TRANSCEND VIVOSCOPE VISIONSCAN-100 Inverted Two-Photon Microscope
| Brand | TRANSCEND VIVOSCOPE |
|---|---|
| Origin | Beijing, China |
| Manufacturer Type | Authorized Distributor |
| Country of Origin | PRC |
| Model | VISIONSCAN-100 |
| Pricing | Upon Request |
| Excitation Wavelength Range | 680–1300 nm |
| Detection Channels | Dual-channel non-descanned detection (NDD) for autofluorescence & SHG |
| Z-Step Resolution | ≤ 0.1 µm (motorized piezo stage) |
| Max Imaging Depth | ≥ 500 µm in scattering tissue |
| XY Field of View | 1.2 mm × 1.2 mm (at 16× objective) |
| Stack Interval | 6.35 µm (user-configurable) |
| Objective Compatibility | 16×/0.8 NA water-dipping, 25×/1.05 NA water-immersion, and 40×/0.8 NA dipping objectives |
Overview
The TRANSCEND VIVOSCOPE VISIONSCAN-100 is an inverted two-photon microscope engineered for high-fidelity, label-free in vivo and ex vivo imaging of thick biological specimens. It operates on the principle of near-infrared (NIR) pulsed laser excitation—typically within 680–1300 nm—to induce simultaneous absorption of two photons, enabling intrinsic contrast generation via endogenous fluorophores (e.g., NAD(P)H, FAD, collagen, elastin) and second-harmonic generation (SHG) from non-centrosymmetric structures such as fibrillar collagen and myosin. Its inverted optical architecture supports stable immersion-based imaging of live tissues, organoids, freshly excised specimens, cryosections, and unstained histological slides—eliminating the need for chemical fixation or exogenous labeling. Designed for reproducible deep-tissue optical sectioning, the system delivers subcellular resolution across depths exceeding 500 µm in highly scattering biological matrices, making it suitable for longitudinal studies requiring minimal phototoxicity and photobleaching.
Key Features
- Inverted platform with motorized XYZ translation and high-stability piezo-driven Z-scanning (≤ 0.1 µm step resolution), optimized for long-term time-lapse imaging of dynamic biological preparations.
- Dual-channel non-descanned detection (NDD) path with spectral separation for simultaneous acquisition of autofluorescence (AF) and second-harmonic generation (SHG) signals—enabling structural-functional correlation without spectral crosstalk.
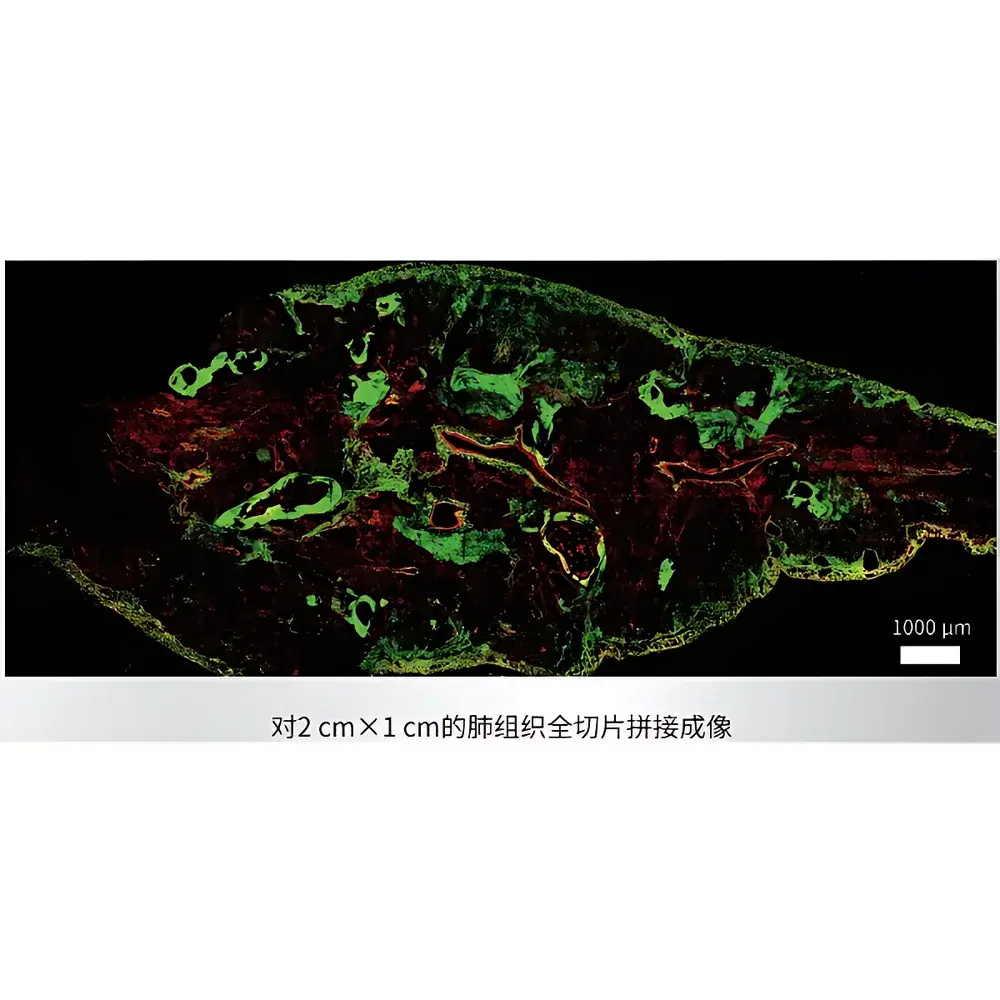
- Configurable stack interval (default 6.35 µm) and automated 2D/3D tile scanning with sub-pixel stitching accuracy, supporting large-area mosaic reconstruction of whole-tissue sections or organoid arrays.
