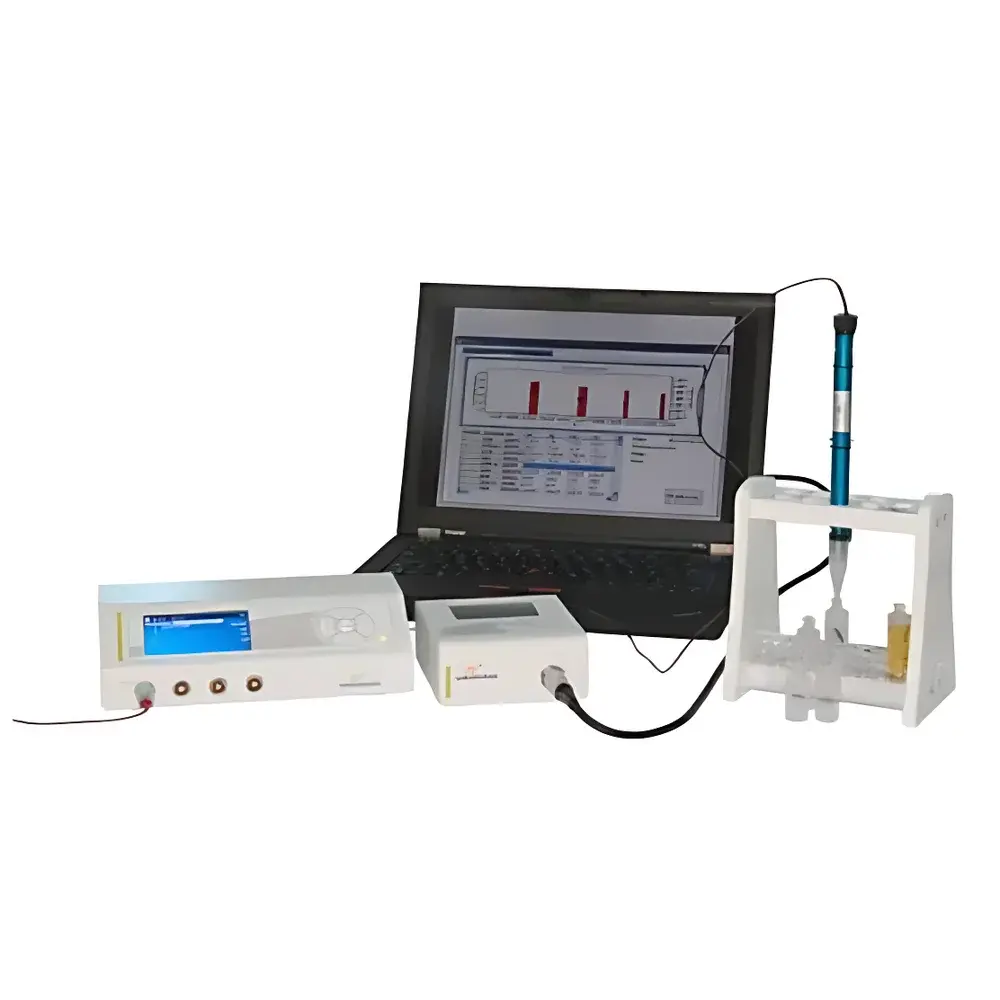
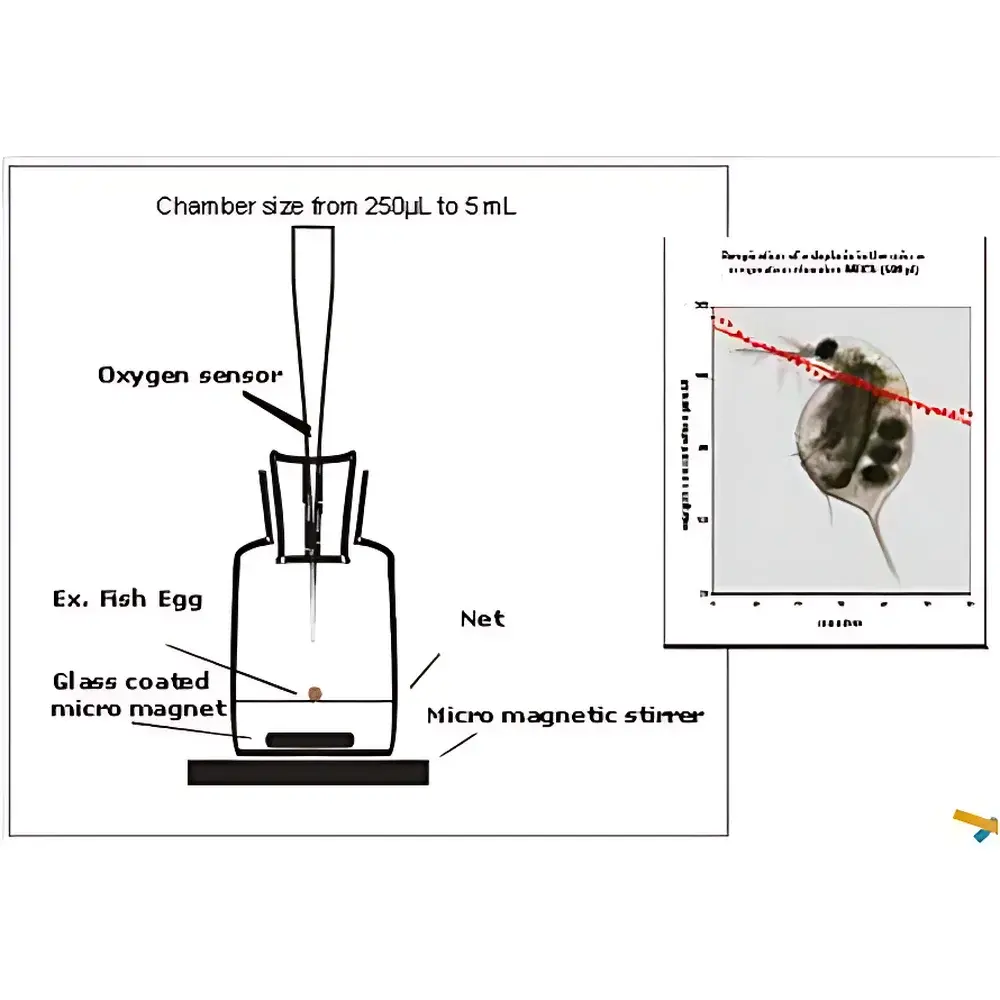
Unisense microrespiration System
| Brand | Unisense |
|---|---|
| Origin | Denmark |
| Model | microrespiration |
| Detection Principle | Amperometric Microsensor-Based Closed-Circuit Respirometry |
| Measurement Range (O₂) | 0–500 µM |
| Current Resolution | 10⁻¹² A |
| Detection Limits | O₂: 0.3 µM |
| NO | 20–30 nM |
| Sample Volume Capacity | 0.5–8 mL (dual-port microrespiration vials) |
| Electrode Compatibility | O₂, H₂, H₂S, NO, N₂O, pH, Redox, Temperature, Flow, Diffusivity, NO₂⁻, NO₃⁻ |
| Signal Processing | Low-Noise Analog Amplification + 24-bit ADC Conversion |
| Compliance | GLP-Compatible Data Acquisition Architecture |
Overview
The Unisense microrespiration System is a high-sensitivity, closed-circuit amperometric respirometry platform engineered for quantitative real-time monitoring of physiological and biochemical oxygen consumption and metabolite dynamics in minute biological samples. Operating on the principle of microsensor-based electrochemical detection, the system measures faradaic current generated by selective redox reactions at the tip of miniaturized Clark-type or specialized gas-selective microelectrodes (e.g., O₂, H₂S, NO), enabling non-invasive, continuous, and label-free tracking of respiratory fluxes and intermediary metabolite kinetics. Designed specifically for low-volume, high-value samples—such as single-cell suspensions, isolated mitochondria, microalgae cultures, fish larvae, plant embryos, and enzymatic reaction mixtures—the microrespiration System delivers nanomolar-per-hour resolution in metabolic rate quantification (e.g., O₂ uptake: down to <1 nM·L⁻¹·h⁻¹), with sub-picoampere current detection stability (10⁻¹² A). Its architecture supports both static incubation and dynamic perturbation experiments, making it suitable for mechanistic studies of electron transport chain function, substrate utilization efficiency, inhibitor response profiling, and discovery of novel anaerobic or alternative respiratory pathways.
Key Features
- Modular three-subsystem design: electrochemical sensor array, ultra-low-noise signal conditioning unit, and precision-engineered microrespiration chamber assembly.
