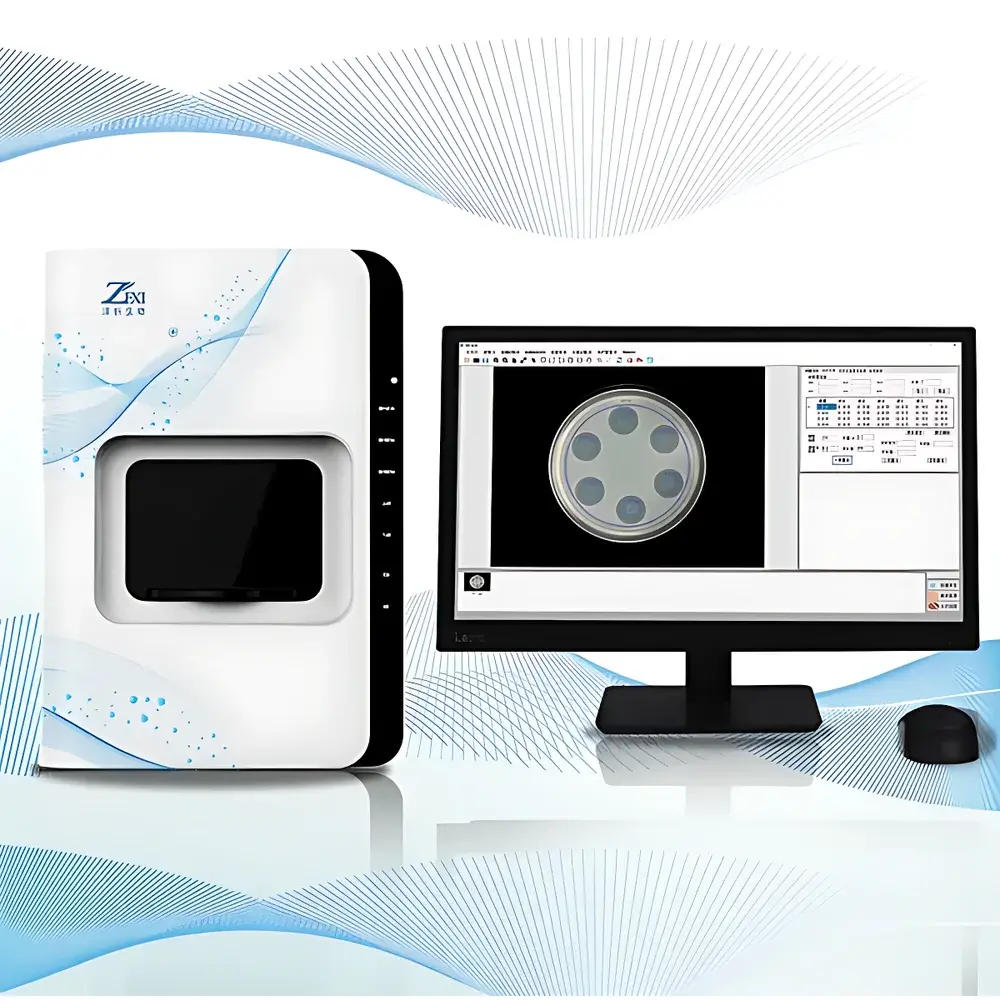
Zstream DTS2 Automatic Zone of Inhibition Measurement and Potency Analysis System
| Brand | Zstream |
|---|---|
| Origin | Zhejiang, China |
| Manufacturer Type | Direct Manufacturer |
| Model | DTS2 |
| Imaging Sensor | 16 MP Color CMOS Camera with 5 MP High-Resolution 8 mm Lens |
| Illumination | Triple-LED Visible Light (Adjustable Brightness & On/Off Control for Top/Bottom Sources), Auto-Color-Temperature Calibration, Integrated 254 nm UV Sterilization Lamp |
| Measurement Accuracy | Repeatability ≤0.001 mm (Relative Error ≤0.01%), Uniformity ≤0.03%, Inter-unit Variation ≤0.1%, Potency Reproducibility ≤0.2% (Relative Error) |
| Software Compliance | 21 CFR Part 11–Compliant Audit Trail, Multi-User Role-Based Access, Immutable Raw Data Storage, PDF Report Generation |
| Configuration | DTS2 Instrument Host, Zstream ZOI & Potency Analysis Software, Commercial Desktop PC |
Overview
The Zstream DTS2 Automatic Zone of Inhibition Measurement and Potency Analysis System is a purpose-built, regulatory-grade instrument engineered for quantitative microbiological assay workflows in pharmaceutical quality control, antibiotic development, and clinical microbiology laboratories. It implements high-resolution digital imaging coupled with algorithm-driven edge detection and geometric modeling to determine the diameter of inhibition zones formed on agar diffusion plates—enabling precise, traceable, and auditable antibiotic potency quantification per pharmacopoeial standards. The system operates on the principle of standardized agar diffusion assays (e.g., cylinder-plate and disc-diffusion methods), where microbial growth inhibition is visualized as clear circular halos around antibiotic-loaded discs or cylinders. By digitizing these zones with calibrated optics and applying validated image segmentation algorithms—including automatic contour detection, arc-based circle reconstruction, and manual three-point circle fitting—the DTS2 delivers metrologically sound measurements that serve as primary inputs for statistical potency estimation.
