- All
- Favorite
- Popular
- Most rated
| Brand | Anvajo |
|---|---|
| Origin | Germany |
| Model | Fluidlab R-300 |
| Detection Time | ≤20 s (cell counting) |
| Cell Concentration Range | 1×10⁴–2×10⁷ cells/mL |
| Sample Volume | <20 µL |
| Sample Throughput | 3–80 µL |
| Wavelength Range | 375–700 nm |
| Weight | 240 g |
| Sterilization Method | Alcohol-wipe compatible surface |
| Brand | Abeyance |
|---|---|
| Origin | USA |
| Model | A220 |
| Capacity | Up to 220 × 2 mL vials (standard configuration) |
| Cooling Mode | Vapor-phase cryogenic storage (−150 °C to −190 °C operating range) |
| Interface | 7-inch capacitive touchscreen with Wi-Fi & Ethernet connectivity |
| Alarm System | SMS/email alerts, cloud-based monitoring, audit-trail logging |
| Compliance | Designed for GLP/GMP-aligned biobanking workflows |
| compatible with ISO 20387 | 2018 biobank accreditation requirements |
| Power | 100–240 V AC, 50/60 Hz, <15 W standby consumption |
| Dimensions (W×D×H) | 650 × 650 × 1,120 mm |
| Net Weight | 82 kg |
| Brand | Alexanderwerk |
|---|---|
| Origin | Germany |
| Model | BT-120 |
| Throughput | Up to 8 kg/h (lactose reference) |
| Construction | Fully stainless steel (AISI 316L), FDA-compliant non-metallic components |
| Drive System | Servo-controlled hydraulic roller pressure |
| Control | PLC-based HMI with recipe management |
| Design | cGMP-compliant, modular, tabletop, mobile base option |
| Roller Configuration | Cantilevered roll design with interchangeable surface finishes (smooth, grooved, patterned, square-profile) |
| Sealing | Circumferential roller seals + isolation-seal technology |
| Compliance | EU GMP Annex 15, ISO 9001, FDA 21 CFR Part 11–ready audit trail (optional), ICH Q5A/Q5B relevant for biologics processing |
| Brand | Abeyance |
|---|---|
| Origin | USA |
| Model | A1000 |
| Capacity | Up to 1,000 L (liquid nitrogen hold time ≥ 120 days) |
| Operating Temperature Range | −190 °C to −150 °C (vapor-phase zone) |
| Construction | Dual-walled stainless steel with high-vacuum insulation |
| Control System | Microprocessor-based touchscreen interface with Wi-Fi/Ethernet connectivity |
| Compliance | Designed per ISO 13485 principles for medical device storage environments |
| Alarm Options | SMS, email, and cloud-based notifications via secure TLS-encrypted API |
| Brand | Abeyance |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Product Origin | Imported |
| Model | A440 |
| Price Range | USD 35,000 – 42,000 |
| Brand | Abeyance |
|---|---|
| Origin | USA |
| Model | A700 |
| Capacity | ≥700 L (vapor-phase storage volume) |
| Static Evaporation Rate | ≤0.8 L/day (typical, at 25°C ambient) |
| Temperature Uniformity | ±5°C across storage chamber (–196°C to –150°C operational range) |
