Scienova XDB High-Throughput Dialysis System
| Brand | Scienova |
|---|---|
| Origin | Germany |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | Scienova XDB |
| Dye Method | Colorimetric |
| Removal Method | Solvent-Removable |
| Developer Type | Dry |
| Appearance | Colorless |
| Density | 0.44 g/cm³ |
| Membrane Material | Low-Binding Regenerated Cellulose (Bio-Xell®) |
| MWCO Options | 2, 3.5, 6–8, 12–14, 20, 140 kDa |
| Operating Temperature Range | 1–60 °C |
| pH Stability Range | 5–8 |
| Sterilization | Autoclavable (121 °C, 20 min) |
| Format Compatibility | 96-well microplates, deep-well plates, robotic liquid handlers |
Overview
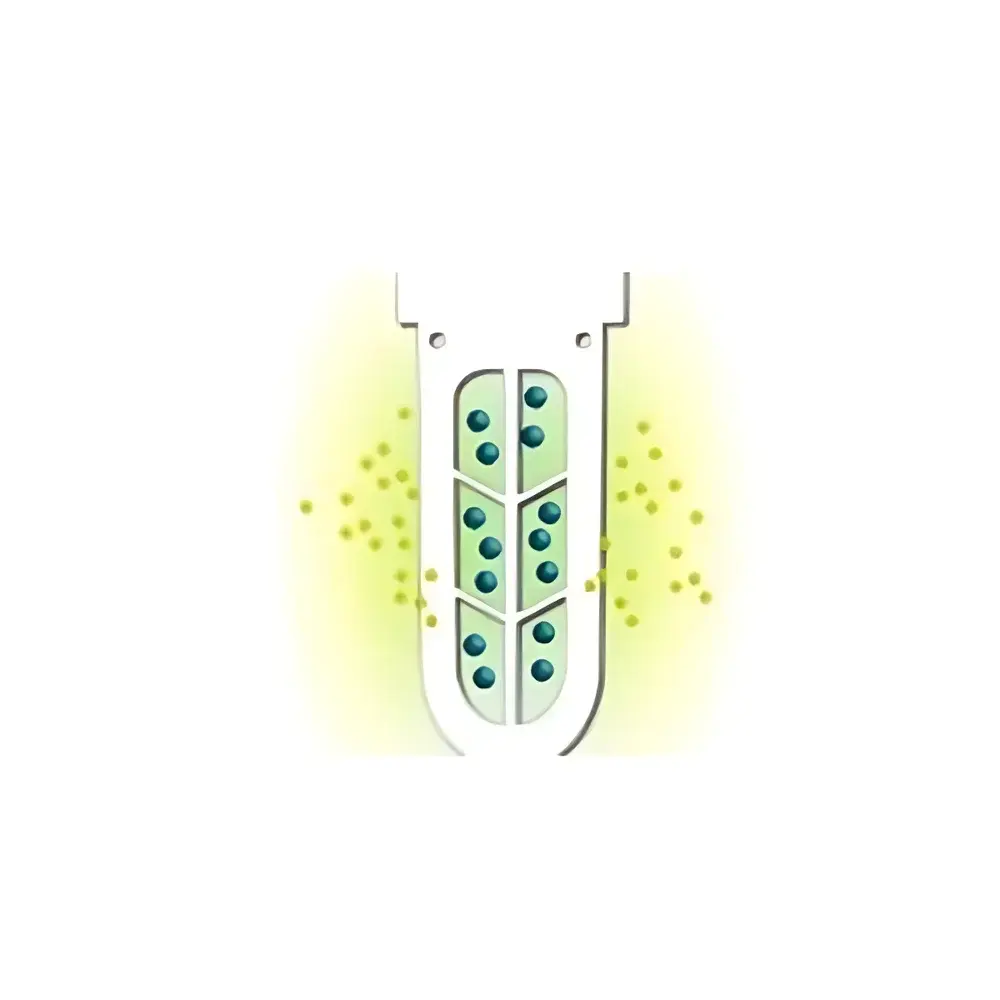
The Scienova XDB High-Throughput Dialysis System is an engineered solution for rapid, scalable, and reproducible buffer exchange and small-molecule removal in biomolecular purification workflows. Unlike conventional dialysis relying on passive diffusion across suspended membrane bags, the XDB employs a microplate-integrated, box-style dialyzer architecture that leverages optimized surface-area-to-volume ratios and minimized diffusion path lengths. Its operation is grounded in the physicochemical principle of concentration-driven solute diffusion across semi-permeable membranes—governed by Fick’s first law—where molecular separation occurs based on hydrodynamic radius relative to the membrane’s nominal molecular weight cutoff (MWCO). Designed specifically for laboratories performing high-volume sample preparation in drug discovery, biopharmaceutical development, and structural biology, the XDB enables parallel processing of up to 96 samples per run without cross-contamination, while maintaining native conformation integrity of proteins, nucleic acids, and protein–ligand complexes. It does not perform chromatographic or electrophoretic separation; rather, it serves as a critical desalting, detergent removal, and buffer-exchange step prior to downstream analytical or functional assays.