Key Features
- Optically isolated dark chamber with triple-LED visible illumination (top/bottom selectable, brightness-tunable via capacitive touch interface) and auto-adjusted color temperature approximating natural daylight (5500 K ±200 K), minimizing chromatic distortion during zone delineation.
- Integrated 254 nm UV-C lamp for routine sterilization of the imaging chamber between runs, reducing cross-contamination risk without requiring chemical disinfectants.
- 16-megapixel color CMOS sensor paired with a fixed-focus 5-megapixel 8 mm lens optimized for flat-field uniformity across standard Petri dish dimensions (90 mm and 150 mm), ensuring sub-pixel spatial fidelity and consistent MTF performance.
- Three-tier measurement logic: fully automated edge detection for crisp contours; arc-based circular interpolation for partially obscured zones; and manual three-point circle definition for low-contrast or irregular halos—each mode preserves full audit trail metadata.
- Pharmacopoeia-aligned potency calculation engine supporting USP <71> one-dose assay, and Chinese Pharmacopoeia (ChP 2010/2015) two-dose and three-dose parallel-line assay models with built-in statistical validation (slope, linearity, validity range).
- Hardware-level calibration traceability: includes on-board uniformity self-test, repeatability verification protocol, and inter-unit consistency benchmarking against NIST-traceable reference standards.
Sample Compatibility & Compliance
The DTS2 accommodates standard Mueller-Hinton, nutrient, or custom-selective agar plates (90 mm and 150 mm diameters), including multi-disc arrays and cylinder-plate configurations. It supports both Gram-positive (e.g., Staphylococcus aureus ATCC 25923) and Gram-negative (e.g., Escherichia coli ATCC 25922) test strains per ISO 20776-1 and CLSI M02-A12 guidelines. All measurement outputs comply with GLP and GMP documentation requirements. The system meets 21 CFR Part 11 criteria for electronic records and signatures through role-based user authentication, immutable raw image/data archiving, time-stamped audit logs capturing operator ID, action type, timestamp, and parameter changes—and secure export of final reports in tamper-evident PDF/A format.
Software & Data Management
Zstream ZOI & Potency Analysis Software is a Windows-based, FDA-auditable application featuring a modular architecture. It enforces strict separation between raw acquisition data (stored in encrypted SQLite databases with SHA-256 hashing), processed results, and report generation layers. Each user account maintains isolated workspace permissions, configurable by administrator-defined roles (e.g., analyst, reviewer, approver). All manual interventions—including edge correction, annotation insertion, and calibration offset adjustment—are logged with contextual metadata. Batch processing supports up to 96 plates per session, with automated outlier flagging based on Cochran’s C-test and parallel-line assay validity thresholds. Data exports include CSV (for LIMS integration), XML (for ELN ingestion), and PDF reports containing plate images, measured zone diameters, calculated potencies, confidence intervals, and assay validity statements.
Applications
- Antibiotic potency release testing in QC labs per ChP, USP, EP, and JP monographs.
- Stability-indicating assay development for solid- and liquid-dose antibiotics.
- Comparative efficacy screening of novel antimicrobial compounds during preclinical R&D.
- Microbial susceptibility profiling in hospital microbiology departments under CLSI M100 guidance.
- Validation and requalification of agar diffusion methods per ICH Q2(R2) analytical procedure requirements.
- Training and proficiency testing for microbiology personnel using standardized digital measurement protocols.
FAQ
Does the DTS2 support ASTM E2698 or ISO 20776-1-compliant workflows?
Yes—the system’s measurement uncertainty budget, calibration traceability, and reporting structure align with ISO/IEC 17025 requirements for microbiological testing laboratories, and its assay logic implements core statistical models referenced in both standards.
Can raw images and measurement data be exported to a Laboratory Information Management System (LIMS)?
Yes—via configurable CSV and XML templates with field-mapped identifiers (sample ID, plate ID, strain, antibiotic, dilution, zone diameter, potency, operator, timestamp). API integration is available upon request.
Is the UV sterilization function validated for bioburden reduction?
The 254 nm UV lamp delivers ≥10 mJ/cm² irradiance at chamber center over 5 minutes, validated to achieve ≥3-log10 reduction of Bacillus atrophaeus spores per ISO 15714, suitable for routine decontamination between batches.
How does the system handle overlapping or fused inhibition zones?
The software provides semi-automated separation tools: users can define boundary constraints manually, and the algorithm applies watershed segmentation with morphological filtering to resolve confluent zones while preserving original pixel integrity for audit purposes.
What validation documentation is supplied with the instrument?
Each unit ships with Factory Acceptance Test (FAT) report, IQ/OQ protocol templates, 21 CFR Part 11 compliance statement, and a traceable calibration certificate issued against NIST SRM 2034 (dimensional standard).