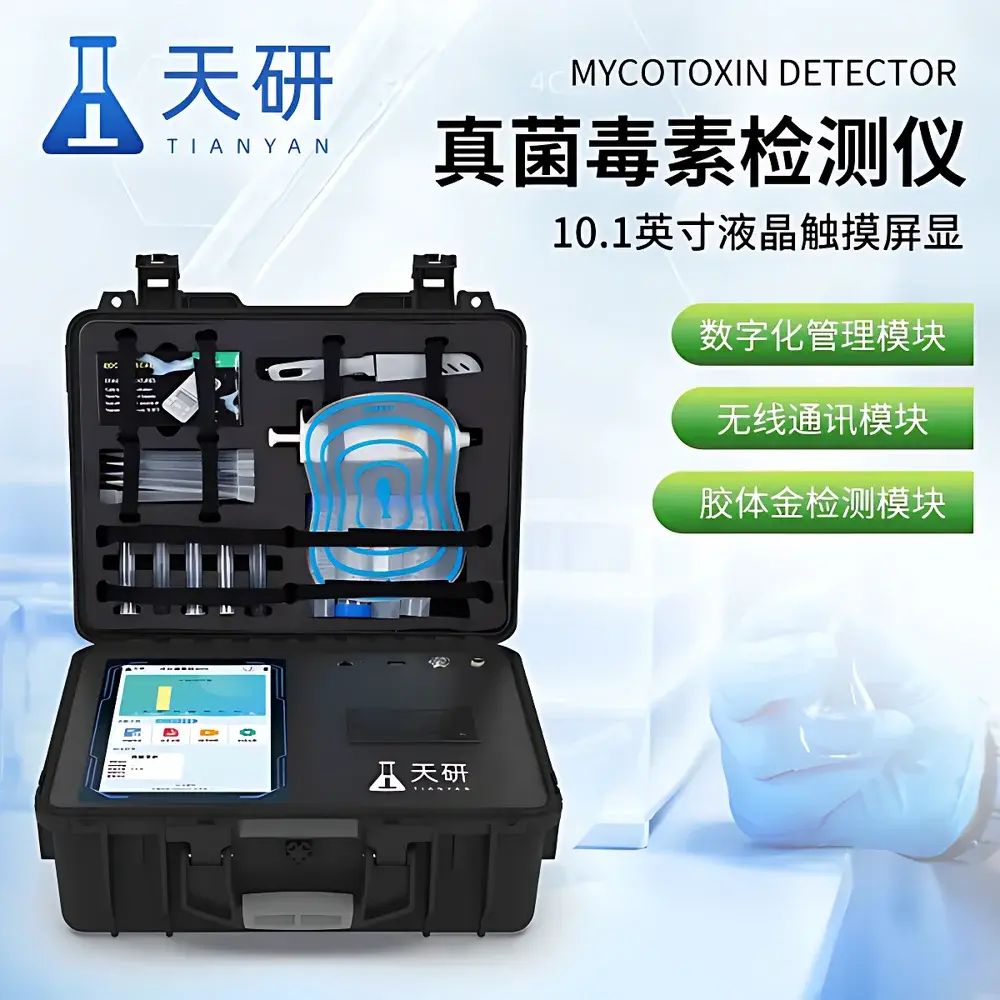
Tianyan TY-L01G Mycotoxin Rapid Quantitative Analyzer (Aflatoxin B1/M1/Total, Deoxynivalenol, Zearalenone, T-2 Toxin, Ochratoxin A, Fumonisin)
| Brand | Tianyan |
|---|---|
| Origin | Shandong, China |
| Manufacturer Type | Direct Manufacturer |
| Model | TY-L01G |
| Detection Principle | Competitive Inhibition Immunoassay with Colloidal Gold Lateral Flow |
| Sample Throughput | Up to 12 tests per run |
| Display | 10.1-inch capacitive touchscreen (Android OS) |
| Processor | ARM Cortex-A7 RK3288, quad-core @ 1.88 GHz |
| Power Supply | AC/DC adapter + built-in 6 Ah Li-ion battery (field-deployable) |
| Dimensions | 43 × 35 × 20 cm |
| Net Weight | 5.1 kg |
| Data Storage | >200,000 records (local USB export to Excel) |
| Connectivity | Wi-Fi, RJ45 Ethernet, optional A4 thermal printer interface |
| Compliance Support | FDA 21 CFR Part 11–ready audit trail (user login, permission tiers, calibration lock), GLP/GMP-aligned data management |
Overview
The Tianyan TY-L01G Mycotoxin Rapid Quantitative Analyzer is an integrated, field-deployable immunoassay platform engineered for precise, quantitative detection of major mycotoxins in food and feed matrices. It operates on the principle of competitive inhibition immunoassay using colloidal gold-labeled antibodies immobilized on lateral flow test strips. When a sample extract is applied, target mycotoxins (e.g., aflatoxin B1, deoxynivalenol, zearalenone) compete with conjugated antigen analogs for binding sites on immobilized antibodies. Signal intensity at the test (T) and control (C) lines—captured via high-resolution optical scanning—is inversely proportional to analyte concentration. The instrument’s embedded algorithm converts raw optical density ratios into quantitative results (µg/kg or ppb) calibrated against certified reference standards traceable to NIST-maintained materials. Designed for regulatory-grade screening, it meets the analytical performance expectations outlined in ISO/IEC 17025–accredited laboratories for rapid triage prior to confirmatory LC-MS/MS analysis.
