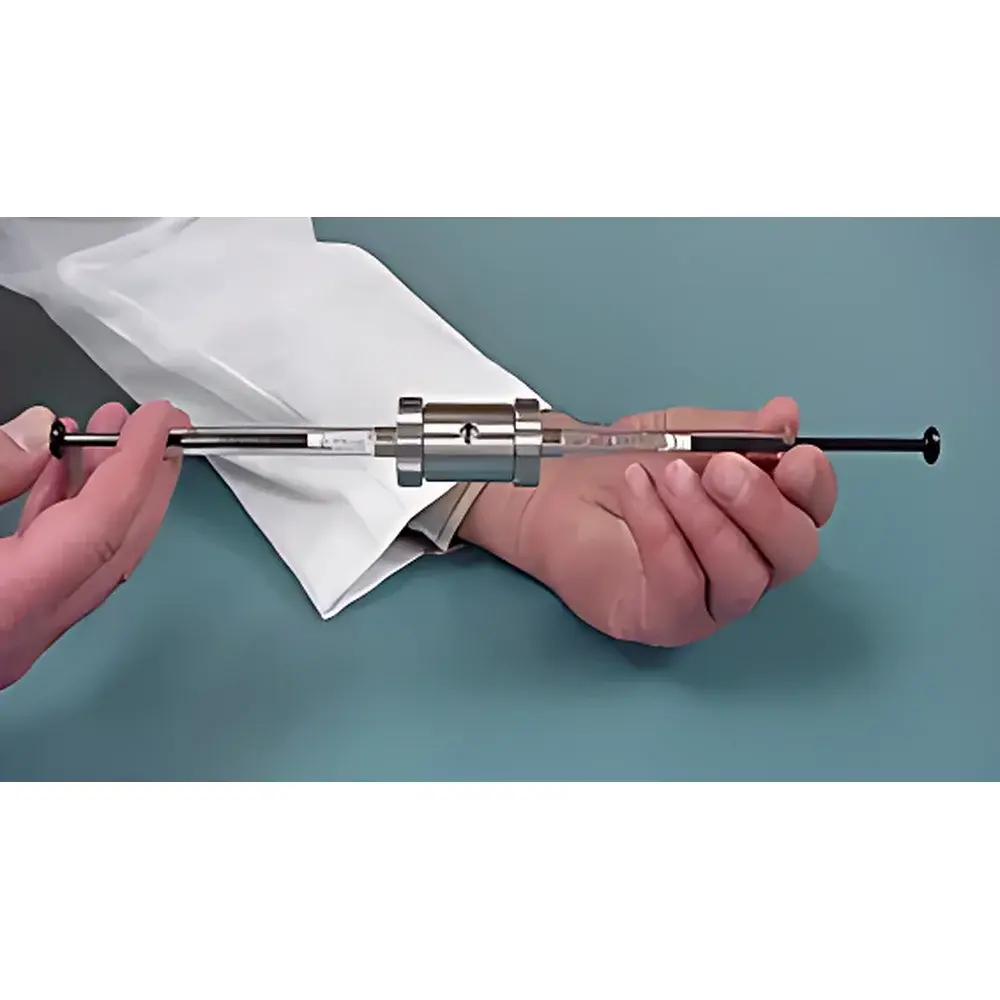
AVESTIN LipoSofast Syringe-Style Liposome Extruder
| Brand | AVESTIN |
|---|---|
| Origin | Canada |
| Model | LipoSofast |
| Sample Volume per Cycle | 1 mL (2 × 500 µL syringes) |
| Minimum Sample Volume | 0.1 mL |
| Residual Volume | 0 µL |
| Available Polycarbonate (PC) Membrane Pore Sizes | 50, 100, 200, 400, and 800 nm |
| Temperature Compatibility | Water-bath compatible for thermal control (e.g., 4–40 °C) |
| Weight | < 2 kg |
| Construction | Borosilicate glass syringes, stainless steel housing, chemically resistant seals |
Overview
The AVESTIN LipoSofast Syringe-Style Liposome Extruder is a precision-engineered, manual extrusion device designed for the controlled size reduction and homogenization of lipid-based nanostructures—including liposomes, lipid nanoparticles (LNPs), exosomes, and other soft vesicular systems. It operates on the principle of mechanical extrusion through track-etched polycarbonate (PC) membranes under defined pressure, enabling reproducible generation of unilamellar or oligolamellar vesicles with narrow polydispersity index (PDI). Unlike high-pressure homogenizers or sonication-based methods, the LipoSofast delivers gentle, low-shear processing—critical for preserving encapsulated therapeutics (e.g., mRNA, siRNA, peptides) and maintaining membrane integrity during repeated extrusion cycles. Its compact, hand-operated architecture eliminates dependency on compressed air, electricity, or complex fluidic systems, making it ideal for benchtop use in formulation development, preclinical research, and GMP-aligned process optimization.