- Multi-analyte capability: simultaneous or sequential measurement of O₂, H₂, H₂S, NO, N₂O, pH, redox potential, temperature, flow velocity, diffusivity, nitrite (NO₂⁻), and nitrate (NO₃⁻) using interchangeable microsensors.
- High-fidelity analog signal amplification with 24-bit analog-to-digital conversion, preserving signal integrity across six orders of magnitude in current output.
- Dual-port microrespiration vials (0.5 mL, 1 mL, 2 mL, 4 mL, and 8 mL volumes) featuring integrated O-rings, magnetic stir bar compatibility, and rigid aluminum-alloy mounting brackets for vibration-damped operation.
- On-the-fly reagent injection via calibrated microsyringe ports—enabling controlled addition of substrates (e.g., succinate, glucose), inhibitors (e.g., rotenone, antimycin A), cofactors, or chelators without system interruption.
- Single-electrode multiplexing capability: one calibrated microsensor can be sequentially deployed across multiple vials using an automated electrode positioning stage (optional accessory).
Sample Compatibility & Compliance
The microrespiration System accommodates diverse biological and biochemical matrices including aqueous suspensions, microbial biofilms, embryonic tissues, excised plant organs, isolated organelles, purified enzyme preparations, and synthetic chemical reaction systems. All hardware components meet IP54 environmental rating standards for laboratory use, and the system’s firmware and data acquisition software comply with GLP documentation requirements—including user-accessible audit trails, electronic signature support, and time-stamped raw data export (CSV, HDF5). While not certified for clinical or IVD use, the platform aligns with ASTM D5210 (standard test method for determining aerobic biodegradation of plastic materials), ISO 8692 (freshwater algal growth inhibition test), and USP <1058> analytical instrument qualification guidelines for research-grade instrumentation.
Software & Data Management
Control and analysis are performed via Unisense’s SensorTrace software suite (v6.5+), a Windows-based application supporting real-time visualization, multi-channel synchronized logging, kinetic curve fitting (Michaelis-Menten, exponential decay, linear regression), and customizable report generation. Raw current-time datasets are stored with full metadata (sensor ID, calibration date, temperature, vial volume, stirring speed), ensuring traceability per FDA 21 CFR Part 11 principles when configured with network authentication and role-based access control. Export formats include ASCII-delimited tables compatible with MATLAB, Python (Pandas), GraphPad Prism, and R for advanced statistical modeling and multivariate metabolic flux analysis.
Applications
- Quantitative mitochondrial respirometry in permeabilized cells and isolated organelles under varying substrate-uncoupler-inhibitor titration (SUIT) protocols.
- Microbial sulfur metabolism studies—real-time H₂S production kinetics during sulfate reduction or elemental sulfur disproportionation.
- NO-mediated signaling dynamics in plant root tips or macrophage cultures under hypoxic stress.
- Early-life-stage toxicology screening: O₂ consumption rate shifts in zebrafish embryos exposed to nanomaterials or endocrine disruptors.
- Photobioreactor optimization: dark-respiration rates and light-dark transition kinetics in Chlamydomonas reinhardtii cultures.
- Enzyme kinetics of terminal oxidases (e.g., cytochrome c oxidase, bd-type quinol oxidases) in reconstituted liposome systems.
FAQ
What sample volumes are supported?
The system supports dual-port microrespiration vials ranging from 0.5 mL to 8 mL, each designed for precise gas-tight sealing and uniform magnetic stirring.
Can the same electrode measure multiple analytes?
No—each microsensor is chemically and physically optimized for a specific analyte (e.g., O₂ vs. NO); however, rapid electrode exchange (<30 s) and auto-recalibration routines minimize experimental downtime.
Is temperature control integrated into the system?
The microrespiration chamber does not include active Peltier heating/cooling; external water-jacketed blocks or incubator integration is recommended for experiments requiring strict thermal regulation (±0.1 °C).
How is calibration performed?
Two-point electrochemical calibration is conducted in vitro using defined standards (e.g., air-saturated and sodium dithionite-deoxygenated buffers for O₂; NO-saturated saline for NO), with slope and offset validated prior to each experimental run.
Does the system support long-term unattended measurements?
Yes—continuous acquisition up to 72 hours is routinely achieved with stable power supply, temperature-stabilized environment, and periodic drift correction enabled via internal reference channel monitoring.