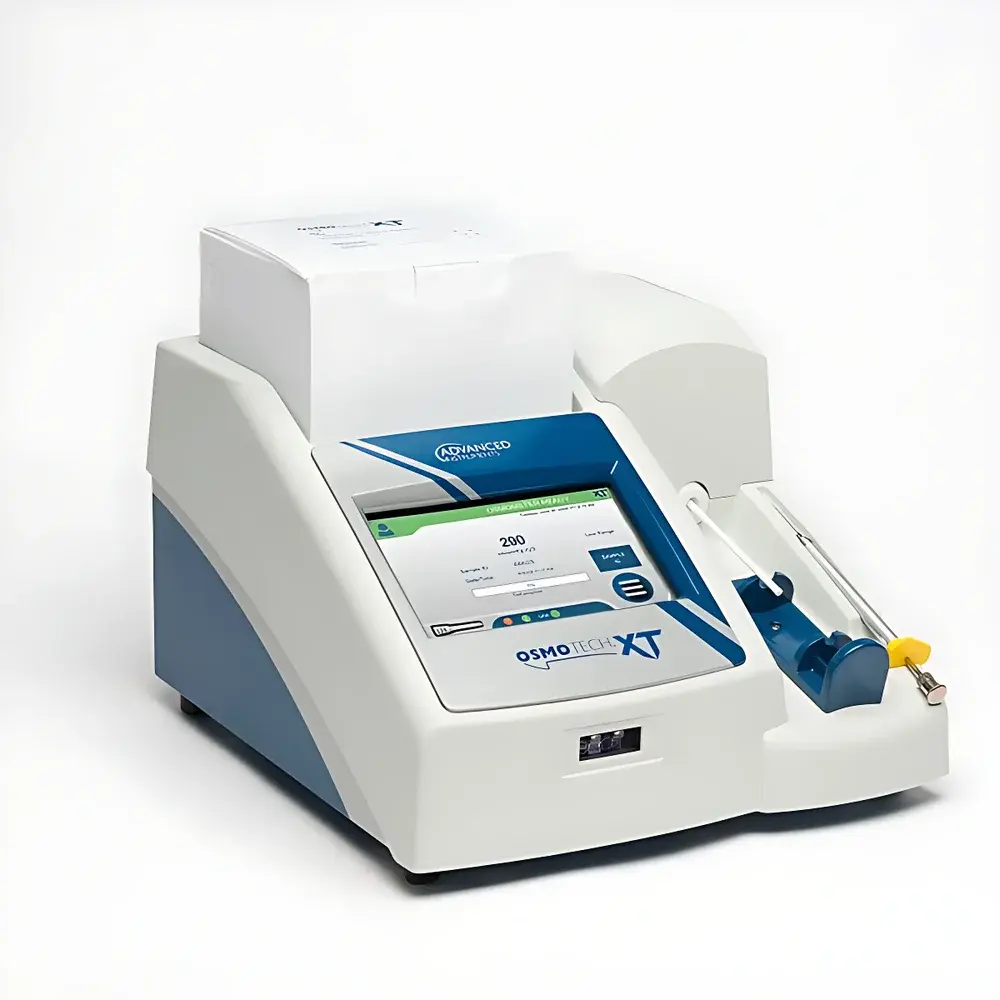
Advanced Instruments OsmoTECH XT Single-Channel Micro-Osmometer
| Brand | Advanced Instruments |
|---|---|
| Origin | USA |
| Manufacturer | Yes |
| Import Status | Imported |
| Model | OsmoTECH XT |
| Price | Upon Request |
Overview
The Advanced Instruments OsmoTECH XT Single-Channel Micro-Osmometer is a cryoscopic (freezing point depression) osmometer engineered for precision and reproducibility in pharmaceutical, biotechnology, and clinical laboratory environments. It determines osmolality by measuring the freezing point depression of aqueous solutions—a thermodynamically rigorous method directly traceable to the colligative properties of solutes. The instrument employs a high-stability thermistor-based detection system with proprietary sample cooling kinetics, enabling accurate quantification across a broad dynamic range up to 4000 mOsm/kg H₂O. Its single-channel architecture prioritizes analytical integrity over throughput, making it especially suited for regulated workflows where sample scarcity, matrix complexity, and method validation are critical considerations.
