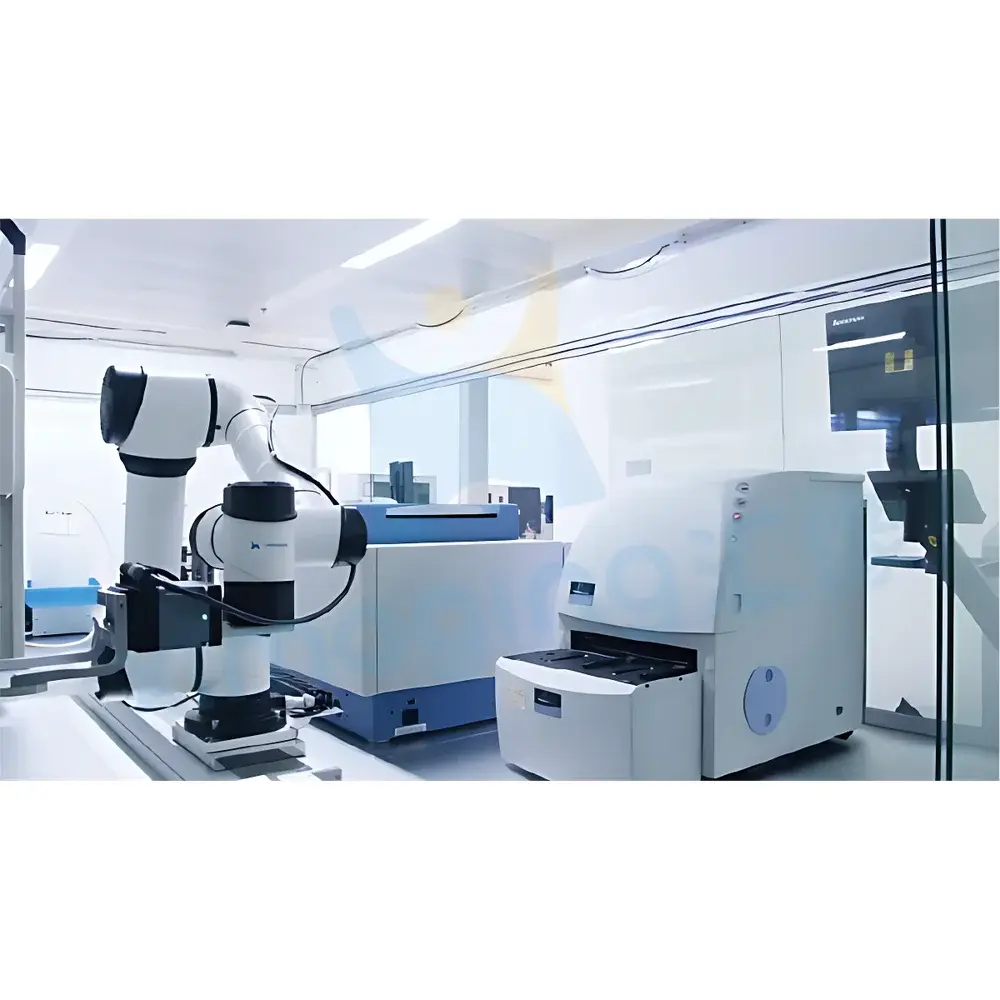
X-imaging Smart Automated Drug Screening System
| Brand | X-imaging |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | OEM Manufacturer |
| Product Category | Domestic |
| Model | Smart Automated Drug Screening System |
| Pricing | Upon Request |
Overview
The X-imaging Smart Automated Drug Screening System is a fully integrated, benchtop-scale laboratory automation platform engineered for precision execution of cell-based and biochemical high-throughput screening (HTS) workflows. Built upon Couette-flow–compatible liquid handling architecture and modular robotic orchestration, the system implements standardized microplate assay protocols—including compound plating, cell seeding, incubation, reagent addition, endpoint or kinetic detection (e.g., luminescence, fluorescence, absorbance), and primary data aggregation—within a single, closed-loop environment. Its core design adheres to fundamental HTS principles defined by the Society for Biomolecular Screening (SBS) and aligns with operational expectations for assay miniaturization (96- to 384-well formats), volumetric accuracy at nanoliter-to-microliter scales, and reproducible biological readout generation under controlled environmental conditions.
