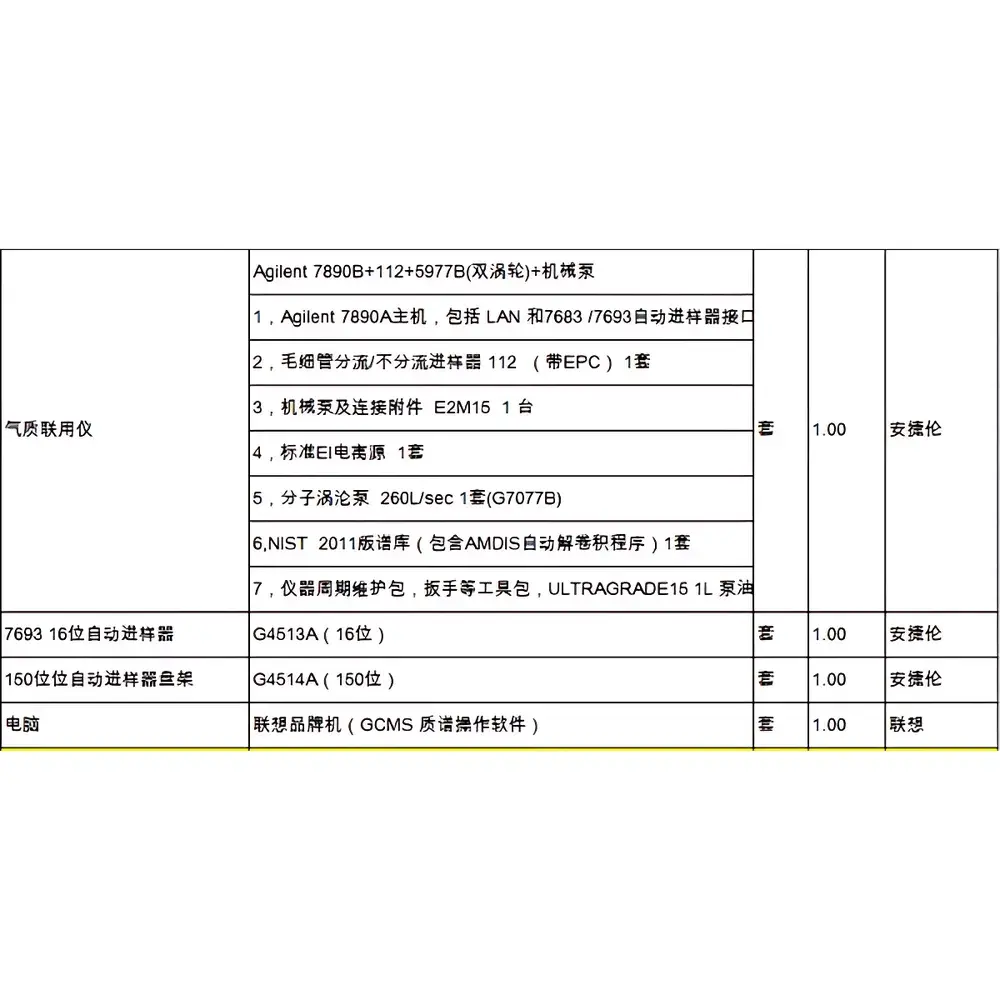
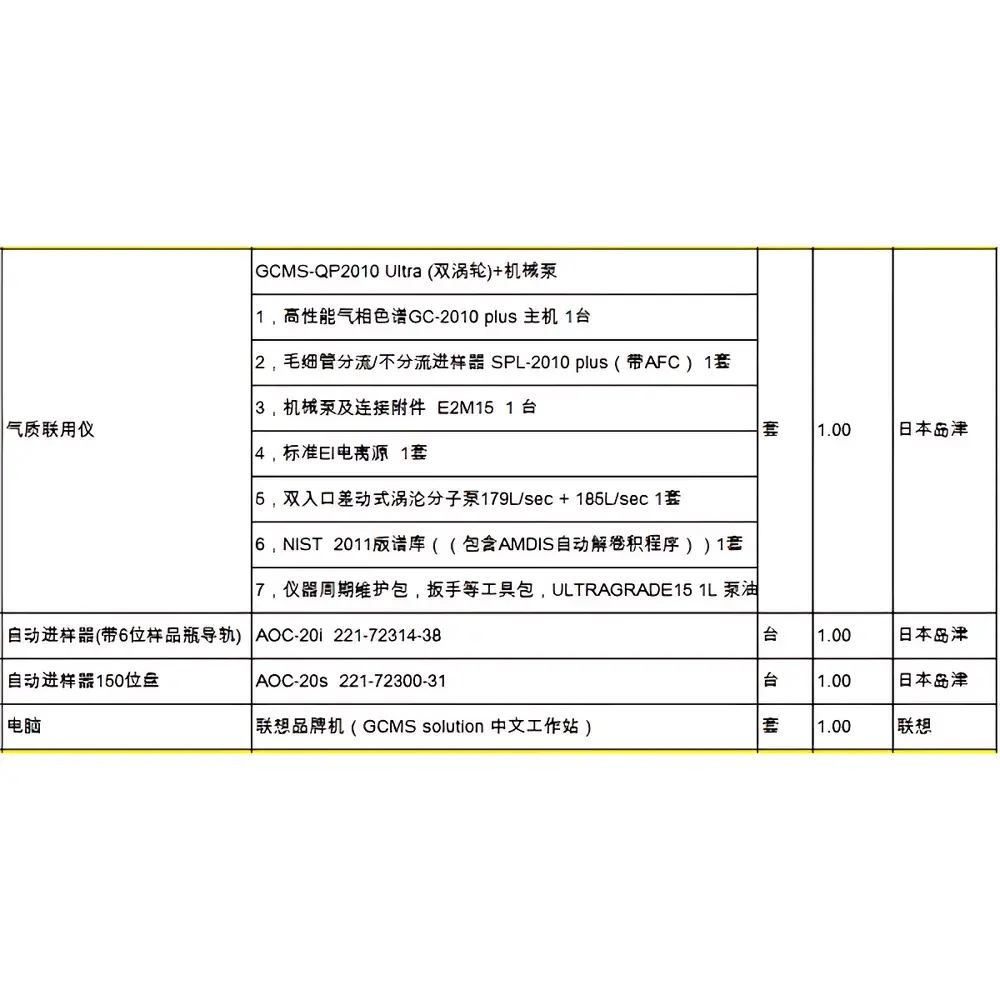
GE AKTA Explorer 100 Biological Purification System
| Brand | Baker Hughes (formerly GE Healthcare) |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Import Status | Imported |
| Model | GE AKTA Explorer 100 Biological Purification System |
| Operational Age | >10 years |
| Dimensions (L×W×H) | 860 × 710 × 660 mm |
| Weight | 116 kg |
| Operating Temperature | 4–35 °C |
| Relative Humidity | 20–95% (non-condensing) |
| Power Consumption | 1100 W (instrument: 800 W, PC: 300 W) |
| UV Detection Range | –6 to +6 AU |
| Conductivity Accuracy | ±0.01 mS/cm or ±2% |
| Temperature Sensor Range | 0–99 °C (±1.5 °C accuracy, 4–45 °C) |
| pH Sensor Range | 0–14 pH (±0.1 pH accuracy, pH 2–12) |
| Fraction Collector Volume Range | 0.1–50 mL |
| Integrated Pressure Sensors | System pump, sample pump, column inlet, column outlet |
| Integrated Air Bubble Detectors | Sample inlet, Pump A, Pump B |
Overview
The GE AKTA Explorer 100 Biological Purification System is a benchtop preparative liquid chromatography platform engineered for robust, reproducible, and scalable biomolecule purification in research, process development, and early-stage GMP environments. Designed and originally manufactured by GE Healthcare (now part of Cytiva, with legacy systems supported under Baker Hughes’ instrumentation portfolio), the AKTA Explorer 100 implements high-precision peristaltic pumping, real-time