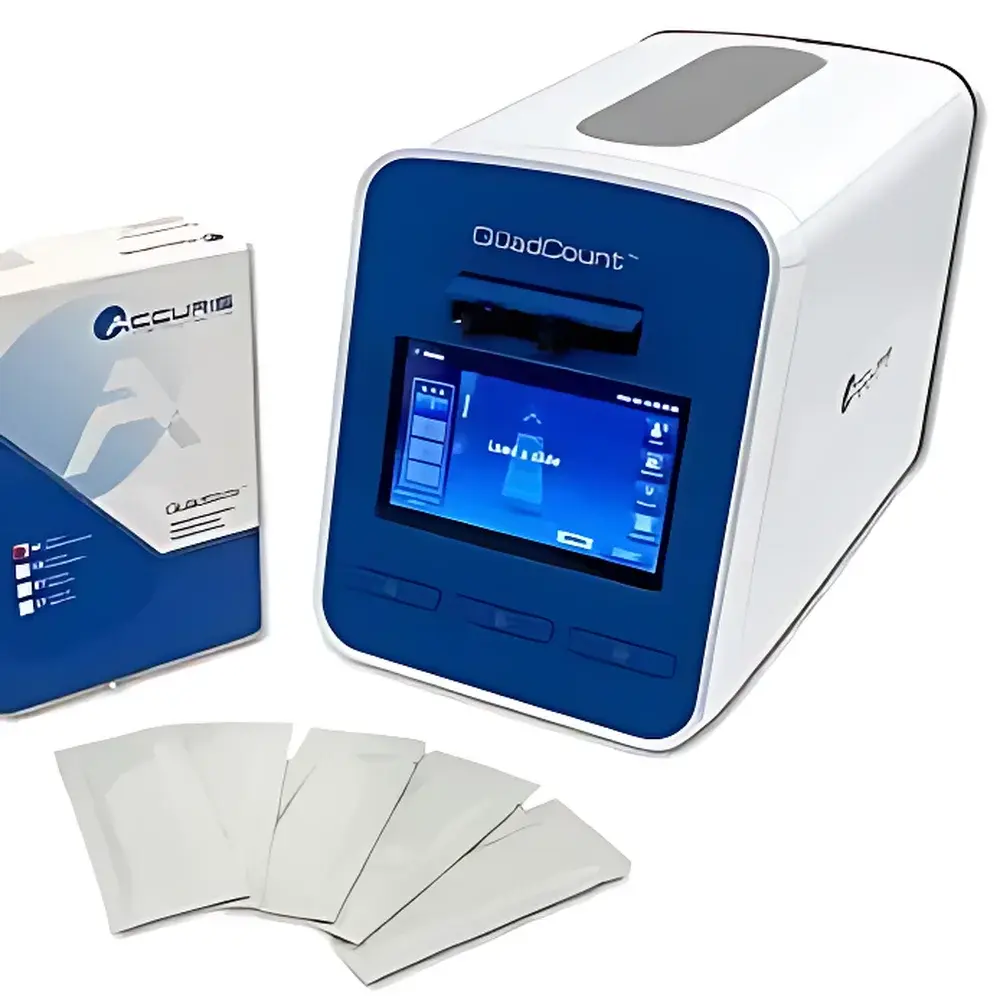
YUYANBIO FACSCOPE B Advanced Automated Cell Counter
| Brand | YUYANBIO |
|---|---|
| Origin | South Korea |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Import Status | Imported Instrument |
| Model | FACSCOPE B |
| Sample Volume | 20 µL per chamber (4 chambers per slide) |
| Cell Size Detection Range | 5–60 µm |
| Optical Magnification | 4× objective |
| Camera | 5 MP CMOS sensor (2592 × 1944 resolution) |
| Light Source | Green LED (50,000 h lifetime) |
| Counting Modes | Rapid (0.15 µL, 20 s), Standard (0.9 µL, 30 s), Precise (3.6 µL, 100 s) |
| Concentration Range | 1 × 10⁴ – 1 × 10⁷ cells/mL |
| Data Storage | 3 user profiles, 1000 datasets per user |
| Interface | USB 2.0 for printer, barcode scanner, keyboard, or USB drive |
| Report Output | Exportable PDF with histogram, viability analysis, and metadata |
| Dimensions | 16.3 × 29.3 × 21.6 cm |
| Weight | 5 kg |
| Power | 115 V / 230 V, 50–60 Hz |
Overview
The YUYANBIO FACSCOPE B is a high-precision, fully automated cell counter engineered for routine and research-grade cell viability and concentration analysis in life science laboratories. It employs brightfield microscopy coupled with green LED illumination and a high-fidelity 4× optical system to deliver consistent, objective quantification of mammalian, yeast, and bacterial suspensions. Unlike manual hemocytometer-based methods—prone to inter-operator variability and sampling bias—the FACSCOPE B integrates motorized XYZ stage control, auto-focus algorithms, and multi-frame image acquisition (up to 24 frames per sample) to ensure reproducible volumetric sampling across heterogeneous populations. Its core measurement principle relies on digital image segmentation and adaptive thresholding, calibrated against standardized particle size and contrast parameters, enabling accurate discrimination between viable and non-viable cells stained with trypan blue or similar dyes. Designed for GLP-compliant workflows, the instrument supports audit-ready data capture, traceable user sessions, and exportable reports aligned with ISO/IEC 17025 documentation requirements.
