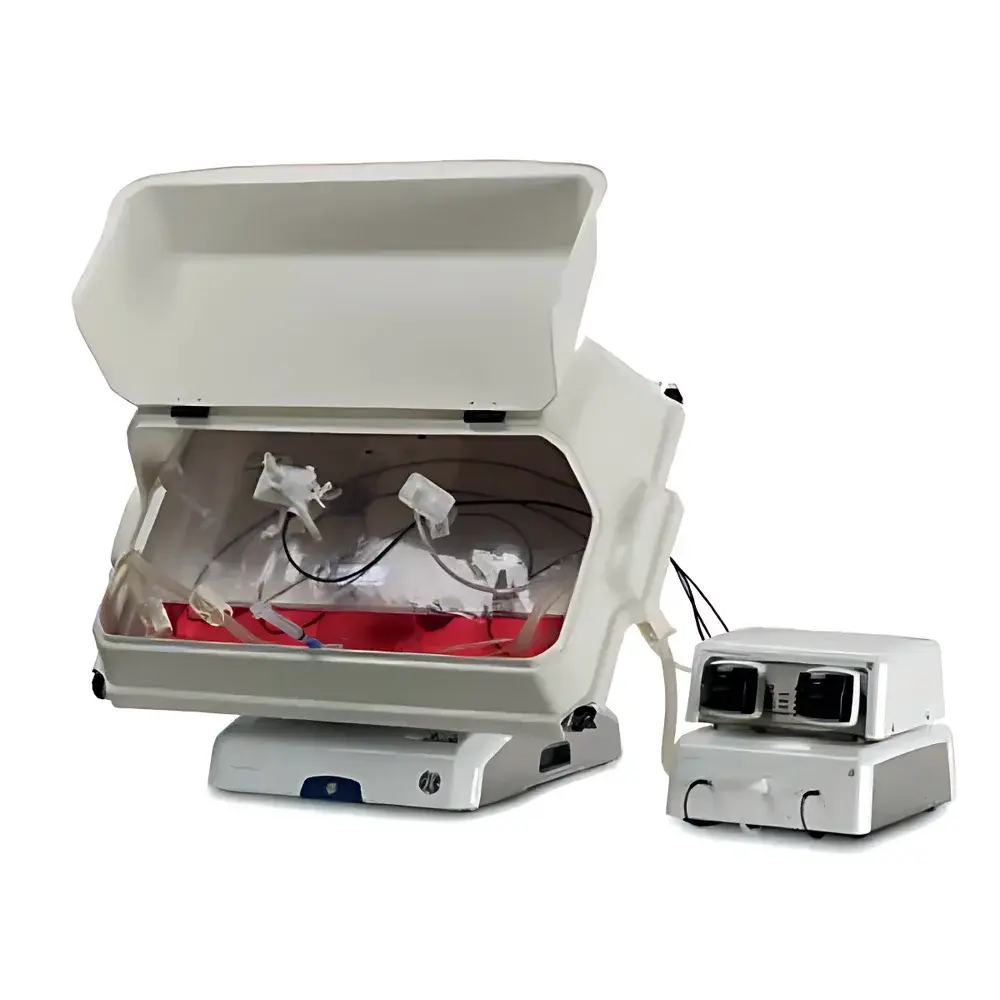
Cytiva ReadyToProcess WAVE 25 Wave Bioreactor
| Brand | Cytiva |
|---|---|
| Origin | United Kingdom |
| Manufacturer Type | Authorized Distributor |
| Product Category | Imported |
| Model | GE ReadyToProcess WAVE 25 Bioreactor |
| Temperature Range | 6–60 °C |
| Working Volume Range | 10–2000 L |
| Number of Vessels | 1 |
| Construction Material | Stainless Steel |
| Sterilization Method | Single-Use Bioreactor System |
Overview
The Cytiva ReadyToProcess WAVE 25 Wave Bioreactor is an engineered, single-use platform designed for scalable mammalian, insect, and plant cell culture under controlled physiological conditions. It operates on the principle of orbital rocking-induced wave motion—a hydrodynamically optimized mixing mechanism that generates gentle yet effective bulk fluid movement within flexible, pre-sterilized Cellbag™ bioreactor bags. This motion enables simultaneous oxygen transfer, CO2 removal, and homogenous nutrient distribution without mechanical agitation or sparging, thereby minimizing shear stress on sensitive suspension or microcarrier-adherent cells. The system integrates a stainless-steel rocking base unit, a programmable gas mixing module (CBCU), and a precision peristaltic pump—each governed by UNICORN™ software to ensure closed-loop control of temperature, pH, dissolved oxygen (DO), liquid addition, and harvest operations. Designed for compliance with current Good Manufacturing Practice (cGMP) environments, the WAVE 25 supports seamless transition from lab-scale process development (10–25 L) to pilot-scale production (up to 2000 L via scalable bag formats), maintaining consistent mass transfer coefficients (kLa) and mixing times across scales.
