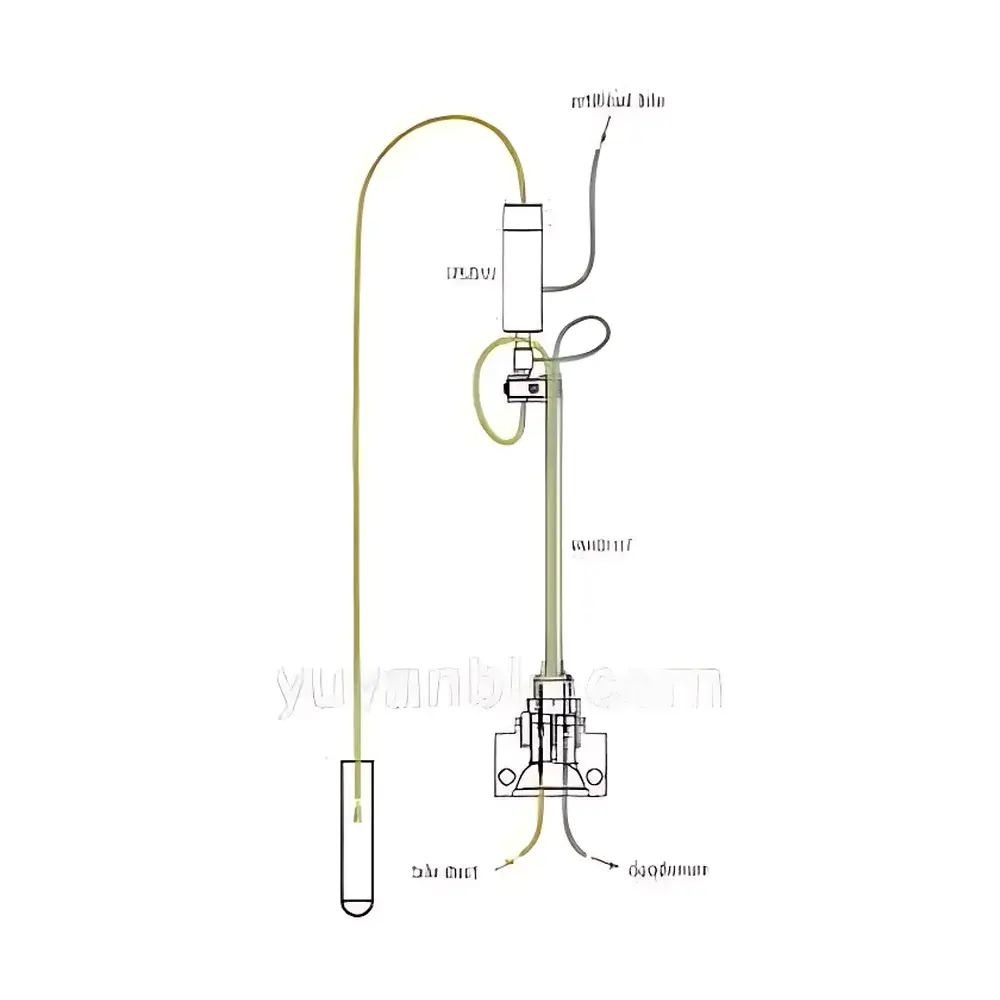
YUYANBIO Large-Animal Dosing Catheters
| Brand | YUYANBIO |
|---|---|
| Origin | USA |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Product Category | Imported Life Science Instrumentation |
| Model | Large-Animal Dosing Catheters |
| Price Range | USD 1,400 – 14,200 |
Overview
YUYANBIO Large-Animal Dosing Catheters are precision-engineered intravascular and extravascular access devices designed for chronic or acute administration, sampling, and physiological monitoring in preclinical large-animal models. These catheters operate on fundamental principles of fluid dynamics and biocompatible material science—leveraging controlled luminal geometry, optimized tip configurations (e.g., rounded, tapered, U-bend, or 90° angled), and pressure-balanced valve mechanisms to ensure reliable patency, minimal thrombogenicity, and hemodynamic stability during prolonged use. Engineered specifically for species including dogs, non-human primates (NHPs), pigs, sheep, rabbits, ferrets, and guinea pigs, each catheter system adheres to anatomical constraints and procedural requirements defined by veterinary surgical standards and GLP-compliant study protocols. The devices support both percutaneous and surgically implanted deployment strategies, with integrated features enabling compatibility with infusion pumps, syringe drivers, and closed-loop sampling systems.