Key Features
- Motorized XYZ precision stage with micron-level positioning for repeatable focal plane alignment and multi-layer imaging
- High-resolution 5 MP CMOS camera (2592 × 1944 pixels) paired with achromatic 4× objective lens for optimal depth-of-field and edge definition
- Three validated counting modes: Rapid (20 s, 0.15 µL), Standard (30 s, 0.9 µL), and Precise (100 s, 3.6 µL), allowing method-specific throughput and statistical confidence
- Dedicated 4-chamber disposable counting slides with hydrophilic, grid-etched surfaces—each chamber independently calibrated to ISO 21501-4 particle sizing tolerances
- Adaptive image analysis engine capable of resolving clustered, irregularly shaped, or low-contrast cells without manual gating or threshold reconfiguration
- Real-time histogram generation by cell diameter (5–60 µm binning), enabling population distribution profiling and outlier detection
- On-device touchscreen interface with intuitive workflow navigation; no external PC required for operation or report generation
Sample Compatibility & Compliance
The FACSCOPE B accommodates a broad range of suspension-based samples including primary mammalian cells (e.g., PBMCs, CHO, HEK293), immortalized lines, yeast (S. cerevisiae), and prokaryotes (E. coli). It is compatible with standard trypan blue, propidium iodide, and acridine orange staining protocols. All counting slides are manufactured under ISO 13485-certified cleanroom conditions and sterilized via ethylene oxide. The instrument meets electromagnetic compatibility (EMC) standards per IEC 61326-1 and electrical safety requirements per IEC 61010-1. For regulated environments, firmware supports optional 21 CFR Part 11-compliant user authentication, electronic signatures, and immutable audit trails when paired with networked data management systems.
Software & Data Management
Embedded firmware provides full control over acquisition parameters—including adjustable size gates (5–60 µm), aggregation sensitivity, and viability classification thresholds—without requiring software installation. Each dataset includes timestamp, operator ID, slide lot number, and environmental metadata (ambient temperature/humidity if logged externally). Up to 3 user accounts can be configured with role-specific permissions. Data exports as password-protected PDF reports containing raw images, annotated cell masks, concentration results, coefficient of variation (CV%), histogram plots, and statistical summaries. USB 2.0 port enables direct export to flash drives or integration with LIMS via CSV-compatible batch reporting.
Applications
- Bioprocess monitoring: Daily viability assessment during upstream bioreactor runs and harvest point determination
- Cell therapy QC: Release testing of CAR-T, NK, and MSC products per USP <71> and ASTM E2877-22 guidelines
- Academic research: Longitudinal studies of cytotoxicity, proliferation kinetics, and transfection efficiency
- Vaccine development: Quantification of dendritic cell yields and activation status post-stimulation
- Stem cell banking: Pre-freeze/post-thaw recovery validation with CV% ≤ 5% across replicates
FAQ
What is the minimum and maximum cell concentration the FACSCOPE B can accurately quantify?
The instrument is validated for concentrations ranging from 1 × 10⁴ to 1 × 10⁷ cells/mL. Below 1 × 10⁴/mL, statistical uncertainty increases due to Poisson sampling limitations; above 1 × 10⁷/mL, dilution is recommended to maintain optimal cell dispersion and image segmentation fidelity.
Does the FACSCOPE B support dual-stain viability assays (e.g., AO/PI)?
No—the current optical configuration uses monochromatic green LED illumination optimized for trypan blue contrast enhancement. Dual-fluorescence capability requires excitation/emission filter sets not included in this model.
Are counting slides reusable or certified for sterility?
FACSCOPE counting slides are single-use, gamma-irradiated disposables supplied with certificate of conformance (CoC) and lot-specific dimensional verification reports.
Can historical data be migrated to a centralized laboratory database?
Yes—PDF reports and CSV-formatted summary files can be imported into ELN or LIMS platforms. Optional API integration is available under enterprise licensing agreements.
Is routine calibration required, and what maintenance does the system need?
The system performs self-diagnostic checks at startup. Annual verification using NIST-traceable polystyrene microsphere standards (10 µm ± 0.2 µm) is recommended for ISO/IEC 17025 accreditation. No consumable optics cleaning or alignment is required under normal operating conditions.