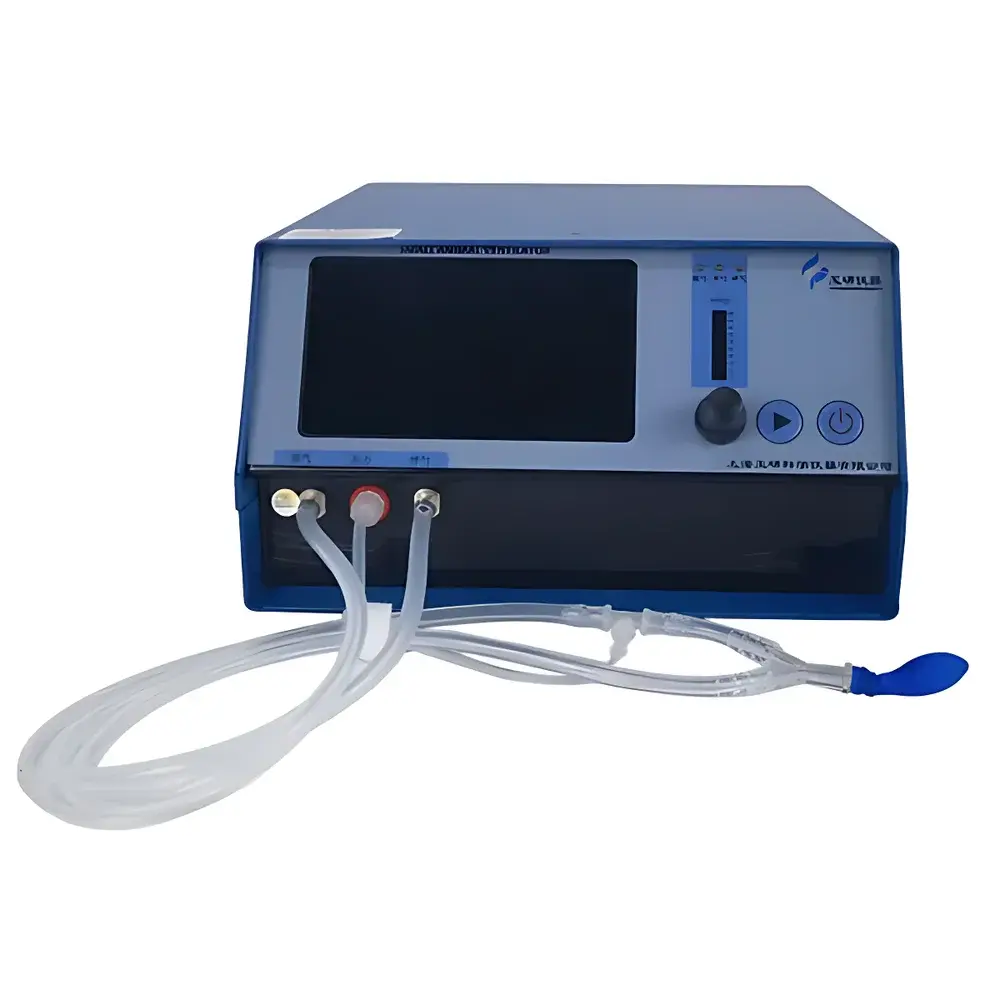
YUYANBIO V-100 Small Animal Ventilator
| Brand | YUYANBIO |
|---|---|
| Origin | USA |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Category | Imported Instrument |
| Model | V-100 |
| Price Range | USD 1,400 – 7,000 |
| Power Supply | 220 V AC |
| Dimensions | 24 × 16 × 10 cm |
| Weight | 1.5 kg |
| Operating Temperature | −10 °C to 60 °C |
| Relative Humidity | 20–80 % RH |
| Respiratory Rate | 1–300 breaths/min |
| Tidal Volume | 0.1–10 mL (0.1 mL step resolution) |
| I | E Ratio: 1:5 to 5:1 (0.1 step) |
| Peak Inspiratory Pressure Limit | 6 kPa |
| Airway Pressure Relief | Automatic overpressure release with user-set threshold |
| Gas Source | Ambient air or external gas supply (no high-pressure gas required) |
| Drive Mechanism | Electrically driven, electronically controlled |
| Interface | Touch-sensitive digital control panel with LED indicators (inspiration/expiration/maintained inspiration) |
| Safety Features | Real-time pressure monitoring, audible/visual alarms, fail-safe pressure limiting |
Overview
The YUYANBIO V-100 Small Animal Ventilator is a precision-engineered, electrically driven, electronically controlled ventilatory support system designed specifically for acute in vivo rodent and small mammal research. It operates on the principle of pressure-limited, volume-targeted intermittent positive pressure ventilation (IPPV), delivering reproducible tidal volumes while maintaining strict control over inspiratory pressure, respiratory rate, and inspiratory-to-expiratory (I:E) ratio. Unlike pneumatic-driven systems requiring compressed gas infrastructure, the V-100 integrates an internal motorized blower and closed-loop flow sensor architecture—enabling stable, quiet, and portable operation in standard laboratory environments. Its design adheres to fundamental physiological constraints observed in murine and lagomorph models: tidal volumes scaled to body weight (e.g., 8–12 mL/kg for mice, 6–10 mL/kg for rats), respiratory frequencies ranging from 40–200 breaths/min, and peak airway pressures constrained to ≤6 kPa to prevent barotrauma. The device is intended for use during terminal surgical procedures, electrophysiology, imaging (e.g., fMRI, PET, optical coherence tomography), and pharmacodynamic studies where precise, artifact-free respiratory control is essential.
