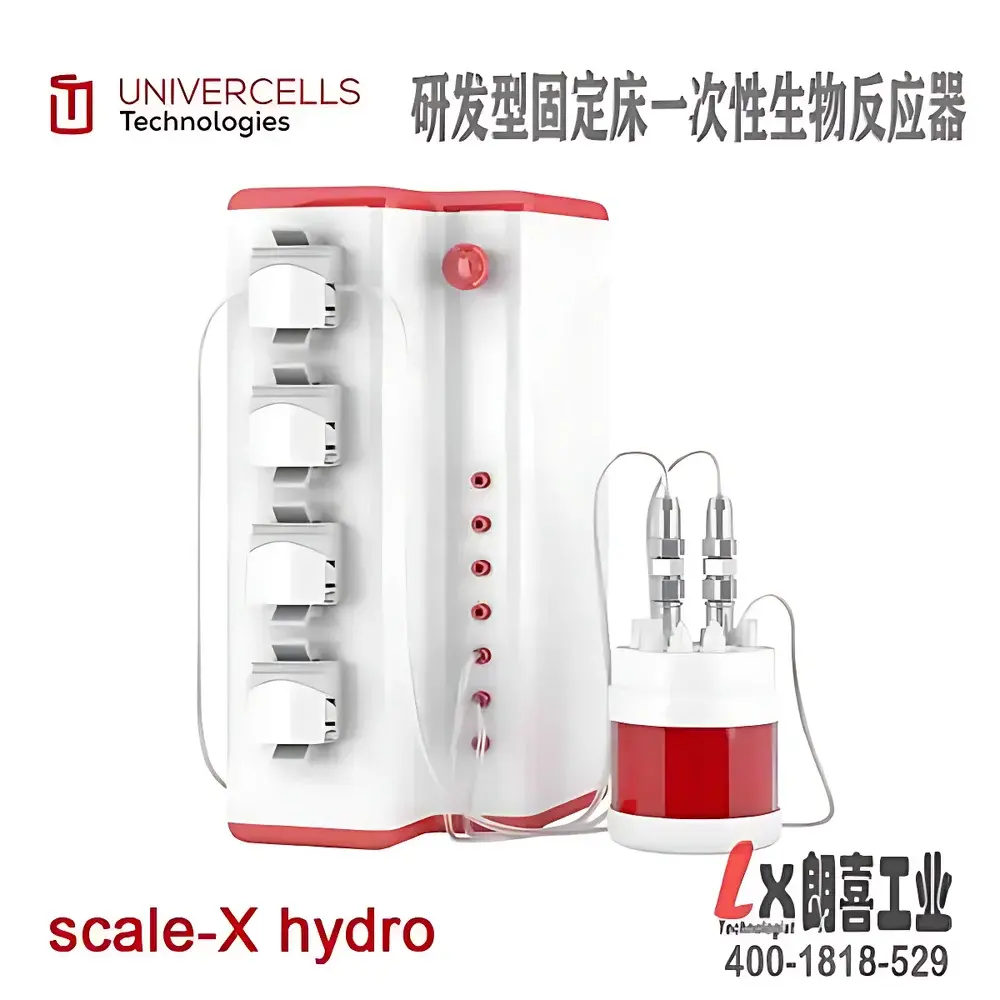
Univercells Scale-X™ hydro Fixed-Bed Bioreactor
| Brand | Univercells Technologies (Belgium) |
|---|---|
| Origin | Belgium |
| Model | Scale-X™ hydro |
| Growth Surface Area | 2.4 m² |
| Configuration | Single-layer fixed-bed module |
| Construction Materials | Structured spiral-wound PET mesh and medical-grade silicone tubing |
| Sterilization | Pre-sterilized single-use disposable cartridge |
| Temperature Control | Ambient to 37 °C (external incubator required) |
| Application Scope | Adherent and suspension cell culture for R&D-scale vaccine and viral vector process development |
| Compliance | Designed for GLP-compliant process characterization |
Overview
The Univercells Scale-X™ hydro Fixed-Bed Bioreactor is an engineered single-use platform for scalable, reproducible adherent cell culture in early-stage bioprocess development—particularly for viral vaccine and oncolytic virus manufacturing. Unlike conventional stirred-tank reactors (STRs) or microcarrier-based systems, the Scale-X™ hydro employs a patented structured spiral-wound polyethylene terephthalate (PET) fixed bed, providing 2.4 m² of high-efficiency growth surface within a compact footprint (~20 L total system volume). Its hydrodynamic design ensures uniform laminar flow across the entire bed depth and circumference, eliminating dead zones and promoting homogeneous cell attachment and proliferation. This architecture enables consistent cell densities up to 2.7 × 10⁵ cells/cm²—comparable to or exceeding those achieved in optimized microcarrier STRs—while reducing reactor volume requirements by 5× at equivalent output. The system operates without internal agitation or sparging, minimizing shear stress and enabling serum-free, perfusion-compatible cultivation of sensitive lines such as Vero, MRC-5, and CHO-S.
