YOKOGAWA Single Cellome™ Unit SU10 Intelligent Nanopipette-Based Intracellular Injection and Aspiration System
| Brand | YOKOGAWA |
|---|---|
| Origin | Japan |
| Manufacturer | YOKOGAWA Electric Corporation |
| Product Category | Imported Instrument |
| Model | Single Cellome™ Unit SU10 |
| Price Range | USD 125,000 – 194,000 (FOB Yokohama) |
Overview
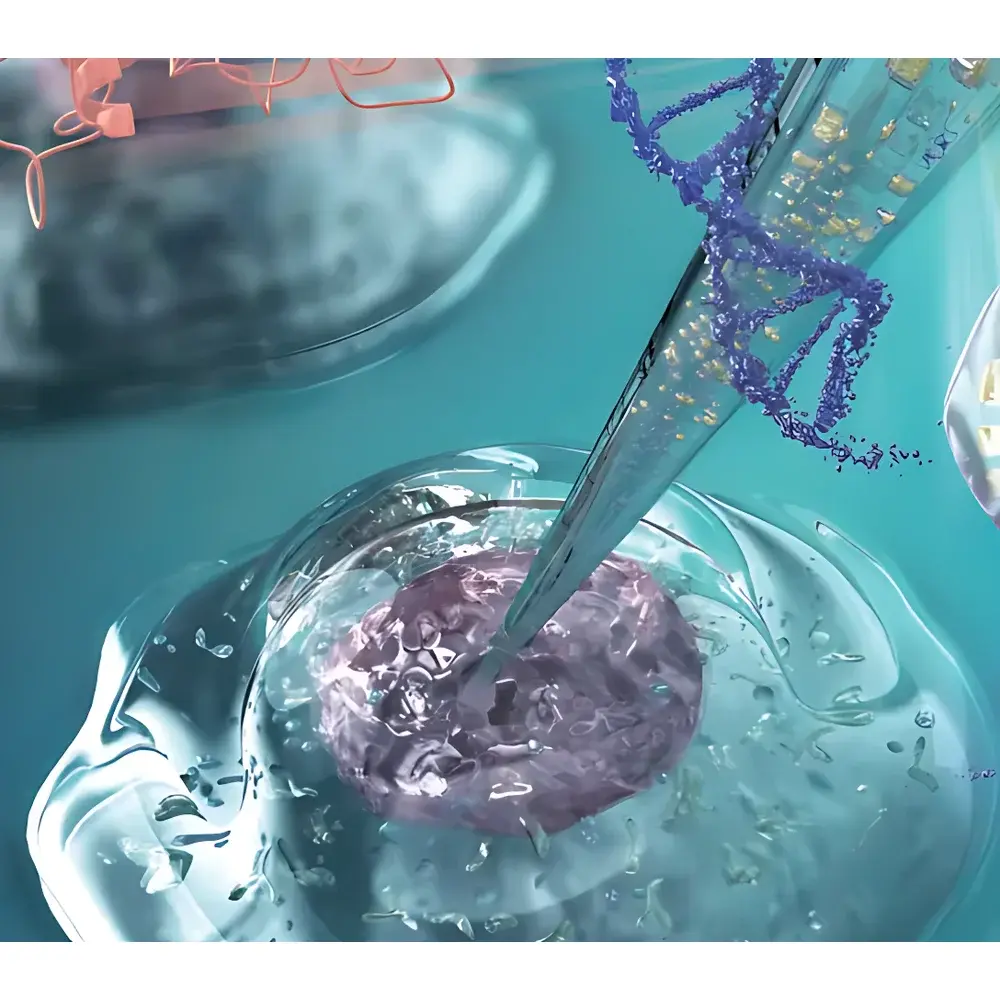
The YOKOGAWA Single Cellome™ Unit SU10 is an automated, nanopipette-based intracellular delivery and aspiration platform engineered for high-precision, minimally invasive manipulation of individual living cells. It operates on the principle of electrophoretically assisted nanoinjection—leveraging controlled electroosmotic flow through a glass nanopipette to achieve programmable, quantitative delivery of biomolecules (e.g., plasmids, CRISPR ribonucleoproteins, siRNA, small-molecule drugs) into subcellular compartments, while simultaneously enabling targeted aspiration of cytosolic or nuclear content. Unlike conventional micropipette systems (typically >1 µm tip diameter), the SU10 employs fused-silica nanopipettes with outer diameters as low as ~100 nm—approximately 1/100th the size of a typical mammalian cell—ensuring negligible membrane disruption, preserved cell viability (>95% post-procedure survival in validated assays), and minimal perturbation of native cellular physiology. The system integrates real-time optical feedback via inverted microscopy, closed-loop Z-axis actuation, and impedance-based contact detection to autonomously identify, approach, penetrate, inject, and retract—all without manual micromanipulation.