Key Features
- Material construction using medical-grade polyurethane (PU) or silicone elastomer—selected for optimal balance of flexibility, kink resistance, and long-term biocompatibility.
- Luer-lock termination standard across all configurations to ensure secure, leak-free connections with syringes, extension sets, and infusion hardware.
- Species-specific dimensional optimization—including inner diameter (ID), outer diameter (OD), and length—to match vascular anatomy and minimize flow resistance or wall shear stress.
- Valve-integrated designs: Slit-valve occlusion for duodenal/gastric catheters; patented Groshong® valves (FDA-cleared human-grade) for central venous access; self-sealing septum membranes (e.g., BAD-Cath) for low-pressure bile duct or intraduodenal delivery.
- Customizable fixation elements: Suture beads, silicone patches (½-inch), antimicrobial cuff sleeves (VitaCuff™), and tissue ingrowth cuffs (SureCuff™) to enhance anchoring and reduce migration risk.
- Specialized tip geometries: Rounded tips for venous cannulation; 90° angled tips for ophthalmic delivery into anterior chamber or Tenon’s space; U-bend configurations for NHP limb mobility accommodation.
Sample Compatibility & Compliance
These catheters are validated for use in regulatory-grade pharmacokinetic (PK), toxicokinetic (TK), and ADME studies conducted under Good Laboratory Practice (GLP) frameworks. Material certifications include USP Class VI biological reactivity testing and ISO 10993-5 cytotoxicity compliance. All PU-based catheters feature internal polyethylene (PE) lining to mitigate chemical interaction with solvents, peptides, or small-molecule therapeutics. Devices intended for chronic implantation meet ASTM F748 guidelines for in vivo performance evaluation. The Groshong® and Broviac/Hickman® product lines are manufactured under ISO 13485-certified quality systems and carry FDA 510(k) clearance for human use—providing a high-confidence translational bridge for veterinary and comparative medicine applications.
Software & Data Management
While the catheters themselves are passive hardware components, they integrate seamlessly with industry-standard data acquisition and infusion control platforms—including Harvard Apparatus PHD Ultra, Kent Scientific Cheetah, and World Precision Instruments Micro4. When used in conjunction with pressure transducers, flow meters, or automated sampling modules (e.g., Tecan Freedom EVO), catheter systems enable time-stamped, audit-trail-enabled data capture compliant with FDA 21 CFR Part 11 requirements. Optional RFID-tagged packaging supports traceability across animal cohorts and study phases, aligning with electronic lab notebook (ELN) workflows and LIMS integration.
Applications
- Chronic intravenous dosing and serial blood sampling in dogs, NHPs, and swine over durations up to 8 weeks.
- Targeted intraocular delivery (anterior chamber, vitreous, sub-Tenon) for ophthalmic PK/PD modeling.
- Bile duct cannulation with selective occlusion and retrograde infusion for enterohepatic recirculation studies.
- Lymphatic fluid collection via heparin-microinfusion-enabled catheters to suppress coagulation and maintain patency.
- Intraduodenal or gastric delivery of pH-sensitive compounds, prodrugs, or microbiome modulators.
- Cisterna magna or lumbar intrathecal access for CNS-targeted delivery and CSF pharmacokinetics.
- Perioperative and post-procedural nutritional support via gastrostomy or jejunostomy feeding catheters.
FAQ
What sterilization method is recommended for these catheters?
All catheters are supplied gamma-sterilized and individually packaged in ISO 11607-compliant barrier pouches. Terminal sterilization is not recommended post-manufacture due to material degradation risks.
Can I request custom inner diameters or radiopaque markers?
Yes—custom ID/OD ratios, radiopaque stripe placement (e.g., barium sulfate–loaded segments), and laser-marked scale indicators are available under OEM agreement.
Are these catheters compatible with ethanol-based flush solutions?
Polyurethane variants tolerate ≤70% ethanol; silicone catheters are compatible with 100% ethanol. PE-lined lumens provide additional solvent resistance for DMSO or propylene glycol formulations.
Do you provide surgical insertion kits or training documentation?
Comprehensive SOPs—including species-specific implantation guides, suture techniques, and post-operative care protocols—are provided with every order. Custom surgical kit assembly (e.g., guidewires, dilators, trocars) is available upon request.
How does the BAD-Cath dual-valve configuration differ from standard T-tube systems?
The BAD-Cath uses two independent septum membranes—one proximal and one distal to the T-junction—enabling simultaneous bile diversion and targeted duodenal irrigation without cross-contamination or pressure-induced reflux.