Key Features
- Patented spiral-wound PET fixed bed: Engineered porosity and mechanical resilience prevent local compaction and ensure full utilization of internal surface area.
- Single-use, pre-sterilized configuration: Eliminates cleaning-in-place (CIP) and sterilization-in-place (SIP) validation burden; reduces cross-contamination risk between campaigns.
- Modular scalability pathway: Direct linear scale-up from Scale-X™ hydro (2.4 m², R&D) → Scale-X™ carbo (10/30 m², clinical manufacturing) → Scale-X™ nitro (200/600 m², commercial production).
- Integrated sampling architecture: Twelve standardized 2.15 cm² biopsy strips allow spatially resolved cell density quantification without bed disruption.
- Low-shear perfusion interface: Compatible with external peristaltic or diaphragm pumps; supports continuous medium exchange and metabolite control.
- Incubator-integrated operation: Designed for placement inside standard CO₂ incubators (37 °C, 5% CO₂); pH controlled via NaHCO₃ or NaOH titration; DO maintained via top-space gas exchange.
Sample Compatibility & Compliance
The Scale-X™ hydro supports both anchorage-dependent (e.g., Vero, MDCK, HEK293) and suspension-adapted cell lines under serum-free or low-serum conditions. It has been validated for use with VP-SFM, EX-CELL® Vero, and other chemically defined media formulations. All wetted components comply with USP Class VI and ISO 10993-5 biological safety standards. The system’s single-use nature aligns with ICH Q5A(R2) guidance on viral clearance validation and facilitates rapid changeover between product campaigns—critical for multi-product facilities. While the base hardware does not include embedded sensors, it interfaces with third-party monitoring systems compliant with 21 CFR Part 11 when paired with validated data acquisition software. Process documentation meets GLP requirements for preclinical batch records and comparability studies.
Software & Data Management
The Scale-X™ hydro operates as a hardware platform requiring external control instrumentation. Users typically integrate it with programmable peristaltic pumps, pH/DO transmitters, and temperature loggers connected to SCADA or LIMS platforms. Univercells provides detailed SOP templates and raw data export protocols (CSV/Excel) for audit-ready reporting. When deployed within the NevoLine™ integrated continuous biomanufacturing platform, the Scale-X™ hydro connects to Univercells’ proprietary control software—enabling automated perfusion scheduling, alarm-triggered intervention logs, and electronic batch record (EBR) generation compliant with Annex 11 and ALCOA+ principles. Audit trails capture user actions, parameter changes, and timestamped sensor readings, supporting regulatory submissions under EMA and FDA review pathways.
Applications
- Vaccine process development: High-density Vero culture for rabies, influenza, and Ebola virus vector production.
- Oncolytic virus manufacturing: Scalable expansion of tumor-selective viruses (e.g., adenovirus, vaccinia) with minimal passage-induced attenuation.
- Cell therapy upstream support: Feeder-layer-free expansion of primary human cells (e.g., mesenchymal stromal cells) under xeno-free conditions.
- Process intensification studies: Side-by-side comparison of fixed-bed vs. microcarrier kinetics, nutrient consumption profiles, and metabolic waste accumulation.
- Platform technology transfer: Bridging lab-scale data to clinical GMP batches using identical bed geometry and flow dynamics across scale-X™ variants.
FAQ
Is the Scale-X™ hydro suitable for suspension cell lines?
Yes—it supports both adherent and suspension-adapted cell lines, including CHO-S and HEK293, when cultured in appropriate microcarrier-free media formulations.
What is the maximum recommended operating pressure across the fixed bed?
The PET matrix is rated for ≤0.5 bar differential pressure; typical perfusion rates range from 0.1–0.5 mL/min/cm² bed surface area.
Can the fixed bed be reused after sterilization?
No—Scale-X™ hydro is strictly a single-use system; the PET/silicone assembly is gamma-irradiated and supplied pre-sterilized.
How is pH controlled without internal electrodes?
pH is monitored externally via inline or grab-sample analysis, with correction achieved through automated NaOH or NaHCO₃ dosing into the recirculation loop.
Does Univercells provide protocol support for Vero cell seeding and harvest?
Yes—comprehensive technical notes, including inoculum density optimization (0.5–2.0 × 10⁴ cells/cm²), trypsinization timing, and harvest yield estimation, are available under NDA.