- Integrated compatibility with standard water-dipping and water-immersion objectives (16×/0.8 NA, 25×/1.05 NA, 40×/0.8 NA), facilitating flexible magnification and working distance selection for diverse sample geometries—from intact explants to microfluidic chamber-mounted cultures.
- Modular laser interface accommodating tunable femtosecond sources (e.g., Chameleon Vision S, Mai Tai DeepSee), ensuring optimal excitation wavelength selection per endogenous contrast mechanism and tissue type.
Sample Compatibility & Compliance
The VISIONSCAN-100 is validated for imaging unprocessed biological specimens including fresh surgical resections, OCT-embedded frozen sections, unstained paraffin-removed “white slides”, and 3D organoid cultures maintained in Matrigel or synthetic hydrogels. Its NIR excitation minimizes absorption by hemoglobin and water, reducing thermal damage and enabling extended acquisition windows under physiological conditions. The system adheres to ISO 13485-aligned manufacturing practices for medical research instrumentation and supports audit-ready documentation workflows compliant with GLP and preclinical GMP requirements. While not a diagnostic device, its imaging outputs are compatible with digital pathology pipelines certified under CAP/CLIA guidelines when integrated with validated analysis software.
Software & Data Management
Acquisition and reconstruction are managed via VIVOVIEW™ v3.x, a MATLAB-based platform offering real-time preview, multi-channel registration, drift correction, and batch-processing of tiled Z-stacks. Raw data is saved in standardized TIFF or OME-TIFF formats with embedded metadata (objective ID, laser power, PMT gain, z-step, timestamp), ensuring FAIR (Findable, Accessible, Interoperable, Reusable) data principles. Export modules support direct import into Imaris, Arivis Vision4D, and QuPath for quantitative morphometry, collagen fiber orientation analysis (via SHG tensor modeling), and spatial phenotyping. Audit trail logging meets FDA 21 CFR Part 11 requirements for electronic records and signatures in regulated environments.
Applications
- Label-free histopathology validation: Correlative imaging of unstained tissue sections alongside conventional H&E staining to assess nuclear morphology, stromal architecture, and collagen distribution—supporting development of AI-assisted digital pathology classifiers.
- Organoid developmental dynamics: Time-resolved 3D mapping of metabolic activity (NAD(P)H/FAD ratio) and extracellular matrix remodeling (SHG intensity & anisotropy) during differentiation and maturation of patient-derived organoids.
- Intravital tissue monitoring: Longitudinal tracking of immune cell infiltration, vascular permeability, and fibrotic progression in murine models using cranial window or dorsal skinfold chamber preparations.
- Neuroscience structural imaging: High-resolution visualization of dendritic spines, microglial surveillance, and amyloid plaque deposition in cleared brain slices or thinned-skull preparations.
FAQ
Does the VISIONSCAN-100 require fluorescent labeling?
No—it is specifically designed for label-free imaging using endogenous contrast mechanisms: autofluorescence from metabolic co-factors and SHG from structural proteins.
Can it image through intact skull or skin?
Yes—when paired with appropriate NIR excitation wavelengths and high-NA water-dipping objectives, it achieves functional imaging depth up to 500 µm in scattering tissue, including thinned-skull preparations.
Is Z-stack interval fixed at 6.35 µm?
No—the 6.35 µm value reflects a typical setting for organoid imaging; users may configure intervals from 0.2 µm to 50 µm depending on Nyquist sampling requirements and specimen thickness.
What regulatory standards does the system support?
It complies with IEC 61000-6-3 (EMC) and IEC 60601-1 (safety); software features audit trails and electronic signature support aligned with 21 CFR Part 11 for regulated preclinical research.
Is remote operation supported?
Yes—VIVOVIEW™ includes secure network-enabled control and real-time streaming capabilities for collaborative multi-site experiments and core facility shared-access models.