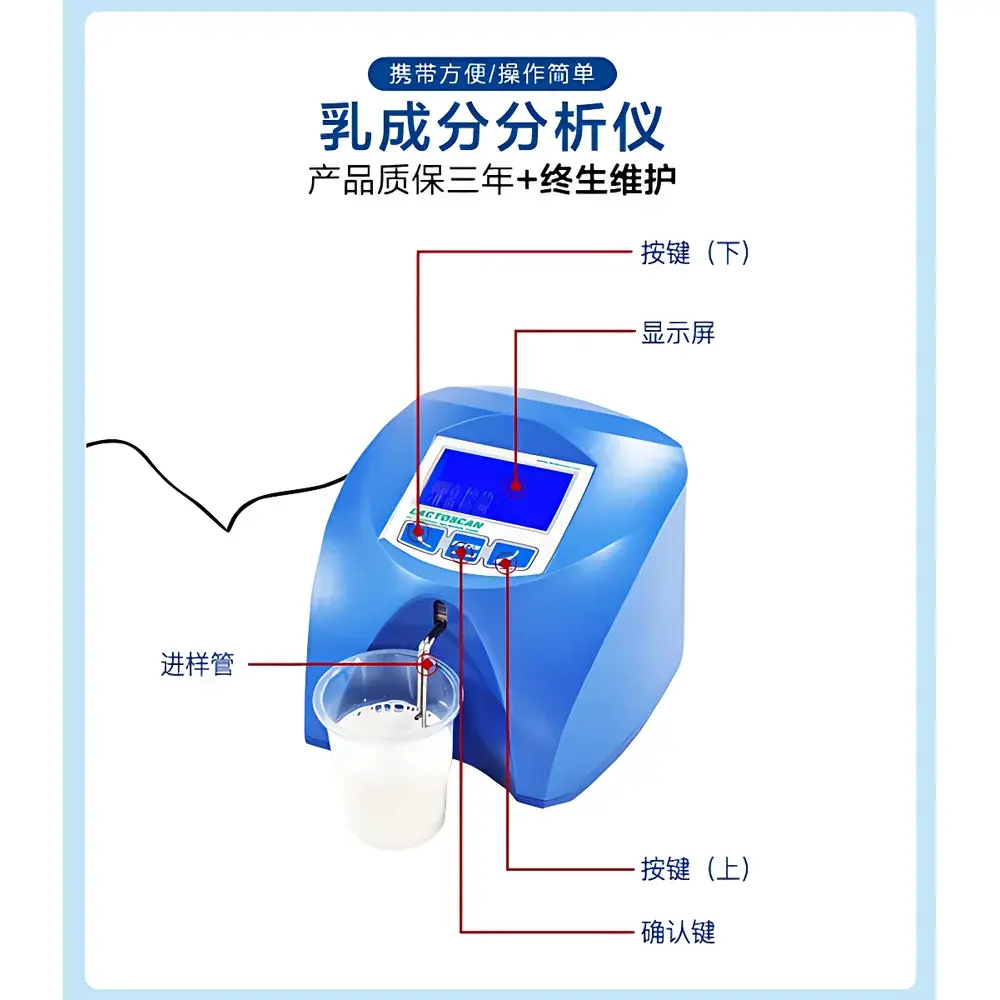
Neo LACTOSCAN MILKYWAY-S Dairy Analyzer
| Brand | Neo |
|---|---|
| Origin | Zhejiang, China |
| Manufacturer Type | Authorized Distributor |
| Country of Origin | China |
| Model | MILKYWAY-S |
| Detection Parameters | Fat (0.5–12.00%), Non-Fat Solids (6.00–12.00%), Density (1.0260–1.0330 g/cm³), Protein (2.00–6.00%), Freezing Point (0.000–1.000 °C), Water Adulteration (0–60.00%), Lactose (1.5–7.0%), Ash (0–30%), Temperature (1–40 °C) |
| Analysis Time | 60–90 seconds |
| Repeatability | < 0.5% CV |
| Accuracy | Fat ±0.1%, Protein ±0.2%, Lactose ±0.15% |
| Automation Level | Semi-Automatic |
| Application Category | Food & Dairy Quality Control Instruments |
Overview
The Neo LACTOSCAN MILKYWAY-S Dairy Analyzer is a semi-automated, benchtop instrument engineered for rapid, reliable quantification of key compositional parameters in raw and processed milk. It employs near-infrared (NIR) spectroscopy combined with chemometric calibration models validated against reference methods (e.g., Gerber, Kjeldahl, cryoscopic freezing point depression) to deliver real-time, multi-parameter analysis without reagents or sample preparation. Designed for routine quality assurance in dairy collection centers, processing plants, and regulatory laboratories, the MILKYWAY-S supports compliance-driven workflows by delivering traceable, reproducible results aligned with ISO 9622:2013 (Milk — Determination of fat, protein, lactose, total solids and urea — Guidance on instrumental methods) and national dairy standards (e.g., GB 5413.1–2010 series). Its compact footprint, low maintenance architecture, and intuitive interface enable seamless integration into high-throughput QC environments where speed, robustness, and method equivalence are critical.
