X-imaging Smart Mass Spectrometry Pre-analytical Automation System
| Brand | X-imaging |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Product Category | Domestic |
| Model | Smart Mass Spectrometry Pre-analytical Automation System |
| Pricing | Available upon Request |
Overview
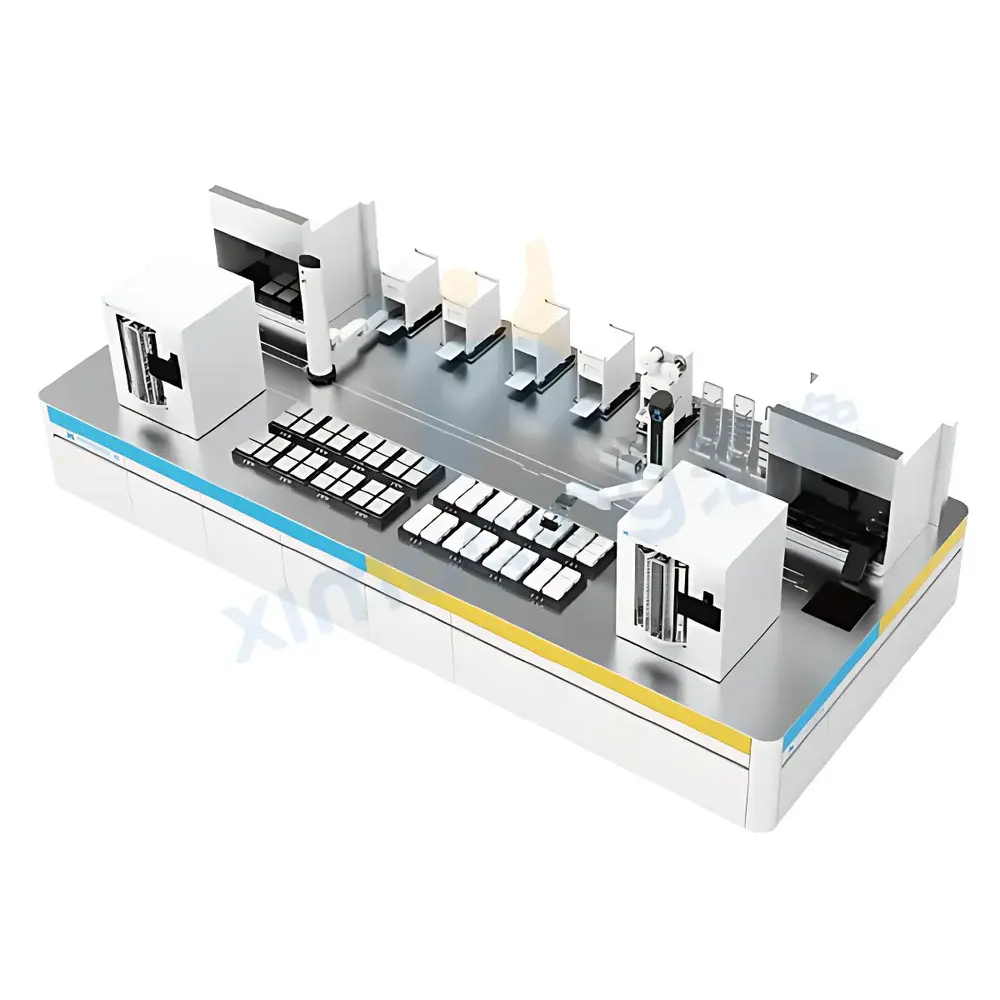
The X-imaging Smart Mass Spectrometry Pre-analytical Automation System is an integrated laboratory automation platform engineered for high-precision, reproducible sample preparation prior to mass spectrometric analysis. Designed specifically for complex biological matrices—including whole blood, plasma, serum, urine, tissue homogenates, and cultured cells—the system implements a modular liquid handling architecture grounded in positive displacement pipetting and temperature-controlled incubation modules. It executes sequential pre-analytical workflows including protein precipitation, solid-phase extraction (SPE), liquid–liquid extraction (LLE), enzymatic digestion, internal standard addition, quality control (QC) sample spiking, supernatant transfer, and MALDI target spotting—all under programmable, audit-trail-enabled protocols. Unlike generic liquid handlers, this system incorporates real-time process monitoring via integrated optical sensors and pressure feedback loops, enabling detection of clogged tips, insufficient aspiration volume, or incomplete phase separation—critical for maintaining analytical integrity in regulated environments.