Key Features
- Freezing point depression measurement principle compliant with USP <785>, EP 2.2.35, and ISO 11348-3 for osmolality determination
- Minimum sample requirement of only 20 µL—ideal for monoclonal antibodies, viral vectors, lyophilized reconstitutes, and other high-value biopharmaceuticals
- Automated calibration verification using dual-point reference standards (e.g., 0 mOsm/kg and 2000 mOsm/kg NaCl solutions)
- Integrated thermal equilibration chamber minimizing ambient drift and ensuring inter-run consistency
- Robust mechanical design with sealed optical path and vibration-damped sample holder to suppress nucleation artifacts
- Self-diagnostic routines for thermistor linearity, refrigeration cycle stability, and stirrer motor performance
Sample Compatibility & Compliance
The OsmoTECH XT accommodates a wide range of sample types—including protein formulations, cell culture supernatants, dialysates, ophthalmic solutions, and vaccine suspensions—without requiring dilution or pre-treatment in most cases. Its optimized freezing protocol mitigates supercooling in viscous or surfactant-containing matrices (e.g., polysorbate-80–stabilized mAb formulations). The system meets essential regulatory expectations for GxP environments: full audit trail capability (user ID, timestamp, result, calibration status), electronic signature support per FDA 21 CFR Part 11, and configurable data retention policies aligned with ALCOA+ principles. All firmware and calibration records are stored internally with SHA-256 hash verification to ensure data integrity during GLP or GMP audits.
Software & Data Management
Instrument control and data analysis are managed via OsmoTECH Connect software (v4.2+), a Windows-based application validated for IQ/OQ/PQ execution. The software supports customizable report templates conforming to internal SOPs or external submission requirements (e.g., CMC sections of IND/BLA dossiers). Raw thermogram data—including temperature vs. time curves, nucleation onset detection points, and derivative-based freezing point identification—are exportable in CSV and PDF formats. Network deployment options include secure LAN integration with LIMS via ASTM E1384-compliant messaging, and optional TLS 1.2–encrypted cloud backup for disaster recovery. Software updates undergo version-controlled release testing with documented change impact assessments.
Applications
- Final container release testing of parenteral drug products per USP <785>
- Stability-indicating assay for freeze-thaw cycling studies of biologics
- Formulation development support—buffer optimization, excipient screening, and tonicity adjustment
- Quality control of raw materials including human serum albumin, polysaccharide carriers, and synthetic peptides
- Process monitoring of ultrafiltration/diafiltration steps in downstream purification
- Supporting comparability protocols during manufacturing site transfers or process changes
FAQ
What sample volume is required for reliable measurement?
A nominal volume of 20 µL is sufficient; however, consistent pipetting technique and low-retention tips are recommended to minimize carryover and surface tension effects.
Does the OsmoTECH XT require daily calibration?
No—calibration verification is performed before each analytical run using certified reference standards; full calibration is recommended weekly or after maintenance.
Can the instrument measure non-aqueous or highly viscous samples?
It is designed exclusively for aqueous solutions; samples with >15 cP viscosity or significant non-volatile organic content may require method adaptation and prior validation.
Is the system compatible with 21 CFR Part 11 compliance?
Yes—audit trail, electronic signatures, role-based access control, and data immutability features are enabled by default in validated software configurations.
How is thermal stability maintained during extended operation?
The instrument uses a dual-stage Peltier cooling system with real-time thermal load compensation and active ambient temperature sensing to maintain ±0.002 °C thermal stability at the measurement cell.