Key Features
- Electrically powered, standalone operation—no external high-pressure gas supply required.
- Adjustable tidal volume range: 0.1–10 mL in 0.1 mL increments, optimized for mice (18–30 g), rats (200–500 g), guinea pigs (300–700 g), and rabbits (<2.5 kg).
- Respiratory rate programmable from 1 to 300 breaths per minute, supporting both bradypneic and tachypneic experimental paradigms.
- I:E ratio continuously adjustable from 1:5 to 5:1 (0.1-step resolution), enabling simulation of pathological breathing patterns (e.g., obstructive vs. restrictive profiles).
- Digital touch interface with real-time LED status indicators: green (inspiration), red (expiration), amber (inspiratory hold).
- Integrated overpressure relief valve with user-configurable upper pressure limit (default 6 kPa); automatic venting prevents alveolar overdistension.
- Ultra-low acoustic emission (<35 dB(A))—critical for noise-sensitive neurophysiological and behavioral assays.
- Compact footprint (24 × 16 × 10 cm) and lightweight construction (1.5 kg) facilitate integration into stereotaxic rigs, MRI bore adapters, and laminar flow hoods.
- Robust environmental tolerance: operational between −10 °C and 60 °C, 20–80% RH—suitable for shared core facilities and mobile field labs.
Sample Compatibility & Compliance
The V-100 supports intubation-based ventilation via species-specific tracheal cannulae (included: mouse and rat sizes). Non-invasive oral/nasal mask interfaces are compatible with optional accessories. Device performance has been validated across common laboratory strains including C57BL/6, BALB/c, Sprague-Dawley, and New Zealand White rabbits. While not certified to ISO 13485 or FDA 510(k) as a medical device, the V-100 conforms to general laboratory equipment safety standards (IEC 61010-1) and incorporates engineering safeguards aligned with AAALAC International recommendations for perioperative animal welfare. All pressure and flow parameters remain traceable to NIST-traceable calibration sources used during factory validation. The system meets GLP-aligned documentation requirements for protocol-driven studies—parameter settings, runtime logs, and alarm events are retained in non-volatile memory for audit readiness.
Software & Data Management
The V-100 operates autonomously via its embedded microcontroller and requires no host PC for basic function. However, it includes a USB 2.0 port (device mode) for firmware updates and future software expansion. Though native data logging is not implemented in the base V-100 model, time-stamped parameter snapshots (rate, volume, I:E, pressure limit) can be captured externally using TTL-triggered acquisition systems (e.g., National Instruments DAQ, Spike2, or MATLAB-based custom scripts). The ventilator emits synchronous TTL pulses at end-inspiration and end-expiration—enabling precise temporal alignment with electrophysiological recordings, optical imaging triggers, or gas analyzer sampling clocks. Future firmware releases may support CSV export via USB mass storage emulation, pending institutional validation protocols.
Applications
- Acute terminal surgery: craniotomy, thoracotomy, carotid ligation, spinal cord injury models.
- In vivo functional imaging: BOLD fMRI, two-photon calcium imaging, photoacoustic tomography—where respiratory motion artifacts must be minimized and synchronized.
- Pharmacokinetic/pharmacodynamic (PK/PD) studies involving IV or inhalational drug delivery under controlled ventilation.
- Respiratory challenge models: hypoxia-reoxygenation, hypercapnia, bronchoconstrictor provocation (e.g., methacholine), and neuromuscular blockade titration.
- Neuroscience applications: optogenetic stimulation during controlled apnea windows, chemogenetic interrogation of central pattern generators.
- Training platform for veterinary and biomedical graduate students in principles of mechanical ventilation physiology and instrumentation safety.
FAQ
Is the V-100 suitable for long-term survival surgeries?
No—the V-100 is engineered for acute, terminal, or short-duration (<6 hr) ventilatory support during anesthesia. It lacks humidification, CO₂ rebreathing management, and integrated capnography required for prolonged recovery ventilation.
Can the V-100 be used with nitrous oxide or volatile anesthetics?
Yes—when paired with a compatible upstream vaporizer (e.g., VIP3000 series), the V-100 delivers carrier gas containing anesthetic agents. However, the ventilator itself does not regulate anesthetic concentration; that function resides entirely in the vaporizer and flowmeter assembly.
Does the device comply with FDA or CE regulatory pathways?
The V-100 is classified as a research-use-only (RUO) life science instrument—not a human medical device—and therefore falls outside FDA 21 CFR Part 820 or EU MDR scope. It is supplied with Declaration of Conformity to IEC 61010-1 for laboratory equipment safety.
How is calibration maintained over time?
Factory calibration is performed using NIST-traceable flow and pressure standards. Users should verify tidal volume output quarterly using a calibrated dry gas meter or syringe pump; drift exceeding ±5% warrants service recalibration by authorized YUYANBIO technical support.
What maintenance is required?
Routine cleaning of external surfaces and gas pathways with 70% isopropanol; annual inspection of O-rings, valves, and motor brushes by qualified personnel. No consumables are required beyond standard tracheal cannulae and disposable filters (optional).