Key Features
- Modular workflow configuration: Supports customizable sequences for proteomic, metabolomic, and small-molecule applications without hardware reconfiguration
- Positive-displacement pipetting with 0.5–1000 µL range and ≤2% CV at 10 µL, ensuring accuracy across viscous and volatile samples
- Integrated temperature-controlled zones: 4–45 °C incubation for enzymatic reactions and cold storage (4 °C) for labile analytes
- On-deck SPE/LLE cartridge handling with vacuum manifold and fraction collection capability
- Automated MALDI target spotting with adjustable droplet volume (0.2–2 µL), spot spacing (500–2000 µm), and drying profile control
- Embedded AI-assisted protocol optimization: Learns from historical run data to recommend optimal dwell times, mixing speeds, and elution gradients
- Full 21 CFR Part 11 compliance: Electronic signatures, role-based access control, and immutable audit trails for all instrument actions and parameter changes
Sample Compatibility & Compliance
The system accommodates primary clinical specimens (EDTA/K2EDTA plasma, heparinized whole blood, dried blood spots, cerebrospinal fluid) and processed extracts (e.g., derivatized amino acids, silylated sterols, acylated lipids). All fluidic pathways are chemically inert (PFA, ETFE, and ceramic components), minimizing adsorption of peptides, phospholipids, and metal-chelating analytes. The platform conforms to ISO/IEC 17025:2017 requirements for testing laboratories and supports method validation per CLSI EP26-A and USP <1225>. It enables full traceability from raw specimen tube barcode to final MS-ready plate, satisfying GLP/GMP documentation standards for clinical assay development and IVD manufacturing.
Software & Data Management
Controlled via X-SciX™ software—a validated, Windows-based application with dual-mode operation (guided wizard and advanced script editor). Protocols are stored as XML-based method files with embedded metadata (reagent lot numbers, calibration timestamps, operator ID). Raw execution logs include timestamped event records (e.g., “Tip wash cycle completed”, “Centrifuge door interlock engaged”) and sensor-derived metrics (aspiration pressure delta, tip seal verification status). Data export supports CSV, PDF e-records, and direct integration with LIMS via HL7 v2.5 or ASTM E1384 interfaces. Software updates undergo formal change control per FDA guidance on computerized systems used in clinical laboratories.
Applications
- Clinical metabolomics: Quantification of catecholamines, bile acids, steroid hormones, vitamins (B12, D, K), folate metabolites, and amino acid profiles in newborn screening and endocrine diagnostics
- Toxicology screening: Extraction and cleanup of heavy metals (As, Pb, Cd), volatile organics (ethanol, methanol, ethylene glycol), and biotoxins (aflatoxins, microcystins) from forensic and occupational specimens
- Therapeutic drug monitoring: Automated preparation of whole blood for tacrolimus, cyclosporine, and sirolimus quantification by LC-MS/MS; simultaneous elemental analysis of antimicrobial agents (e.g., vancomycin-bound Fe/Cu)
- Microbial proteotyping: High-throughput ribosomal protein extraction from Gram-positive/negative isolates for rapid MALDI-TOF identification and resistance marker profiling (e.g., β-lactamase expression)
- Lipidomics: Class-specific enrichment of glycosphingolipids, cardiolipins, and oxidized phospholipids from mitochondrial fractions for structural characterization
FAQ
Does the system support integration with third-party mass spectrometers?
Yes—it outputs standardized MALDI targets compatible with Bruker rapifleX, Sciex TOF 6600+, and Waters SYNAPT G2-Si platforms; LC-MS vial racks follow ANSI/SLAS footprint specifications.
Can it handle high-viscosity samples such as whole blood or tissue lysates?
Yes—positive-displacement pipetting combined with pre-wet and slow-aspirate routines ensures reliable handling of samples up to 15 cP viscosity without carryover or foaming.
Is method validation documentation provided?
X-imaging supplies IQ/OQ/PQ protocols, system suitability test scripts, and a comprehensive validation support package aligned with ISO 15197 and CLIA requirements.
What maintenance intervals are recommended?
Daily tip wash and calibration check; quarterly fluidic path inspection and pressure sensor recalibration; annual full performance verification using NIST-traceable reference standards.
How is cybersecurity managed in networked deployments?
The system operates in air-gapped mode by default; when connected, it complies with IEC 62443-3-3 and supports TLS 1.2+ encryption, certificate-based authentication, and periodic vulnerability scanning via integrated endpoint protection.