multi-parameter monitoring (UV, conductivity, pH, pressure differential), and intelligent fraction collection to support ion exchange, size exclusion, affinity, and hydrophobic interaction chromatography. Its architecture follows the core principles of controlled flow dynamics, column protection via dual pressure sensing, and integrated system-level diagnostics — all coordinated through the UNICORN 6 software platform. The system is purpose-built for method development workflows requiring statistical rigor, regulatory traceability, and seamless scale-up to production-grade AKTA systems such as the AKTA avant or AKTA Pure.
Key Features
- BufferPro Automated Buffer Preparation: Integrated four-channel valve-based online buffer blending enables real-time formulation of up to 26 predefined buffer recipes. Acid/base titration is performed dynamically to achieve target pH and ionic strength, with automatic temperature and conductivity compensation — eliminating manual preparation, reducing variability, and accelerating screening cycles by ~30 minutes per condition.
- Design of Experiments (DoE) Integration: UNICORN 6 embeds statistically rigorous DoE tools compliant with ICH Q5 and Q8 guidelines. Users define factor ranges (e.g., gradient slope, flow rate, load density, pH) and execute multivariate experiments in minimal runs, enabling efficient parameter screening, optimization, and robustness assessment without sequential one-factor-at-a-time (OFAT) limitations.