| Max. Sample Capacity | Up to 40,000 cryovials (2 mL) with standard 10×10 racks |
| Vacuum Integrity | <1×10⁻⁴ Pa after initial pump-down, maintained for ≥15 years per ISO 23756:2021 qualification |
| Compliance | Meets ASTM F2697-22 (Standard Specification for Vapor Phase Liquid Nitrogen Freezers), FDA 21 CFR Part 11–ready software architecture |
| Brand | 3P Innovation (UK) |
|---|---|
| Origin | United Kingdom |
| Manufacturer Type | Authorized Distributor |
| Product Category | Imported Instrument |
| Model | Fill2Weight |
| Application Segment | Biopharmaceuticals |
| Compliance | FDA cGMP, EU GMP Annex 1, NMPA Sterile Manufacturing Guidelines |
| Material Construction | 316L Stainless Steel + Food-Grade Coating |
| Cleaning | CIP/SIP-Compatible |
| Particle Containment | <0.1 µg/m³ (OEL ≤1 µg/m³ compatible) |
| Dose Uniformity (CV) | ≤2.5% |
| Powder Size Range | 1–100 µm |
| Throughput (Commercial Mode) | >2000 units/min |
| Cleanroom Compatibility | ISO Class 5 (Grade A/B environments) |
| Brand | HOF |
|---|---|
| Origin | Germany |
| Model | FT4-38 |
| Temperature Range | −73 °C to +40 °C |
| Temperature Accuracy | ±1 °C |
| Cooling Rate | −1 °C/min |
| Heating Rate | +1 °C/min |
| Batch Capacity | up to 10 × 20 L single-use bags |
| Compliance | FDA 21 CFR Part 11 (audit trail, role-based access control), GMP-ready architecture |
| Software | Integrated PC with validated GUI, PDF report generation |
| Brand | STREX |
|---|---|
| Origin | Japan |
| Model | SP-5000 |
| Pressure Range | 120–300 mmHg |
| Gas Requirement | 5% CO₂ + 95% Air mixture |
| Operating Modes | Continuous Pressurization / Cyclic Pressurization-Decompression |
| Compliance | Designed for ISO 13485-aligned lab environments and GLP-compliant cell culture workflows |
| Brand | BioLife Solutions (formerly Custom Biogenic Systems) |
|---|---|
| Origin | USA |
| Model | 2101 Series |
| Voltage/Frequency | 230 V, 50/60 Hz |
| Dimensions (D×H×W) | 648 mm × 743 mm × 484 mm |
| Temperature Uniformity | ±0.1 °C |
| Temperature Accuracy | ±0.1 °C |
| Control Range | +50 °C to −180 °C |
| Cooling Rate | 0.01–99 °C/min |
| Heating Rate | up to 20 °C/min |
| Chamber Volume | 28 L |
| Software | Series 2101 Control Software (Windows OS) |
| Brand | Esco |
|---|---|
| Origin | Taiwan |
| Manufacturer Type | Authorized Distributor |
| Product Origin | China (Manufactured in Taiwan) |
| Model | CelCradle |
| Temperature Range | 22–40 °C |
| Working Volume Range | 5 L to 500 L |
| Number of Bioreactor Vessels | 1–4 |
| Material | PET (Polyethylene Terephthalate) |
| Sterilization Method | Gamma Irradiation |
| Brand | NanoEntek |
|---|---|
| Origin | South Korea |
| Model | ExTransfection |
| Type | Fully Automated |
| Compliance | CE-IVD, ISO 13485–certified manufacturing environment |