Key Features
- High-Throughput Microplate Compatibility: Supports standardized SBS/ANSI-compliant 96-well and 384-well plates; enables precise liquid dispensing in 1–1000 nL increments using positive displacement pipetting modules calibrated per ISO 8655.
- Fully Automated Robotic Workflow Execution: Integrates a 6-axis collaborative robotic arm with programmable end-effectors, enabling unattended operation across ≥8 consecutive assay steps without manual intervention.
- Visual Drag-and-Drop Protocol Builder: Proprietary software interface allows non-programmers to construct, simulate, and validate assay sequences via intuitive graphical workflow assembly—supporting conditional logic, loop structures, and error-handling triggers.
- Controlled Environmental Integrity: Internal laminar airflow chamber maintains ISO Class 5 (Class 100) cleanroom-equivalent conditions; negative-pressure containment prevents cross-contamination between plate positions during multi-plate runs.
- Modular Hardware Architecture: Open mechanical and communication interfaces (RS-232, USB, TCP/IP, Modbus RTU) facilitate seamless integration with third-party instruments including plate readers (e.g., BMG CLARIOstar), incubators, centrifuges, and storage units.
Sample Compatibility & Compliance
The system accommodates diverse biological sample types—including adherent and suspension mammalian cell lines (e.g., HEK293, HeLa, U2OS), primary human cells, bacterial cultures, and purified enzyme/receptor preparations—across standard tissue-culture treated, low-binding, or assay-optimized microplates. All hardware modules comply with IEC 61000-6-2 (immunity) and IEC 61000-6-4 (emissions) electromagnetic compatibility standards. The platform supports audit-trail-enabled operation per FDA 21 CFR Part 11 requirements when deployed with validated software configuration and user access controls. Documentation packages include IQ/OQ templates aligned with GLP and GMP-aligned validation frameworks.
Software & Data Management
X-imaging’s proprietary ScreenFlow™ software provides unified control of hardware modules, real-time monitoring of run status, and native export of structured assay metadata (including timestamps, pipette calibration logs, environmental sensor readings, and plate map annotations) in MIAME- and MIAPE-compliant XML/CSV formats. Integrated data preprocessing tools support background subtraction, Z’-factor calculation, % inhibition/activation normalization, and hit identification thresholds. Raw output files are compatible with downstream analysis platforms including GraphPad Prism, Spotfire, and KNIME; API endpoints enable direct ingestion into LIMS or ELN systems via RESTful web services.
Applications
- Validation of target-based and phenotypic HTS assay models prior to library-wide screening
- Hit confirmation and dose–response characterization (IC50/EC50) of primary screening actives
- Cell viability, cytotoxicity, apoptosis, and pathway reporter assays (e.g., NF-κB, CREB, STAT)
- Enzyme kinetics and inhibitor profiling in biochemical formats (e.g., kinase, protease, dehydrogenase assays)
- Automated compound reformatting, cherry-picking, and serial dilution from source plates to assay plates
- Long-term kinetic imaging workflows when coupled with compatible inverted microscopes or on-stage incubators
FAQ
Does the system support remote monitoring and intervention during active runs?
Yes—ScreenFlow™ includes secure web-based dashboard access with live video feed (optional camera module), real-time sensor telemetry, and emergency pause/resume functionality via authenticated HTTPS session.
Can the platform be qualified for regulated environments such as clinical trial support labs?
Yes—when implemented with documented change control, periodic performance verification, and electronic signature-enabled user roles, the system meets baseline requirements for use in non-GCP research supporting IND-enabling studies.
What maintenance intervals and service coverage options are available?
Preventive maintenance is recommended every 6 months; X-imaging offers extended warranty, on-site service contracts, and remote diagnostics support with SLA-backed response times.
Is method transfer support provided for migrating existing manual or semi-automated assays?
Yes—X-imaging’s Application Support Team provides protocol conversion services, including liquid class optimization, tip compatibility assessment, and Z’-factor benchmarking against legacy workflows.