Key Features
- True nanoscale intervention: Glass nanopipettes with outer diameters down to 100 nm enable sub-micron precision targeting within single cells
- Fully automated workflow: Autonomous cell surface detection, contact confirmation, membrane penetration (via piezo-assisted gentle indentation), injection/aspiration initiation, and pipette retraction
- Electroosmotic volume control: Programmable injection volumes from 0.1 to 100 fL per pulse, adjustable via voltage amplitude and duration
- High procedural fidelity: ≥95% successful intracellular delivery rate across adherent and suspension cell lines (validated in HeLa, HEK293, primary T cells, and iPSC-derived neurons)
- Real-time monitoring: Integrated differential interference contrast (DIC) and fluorescence imaging compatibility for concurrent morphological and functional assessment
- Modular design: Compatible with standard inverted microscopes (Nikon Ti2, Olympus IX83, Zeiss Axio Observer) and third-party environmental chambers (37°C, 5% CO₂)
Sample Compatibility & Compliance
The SU10 supports a broad range of eukaryotic cell types—including adherent lines (e.g., CHO, A549), suspension cultures (Jurkat, PBMCs), primary cells (human dendritic cells, cardiomyocytes), and stem-cell derivatives—without requiring chemical transfection reagents or viral vectors. All hardware components comply with IEC 61010-1:2012 for laboratory electrical equipment safety. Software architecture conforms to ALCOA+ principles for data integrity; audit trails, electronic signatures, and user-access controls align with FDA 21 CFR Part 11 requirements for regulated environments. System validation documentation supports IQ/OQ/PQ execution under GLP and GMP-aligned workflows.
Software & Data Management
Control and analysis are managed via the proprietary Single Cellome Control Suite v3.2—a Windows-based application supporting script-driven protocol sequencing, multi-parameter event logging (time-stamped injection pressure, voltage, impedance shift, image frame capture), and export of raw data in HDF5 and CSV formats. Built-in machine-learning-assisted focus stabilization compensates for thermal drift during long-duration experiments. Data files include embedded metadata compliant with MIAME and MIAPE standards, facilitating integration with LIMS and ELN platforms (e.g., LabArchives, Benchling). Remote operation via secure TLS-encrypted connection enables off-site monitoring and collaborative experiment coordination.
Applications
- Functional genomics: High-efficiency CRISPR-Cas9 RNP delivery for allele-specific editing without double-strand break accumulation
- Single-cell pharmacology: Intracellular dosing of kinase inhibitors or PROTACs with concurrent live-cell calcium or pH imaging
- Cancer biology: Direct interrogation of tumor heterogeneity via serial aspiration of cytoplasmic content from morphologically distinct subpopulations
- Immunotherapy development: Antigen loading into dendritic cells at defined stoichiometry to assess T-cell activation thresholds
- Regenerative medicine: Precise mitochondrial transfer between stem cells and aged somatic cells to evaluate metabolic rescue kinetics
- Neuroscience: Subcellular injection of optogenetic constructs into dendritic spines for compartmentalized functional mapping
FAQ
What is the minimum viable cell size the SU10 can target?
The system reliably engages cells ≥8 µm in diameter, including small lymphocytes and early-stage progenitors, using adaptive focus-lock and impedance thresholding.
Can the SU10 perform simultaneous injection and aspiration in the same cell?
Yes—sequential or interleaved protocols are supported via time-gated voltage polarity switching, enabling “inject-aspirate” cycles within a single penetration event.
Is nanopipette fabrication included with the instrument?
SU10 ships with a starter kit of 20 pre-fabricated, quality-tested nanopipettes (100–200 nm OD); additional pipettes are available through YOKOGAWA’s certified manufacturing partner BioStinger, Inc.
Does the system support cryo-compatible operation?
No—the SU10 is designed for ambient-temperature, liquid-phase operation only; cryo-integration requires external modification not covered under warranty.
How is calibration traceability maintained?
Each nanopipette lot undergoes SEM-based dimensional certification and electroosmotic flow calibration against NIST-traceable standards; calibration certificates are digitally linked to pipette batch IDs in the software.