Key Features
- Microplate-compatible, single-use dialysis boxes with integrated low-binding regenerated cellulose membranes (Bio-Xell®), available in six MWCO options (2–140 kDa) to match diverse biomolecular size ranges.
- Reduced dialysis time by 2–12× compared to traditional bag-based methods, due to geometric optimization: fixed 0.5 mm diffusion distance, uniform 2.5 cm² effective membrane area per well, and controlled 100–300 µL sample volume capacity.
- 98% average biomolecule recovery rate validated for IgG, BSA, dsDNA (≥5 kb), and tRNA under standard conditions (4°C, 12 h, 3 buffer exchanges), minimizing loss during critical pre-analytical steps.
- Fully compatible with automated liquid handling systems—including Hamilton STAR, Tecan Freedom EVO, and Agilent Bravo—enabling GLP-compliant, walk-away processing in regulated environments.
- Autoclavable design (121°C, 20 min) supports terminal sterilization without membrane degradation or leachables, meeting ISO 13485 requirements for device manufacturing and QC labs.
- No pre-wetting or conditioning required; ready-to-use format eliminates variability from manual membrane hydration and reduces hands-on time by >70% versus roll-type dialysis tubing.
Sample Compatibility & Compliance
The Scienova XDB accommodates a broad range of biological matrices including clarified lysates, plasma fractions, enzymatic digests, synthetic oligonucleotides, and virus-like particles (VLPs). It has been verified for compatibility with common formulation buffers (e.g., PBS, Tris-HCl, HEPES), chaotropic agents (≤1 M urea), non-ionic detergents (e.g., Triton X-100, Tween-20), and organic co-solvents (≤10% DMSO, acetonitrile). All membrane lots undergo endotoxin testing (<0.1 EU/mL) and heavy metal screening (Pb, As, Cd <1 ppm) per USP <85> and <232>. The system conforms to ISO 10993-5 (cytotoxicity), ISO 10993-10 (irritation/sensitization), and ASTM F2459-20 (performance standards for dialysis devices). For regulated applications, full traceability documentation—including CoA, CoC, and lot-specific MWCO verification reports—is provided with each shipment.
Software & Data Management
While the XDB itself is hardware-only, its integration into digital lab ecosystems is supported via standardized plate map definitions (.csv/.xlsx) and LIMS-compatible barcoding (1D/2D QR codes on individual units). When deployed with compatible automation platforms, process parameters—including dwell time, buffer exchange cycles, temperature logging, and pipetting coordinates—are recorded with audit-trail capability compliant with FDA 21 CFR Part 11 and EU Annex 11. Instrument control scripts (Python, LabVIEW) are available upon request for custom protocol development. All validation protocols—including recovery efficiency, carryover assessment, and MWCO verification—align with ICH Q5A(R2) and USP <1058> guidelines for analytical instrument qualification.
Applications
- High-throughput desalting of enzymatically digested peptides prior to LC-MS/MS analysis.
- Buffer exchange of monoclonal antibodies from ammonium sulfate precipitation eluates into formulation buffers for stability studies.
- Removal of free fluorophores or crosslinkers after labeling reactions to reduce background in SPR and BLI binding assays.
- Preparation of ligand-free protein samples for ITC and DSF thermal shift assays.
- Purification of CRISPR-Cas ribonucleoprotein (RNP) complexes from residual sgRNA and Cas9 contaminants prior to cellular delivery.
- Downstream processing of extracellular vesicles (EVs), where detergent removal and isotonic buffer exchange are essential prior to NTA or cryo-EM analysis.
FAQ
Can the XDB be reused after autoclaving?
No. Each unit is designed for single-use only, even after sterilization, to ensure consistent MWCO performance and eliminate carryover risk.
Is the Bio-Xell® membrane compatible with acidic or alkaline buffers?
Yes, within pH 5–8. Outside this range, prolonged exposure may cause hydrolytic degradation of the cellulose matrix.
What is the maximum recommended sample viscosity for reliable diffusion kinetics?
Viscosities up to 15 cP (e.g., 20% glycerol solutions) are supported; above this, diffusion rates decline nonlinearly and require extended equilibration times.
Does the XDB support kinetic dialysis monitoring?
Not natively—but real-time sampling is possible using compatible robotic arms equipped with low-volume probes, enabling time-resolved small-molecule depletion profiling.
How is MWCO verified for each production lot?
Via standardized dextran fractionation assay (ASTM D5225-18) with HPLC-SEC quantification, with ±10% tolerance against nominal cutoff values.