Key Features
- 10.1-inch Android-based touchscreen interface with intuitive navigation and multilingual UI support (English, Spanish, Arabic, Vietnamese preloaded)
- Dual-mode operation: standalone field use (6 Ah rechargeable Li-ion battery, 8+ hours continuous runtime) or lab-integrated deployment (AC power + Ethernet/Wi-Fi connectivity)
- Automated track-driven colloidal gold strip handling: real-time image capture, auto-alignment, CT-line recognition, and dynamic threshold adjustment without manual intervention
- Universal strip compatibility: validated with third-party CE-marked and AOAC-RI-certified lateral flow assays—no vendor lock-in for consumables
- On-device data intelligence: weekly/monthly statistical dashboards with bar charts, pass/fail rate summaries, and trend visualization—no PC required
- Pre-configurable test templates: batch sample metadata (sample ID, matrix type, testing protocol, submitting unit) stored and recalled with one tap
- Robust data governance: role-based user authentication (admin/operator/auditor tiers), encrypted local storage, and tamper-evident audit logs compliant with GLP documentation requirements
- Firmware-over-the-air (FOTA): secure remote updates per instrument ID; no physical return or downtime required for method expansion or security patches
Sample Compatibility & Compliance
The TY-L01G supports extraction-ready samples from cereals (corn, wheat, barley, rice, bran), oilseeds (soybean meal, peanut cake), finished feeds, edible oils, and seasonings. Extraction protocols follow AOAC Official Method 2005.02 (aflatoxins), EN 14123 (DON), and ISO 15882 (zearalenone), ensuring interlaboratory reproducibility. Instrument output reports include mandatory fields for regulatory submission: analyst ID, calibration date, lot numbers of test strips and extraction reagents, limit of quantitation (LOQ) per analyte, and uncertainty estimation per ISO/IEC Guide 98-3. Data export formats comply with EFSA’s Data Collection Framework (DCF) and USDA-FDA Food Safety Modernization Act (FSMA) recordkeeping mandates.
Software & Data Management
The embedded Android OS hosts a purpose-built LIMS-lite application supporting full traceability from sample intake to report generation. All results are timestamped, digitally signed, and stored with immutable metadata—including environmental temperature/humidity during testing. Data sync occurs automatically to cloud-hosted or on-premise regulatory platforms via TLS 1.2–encrypted Wi-Fi or wired Ethernet. Optional A4-report printing includes QR-coded result verification, chain-of-custody fields, and compliance headers aligned with GB 2761–2023 (China), EC No. 1881/2006 (EU), and FDA Action Levels. USB export generates ISO-standard CSV/Excel files compatible with SAS JMP, Minitab, and enterprise SAP QM modules.
Applications
This analyzer serves as a frontline tool across the agri-food value chain: grain elevators perform inbound dock-side screening to reject contaminated lots before unloading; feed mills conduct in-process QC on raw ingredients and finished mash; dairy processors monitor milk for aflatoxin M1 carryover; regulatory agencies execute market surveillance under national residue monitoring plans (NRMPs); and contract labs deploy mobile units for farm-gate audits. Its portability and battery operation make it suitable for USDA APHIS border inspection points, FAO emergency response deployments, and World Food Programme (WFP) field assessments in low-infrastructure regions.
FAQ
What mycotoxins does the TY-L01G quantitatively measure?
It delivers quantitative results for aflatoxin B1, B2, G1, G2, M1, and total aflatoxins; deoxynivalenol (DON); zearalenone (ZEN); T-2 toxin; ochratoxin A (OTA); and fumonisins B1/B2—all within a single software environment.
Is method validation documentation available?
Yes: Tianyan provides instrument-specific validation reports per ICH Q2(R2), including precision (intra-/inter-day CV ≤12%), accuracy (recovery 85–115%), linearity (R² ≥0.992), and specificity data against cross-reactive analogs.
Can the device integrate with existing laboratory information systems (LIS)?
It supports HL7 v2.5.1 and ASTM E1384-compliant data exchange via configurable API endpoints or flat-file FTP push—no proprietary middleware required.
How is calibration maintained across extended field use?
Each test strip batch includes reference controls; the system auto-validates optical baseline before every run. Full calibration verification is performed quarterly using NIST-traceable mycotoxin reference standards (SRM 2371, SRM 2372).
Does the instrument meet international regulatory data integrity requirements?
Yes: audit trails record all user actions (login/logout, result edits, calibration events), enforce electronic signatures per 21 CFR Part 11 Subpart B, and prevent deletion of raw image files or intermediate calculations.