Key Features
- Zero-residual-volume design ensures quantitative recovery of precious samples—no loss due to dead volume or internal retention.
- Modular, fully disassemblable construction enables rapid cleaning, sterilization (autoclavable components), and compatibility with ultrasonic bath decontamination.
- Borosilicate glass syringes provide optical clarity for real-time visual monitoring of extrusion progress and bubble detection.
- Stainless steel body and fluoropolymer-sealed plunger mechanisms ensure chemical resistance to organic solvents (e.g., chloroform, ethanol) and long-term dimensional stability.
- Water-jacket compatibility allows integration with circulating water baths for precise temperature management—essential for thermolabile formulations such as PEGylated liposomes or temperature-sensitive cargo (e.g., CRISPR ribonucleoproteins).
- Ergonomic dual-syringe configuration supports bidirectional extrusion without sample transfer, minimizing contamination risk and operator variability.
Sample Compatibility & Compliance
The LipoSofast accommodates aqueous dispersions, organic-aqueous mixtures, and detergent-stabilized vesicle suspensions. It is routinely validated for use with phosphatidylcholine (PC), DSPC, DOPE, cholesterol, and ionizable lipids across pH 4–9. Membrane pore sizes (50–800 nm) are certified per ASTM F838-22 for bacterial retention and meet ISO 13485-compliant manufacturing standards. Device materials comply with USP Class VI biocompatibility requirements. While not an automated GMP system, its traceable operation (manual force application, defined cycle count, documented membrane lot numbers) supports GLP documentation and FDA 21 CFR Part 11–aligned data recording when paired with electronic lab notebooks (ELNs).
Software & Data Management
The LipoSofast is a standalone mechanical instrument with no embedded electronics or firmware; therefore, it does not include proprietary software. However, its operational parameters—extrusion cycles, membrane pore size, batch temperature, and syringe volume—are fully documentable within laboratory information management systems (LIMS) or ELNs. Users may integrate extrusion logs with dynamic light scattering (DLS) or nanoparticle tracking analysis (NTA) data for correlation of process inputs with final particle size distribution (PSD). AVESTIN provides comprehensive SOP templates aligned with ICH Q5A(R2) and USP guidance for liposome characterization workflows.
Applications
- Preparation of uniform liposomal drug carriers for oncology, vaccine delivery (e.g., mRNA-LNPs), and gene therapy vectors.
- Downstream processing in academic and industrial labs where scalability from microgram-to-milligram scale is required prior to pilot-scale extrusion or microfluidics.
- Quality-by-Design (QbD) studies evaluating the impact of membrane pore size, extrusion temperature, and cycle number on vesicle lamellarity and encapsulation efficiency.
- Reconstitution and sizing of commercially available liposome stocks (e.g., Lipoid, Avanti Polar Lipids) for assay standardization.
- Exosome and extracellular vesicle (EV) size homogenization for flow cytometry calibration and biodistribution studies.
FAQ
What is the recommended number of extrusion cycles for achieving monodisperse liposomes?
Typical protocols specify 11–21 cycles (5–10 passes in each direction) using progressively smaller pore sizes—e.g., 800 nm → 400 nm → 100 nm—to achieve Dv,50 < 120 nm with PDI < 0.15 (measured by DLS).
Can the LipoSofast be used with organic solvents during lipid film rehydration?
Yes—glass syringes and stainless steel housing are compatible with common reconstitution solvents including chloroform/methanol mixtures, ethanol, and isopropanol; however, avoid prolonged exposure to strong oxidizers or hydrofluoric acid.
Is membrane reuse permitted?
PC membranes are single-use per ISO 13408-1 recommendations to prevent cross-contamination and pore deformation; AVESTIN supplies certified, lot-traceable membranes with certificate of conformance.
How is temperature control implemented during extrusion?
The device fits standard 50–100 mL water baths; users secure the assembled extruder vertically using clamps and circulate thermostatically controlled water (±0.5 °C) around the syringe barrel assembly.
Does the LipoSofast support regulatory filing submissions?
While the device itself is not 510(k)-cleared, its design, material certifications, and operational repeatability support inclusion in CMC sections of IND/IMPD dossiers when accompanied by validation reports per ICH Q5B and Q5E.