Key Features
- NIR-based multi-component analysis with factory-installed, pre-validated calibrations for fat, protein, lactose, non-fat solids, density, freezing point, ash, and water adulteration
- Analysis cycle completed in 60–90 seconds per sample—optimized for batch testing of incoming raw milk lots
- High repeatability (< 0.5% coefficient of variation) and documented accuracy: ±0.1% for fat, ±0.2% for protein, ±0.15% for lactose relative to reference methods
- Temperature-controlled sample chamber (1–40 °C operating range) ensuring measurement stability across seasonal ambient variations
- Semi-automatic operation with manual pipetting and automated optical path alignment—minimizing operator dependency while maintaining procedural control
- Robust mechanical design with stainless-steel fluidic pathways and sealed optical module for long-term reliability in industrial settings
Sample Compatibility & Compliance
The MILKYWAY-S accepts homogenized, unfiltered raw cow’s milk, buffalo milk, and standardized pasteurized samples (1–5 mL volume). It accommodates minor particulate presence but requires removal of gross sediment or cream separation prior to analysis. The system meets functional requirements for ISO/IEC 17025-compliant laboratories when operated under documented SOPs. Calibration verification is supported via certified reference materials (CRM) traceable to NIST SRM 1846 (Whole Milk Powder) and internal validation protocols. While not FDA 21 CFR Part 11–compliant out-of-the-box, audit trails and user access logs can be implemented via external LIMS integration for GLP/GMP-aligned environments.
Software & Data Management
Instrument control and data acquisition are managed through Neo’s proprietary Windows-based software (v3.2+), supporting CSV and Excel export, customizable report templates (PDF/Excel), and batch summary statistics (mean, SD, CV, pass/fail flags). Raw spectral data and calibration metadata are stored with timestamped audit trails. Software allows user-defined acceptance criteria per parameter, automatic outlier detection, and trend charting over time. Data security follows ISO 27001 principles; local database encryption and role-based user permissions (admin/operator/viewer) ensure integrity during multi-shift operations. Integration with enterprise systems (e.g., SAP QM, LabWare LIMS) is available via ODBC and HL7-compatible APIs.
Applications
- Raw milk reception testing at dairy cooperatives and collection hubs
- In-process monitoring of standardization, homogenization, and thermal treatment efficacy
- Verification of compositional claims on finished products (UHT, skim, fortified milks)
- Adulteration screening—including water dilution and reconstitution ratio estimation
- Freezing point depression analysis for detecting added water per Codex Alimentarius Standard 206–1999
- Supporting HACCP plan verification and daily equipment performance checks
FAQ
What reference standards are used to validate the MILKYWAY-S calibrations?
Calibrations are developed and verified using AOAC Official Methods and ISO 9622–aligned protocols, with primary validation against Gerber fat, Kjeldahl protein, and cryoscopic freezing point reference measurements.
Is the instrument suitable for goat or sheep milk analysis?
Yes—though default calibrations are optimized for bovine milk, species-specific models can be developed upon request using customer-provided representative samples and reference lab data.
Does the system require daily recalibration?
No—factory calibrations are stable for ≥6 months under normal use; quarterly verification with CRM or reference samples is recommended per ISO/IEC 17025 clause 7.7.
Can results be exported to an ERP or LIMS platform?
Yes—via configurable file export (CSV, XML) or direct database connection using standard ODBC drivers and secure API endpoints.
What maintenance is required beyond routine cleaning?
Annual optical alignment verification and NIR source intensity check are advised; no consumables or lamp replacements are needed within the first 5 years of operation.