| Application Focus | Primary cells, stem cells, CRISPR RNP delivery, GMP-compatible process development |
| Brand | TAIYO NIPPON SANSO |
|---|---|
| Origin | Japan |
| Model | MR-LN-6000 |
| Capacity | 6 L (liquid nitrogen hold time ≥4 h at ≤−150 °C in 25 °C ambient) |
| Temperature Monitoring | Integrated digital temperature recorder with transparent lid display |
| Insulation | High-vacuum multilayer insulation (MLI) + adsorbent-based vapor-phase stabilization |
| Compliance | Designed for ISO 13485-aligned biologics handling environments |
| Regulatory Context | Supports GLP-compliant sample chain-of-custody documentation |
| Brand | Eureka Bio |
|---|---|
| Origin | Shenzhen, China |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Domestic (China) |
| Model | CellSep Pro |
| Price Range | USD 70,000 – 140,000 (approx.) |
| Type | Fully Automated |
| Sample Volume | 10 mL – 500 mL |
| Cell Processing Capacity | Configurable per protocol (e.g., PBMC isolation from 30–150 mL whole blood |
| Temperature Control | Ambient (no active thermal regulation) |
| Maximum Culture Volume | Customizable upon request |
| Gas Pressure Control | Not applicable |
| Brand | BioLife Solutions |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Product Category | Imported Instrument |
| Model | BioT ULT Workstation |
| Power Consumption | 1 kW |
| Dimensions (W×D×H) | 152.4 × 71.1 × 44.5 cm |
| Temperature Uniformity | ±1°C |
| Temperature Accuracy | ±0.1°C |
| Control Range | −50°C to +70°C |
| ULT Operating Range (dry ice mode) | −75°C to −50°C |
| Stabilization Time to <−50°C | <30 min |
| Hold Time at <−50°C (lid open, single dry ice charge) | >15 h |
| Effective ULT Zone Depth | 10 inches (25.4 cm) below chamber top |
| Brand | Flow Sciences (FSI) |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | FSOEB |
| Pricing | Available Upon Request |
| Brand | Sony |
|---|---|
| Origin | Japan |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | CGX10 |
| Pricing | Upon Request |
| Type | Temperature-Controlled (Low-Temperature Capable) |
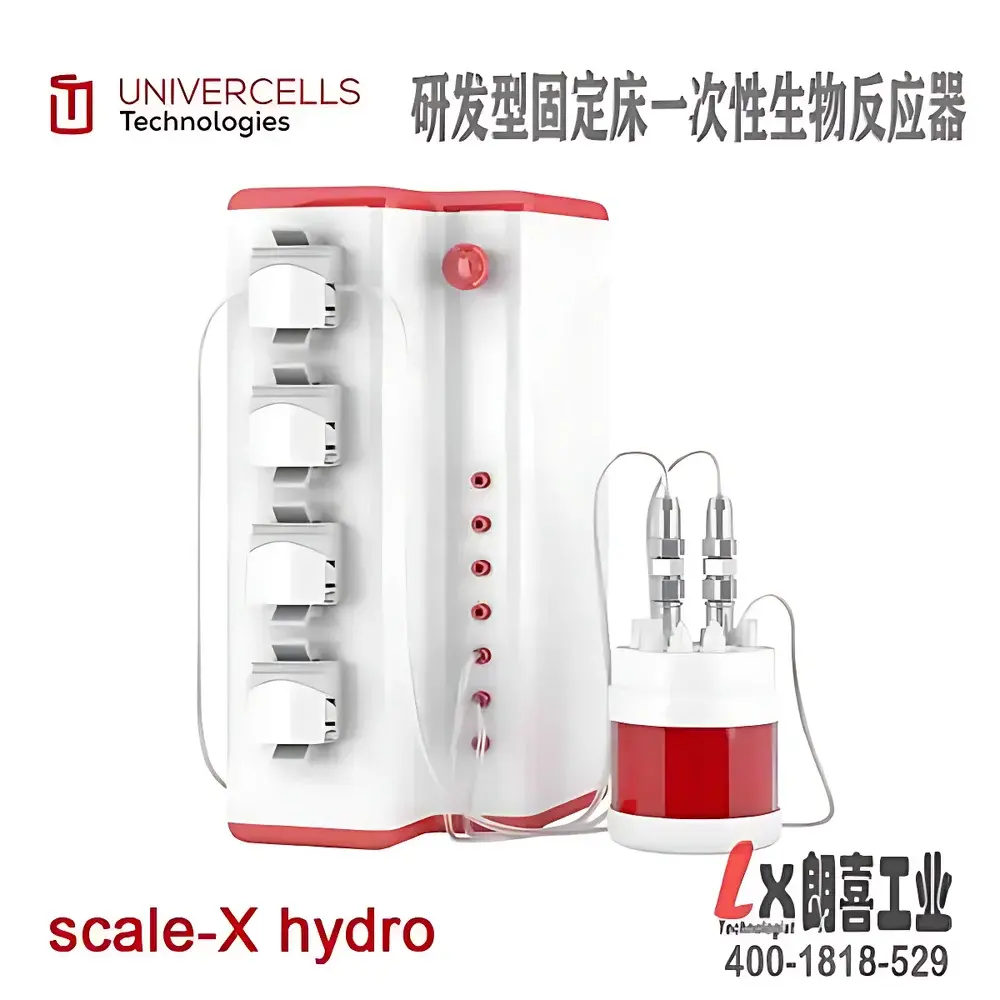
| Brand | Univercells Technologies (Belgium) |
|---|---|
| Origin | Belgium |
| Model | Scale-X™ hydro |
| Growth Surface Area | 2.4 m² |
| Configuration | Single-layer fixed-bed module |
| Construction Materials | Structured spiral-wound PET mesh and medical-grade silicone tubing |
| Sterilization | Pre-sterilized single-use disposable cartridge |
| Temperature Control | Ambient to 37 °C (external incubator required) |
| Application Scope | Adherent and suspension cell culture for R&D-scale vaccine and viral vector process development |
| Compliance | Designed for GLP-compliant process characterization |
Show next