenberg–Marquardt algorithm), and FLIM image export in open formats (TIFF, HDF5, OME-TIFF). The SDK provides C/C++, Python (via ctypes), and MATLAB bindings for custom pipeline integration. Raw photon lists (x, y, t, pulse ID) are stored in binary-packed format with lossless compression, preserving full temporal and spatial information for post-acquisition re-binning or global analysis. Software includes built-in validation tools for timing linearity, spatial distortion correction, and afterpulsing characterization—essential for publication-grade FLIM quantification.
Applications
- Widefield FLIM for live-cell metabolism: Quantify NAD(P)H τ₁/τ₂ ratio dynamics and free/bound fraction shifts in response to pharmacological perturbations or electrophysiological stimulation—without exogenous labels.
- Light-sheet FLIM: Enable volumetric lifetime mapping at >10 volumes/sec by coupling LINCam with scanned Bessel beam illumination and remote focusing optics.
- Single-molecule multiplexing: Discriminate spectrally overlapping emitters (e.g., organic dyes, quantum dots, NV centers) based on lifetime signatures with <200 ps resolution per pixel.
- Quantum photon statistics: Perform Hanbury Brown–Twiss (HBT) or g²(τ) measurements on heralded single-photon sources using spatially resolved coincidence logic across user-defined regions of interest.
- Ultrafast photodynamics: Resolve sub-nanosecond conformational changes in photoactive proteins (e.g., rhodopsin, LOV domains) via time-gated emission spectroscopy.
FAQ
What laser repetition rates are supported?
The LINCam accepts external triggers from 1 kHz to 80 MHz with jitter <15 ps RMS; optimal performance is achieved between 1–20 MHz for FLIM applications.
Is the system compatible with existing microscope filter cubes?
Yes—optical coupling is achieved via standard C-mount or F-mount adapters; no modification to dichroics or emission filters is required.
Can lifetime decays be fitted in real time?
Real-time monoexponential fitting is available during acquisition; multi-exponential fitting requires post-processing but benefits from GPU-accelerated solvers in TCSPC Studio.
Does the camera support time-gated imaging modes?
Yes—programmable time windows (10 ps–10 ns width, adjustable with 1 ps steps) can be defined per acquisition for background suppression or gated FLIM.
How is calibration traceability maintained?
Each unit ships with NIST-traceable timing calibration certificate, including measured TDC nonlinearity (<0.05% over full range) and spatial distortion map derived from precision pinhole array imaging.