- Method Editor & Visual Workflow Design: Drag-and-drop stage-based method construction allows rapid protocol assembly using preconfigured modules (equilibration, load, wash, elution, regeneration). Advanced users retain full scripting access via UNICORN’s Method Language (UML), supporting conditional logic, loops, and external device integration.
- Column Protection Architecture: Dual pressure transducers (inlet and outlet) continuously calculate ΔP across the column bed. Real-time differential pressure monitoring safeguards resin integrity during packing and operation; automated flow adjustment prevents over-pressurization while maintaining optimal linear velocity.
- UNICORN 6 Regulatory Compliance: Fully compliant with FDA 21 CFR Part 11, featuring electronic signatures, role-based user permissions, immutable audit trails, and secure database logging. All method versions, run logs, instrument events, and electronic records are timestamped and cryptographically sealed within a single relational database.
- UniTag & Column Logbook: QR-code-based column tracking enables full lifecycle management — including total runs, cumulative pressure exposure, cleaning-in-place (CIP) history, and maintenance alerts. Prepacked columns (e.g., HiScreen, Capto, MabSelect) are auto-recognized; custom-packed columns can be registered manually or via UniTag labels.
Sample Compatibility & Compliance
The AKTA Explorer 100 supports purification of monoclonal antibodies, recombinant proteins, plasmid DNA, viral vectors, and other therapeutic biomolecules across molecular weights from 5 kDa to >1 MDa. It accommodates standard analytical and semi-preparative columns (1–26 mm ID) and HiScreen prepacked screening columns (10 mm × 20 mm, 10 mm × 50 mm). All fluidic pathways are constructed from biocompatible, low-binding materials (PEEK, stainless steel, PTFE) validated per USP and ISO 10993-5. System validation documentation, including IQ/OQ protocols and 21 CFR Part 11 readiness reports, is available through authorized service partners. The platform meets essential requirements for GLP-compliant process characterization studies and supports alignment with ICH Q5A(R2), Q5B, and Q5D.
Software & Data Management
UNICORN 6 serves as the central control, analysis, and data governance layer. It provides synchronized acquisition of UV280/254 nm, conductivity, pH, temperature, and pressure signals at ≥1 Hz sampling resolution. All raw and processed data are stored in an encrypted, tamper-evident SQLite database with automatic daily backups. Reporting modules generate PDF/Excel-compliant summary reports with embedded chromatograms, peak tables, and metadata (user, timestamp, method version, instrument configuration). Audit trail entries record every action — from method modification to fraction collection trigger — with operator ID and justification fields where required. Remote access is supported via secure RDP or VNC, subject to network policy controls.
Applications
- Early-stage purification method development for mAbs and bispecifics using DoE-driven gradient and pH scouting
- HiScreen-based rapid screening of resin selectivity, binding capacity, and elution stringency
- Process characterization studies under defined DoE matrices for regulatory filing (e.g., BLA submissions)
- Small-scale polishing step optimization prior to transfer to AKTA avant or AKTA Pure platforms
- Training and competency assessment in academic and contract development laboratories
- Stability-indicating assays for forced degradation studies (oxidation, deamidation, aggregation)
FAQ
Is the AKTA Explorer 100 compatible with modern BioProcess resins such as Capto Core 700 or MabSelect PrismA?
Yes. The system’s maximum flow rate (10 mL/min), pressure limit (20 MPa), and low-dead-volume fluidics are fully aligned with the operational specifications of Cytiva’s BioProcess resins. Method parameters established on Explorer 100 demonstrate high predictability during scale-up.
Can UNICORN 6 export data to third-party analysis tools like Empower or Chromeleon?
Raw chromatographic data (UV, conductivity, etc.) can be exported in ASCII (.txt) or CSV format with full time-stamped metadata. Binary .chm files require UNICORN-native viewers but are convertible via UNICORN’s built-in report engine.
What validation support is provided for second-hand units?
Pre-delivery performance qualification (PQ) is conducted per ASTM E2500-13. Full documentation packages — including calibration certificates, PQ reports, and 21 CFR Part 11 configuration summaries — are delivered with each unit. Optional IQ/OQ services are available through certified field engineers.
Does the system include a PC, and is it upgradeable to newer UNICORN versions?
The system ships with a dedicated industrial PC preloaded with UNICORN 6.4. Upgrades to UNICORN 7.x are supported subject to hardware compatibility and license entitlement — verified during pre-installation assessment.
How is column lifetime tracked, and can alerts be configured for maintenance?
Column Logbook automatically records run count, cumulative ΔP, and CIP cycles. Threshold-based notifications (e.g., “Alert after 50 runs” or “Warn at 80% of max rated ΔP”) are configurable in UNICORN’s Column Manager module.