Key Features
- Integrated rocking platform with precision servo-controlled motion: adjustable speed (0–40 rpm), tilt angle (0–12°), and acceleration profile (15–100% waveform modulation) to tailor hydrodynamic conditions for suspension, microcarrier, or low-shear adherent cultures.
- Dual mirrored solid-state drives (SSDs) embedded in the rocker base ensure continuous, network-independent data logging with redundancy—no data loss even if one drive fails during active cultivation.
- Real-time, closed-loop control of temperature (6–60 °C), pH (via CO2/base titration), and DO (via O2/N2 blending) using calibrated optical sensors integrated into the Cellbag™ and mass flow controllers in the CBCU module.
- Ergonomic tilting function (up to 30°) enables gravity-assisted sampling and harvest—eliminating manual lifting and reducing operator intervention while preserving sterility.
- UNICORN™ software v7.0+ compliant with FDA 21 CFR Part 11 and GAMP® 5: supports electronic signatures, audit trails, role-based access control, and automated IQ/OQ documentation generation.
- Modular architecture allows up to three WAVE 25 units to be synchronized and monitored from a single UNICORN client workstation via OPC UA communication protocol.
Sample Compatibility & Compliance
The WAVE 25 accommodates standardized, gamma-irradiated Cellbag™ bioreactor bags in volumes from 10 L to 25 L (with scalable bag options supporting up to 2000 L in aligned process configurations). Bags feature integrated optical pH/DO sensors, sterile connectors, and gamma-stable polymeric construction validated per USP and cytotoxicity and extractables testing. All hardware components meet IEC 61000-6-2/6-4 electromagnetic compatibility standards and are CE-marked for industrial use. System qualification packages include full material traceability (CoA, CoC), FAT/SAT reports, and vendor-supplied IQ/OQ protocols aligned with ISO 13485 and ASTM E2500-13 guidelines for pharmaceutical equipment validation.
Software & Data Management
UNICORN™ serves as the central control and data acquisition engine, storing all sensor readings, actuator commands, and alarm events in a structured SQL database with timestamped metadata. Each run generates a compressed .u3d archive containing raw signals, calculated parameters (e.g., kLa, volumetric power input), and user-defined annotations—exportable in CSV, PDF, or ASAM ATX format for regulatory submission. Audit trails record every parameter change, user login/logout, method edit, and report generation event with immutable timestamps and operator ID. Data integrity is further enforced through automatic checksum verification upon file transfer and periodic database consistency checks.
Applications
- Monoclonal antibody (mAb) and recombinant protein production in CHO and HEK293 suspension cultures.
- Process characterization and scale-down modeling for upstream bioprocess development per ICH Q5D and Q8(R2).
- Microcarrier-based expansion of mesenchymal stem cells (MSCs) and induced pluripotent stem cells (iPSCs) under serum-free, xeno-free conditions.
- High-yield baculovirus expression vector system (BEVS) cultivation in Sf9 insect cells.
- Continuous perfusion culture with integrated harvest pumps and inline density monitoring.
FAQ
Is the WAVE 25 suitable for GMP manufacturing?
Yes—the system is qualified for use in regulated environments, with UNICORN software validated to 21 CFR Part 11 and GAMP 5 requirements, and hardware meeting ISO 13485 design controls.
Can the same Cellbag™ be used across different WAVE platforms?
Cellbag™ bags are platform-specific; WAVE 25 bags are mechanically and sensor-compatible only with WAVE 25 and WAVE 250 systems—not with legacy WAVE 10 or WAVE 500 models.
What is the maximum allowable working volume for a single Cellbag™ on the WAVE 25?
The standard Cellbag™ for WAVE 25 supports up to 25 L working volume; however, the system’s control algorithms and hardware calibration are validated for operation between 10 L and 25 L.
How is temperature uniformity ensured across large-volume bags?
Heating output is dynamically modulated based on real-time weight measurement and bag geometry—ensuring ±0.3 °C stability at setpoint and <1.5 °C spatial gradient across the liquid column.
Does the system support remote monitoring via SCADA?
Yes—OPC UA server functionality enables secure, firewall-friendly integration with third-party MES, SCADA, or DCS platforms for centralized batch oversight and alarm forwarding.