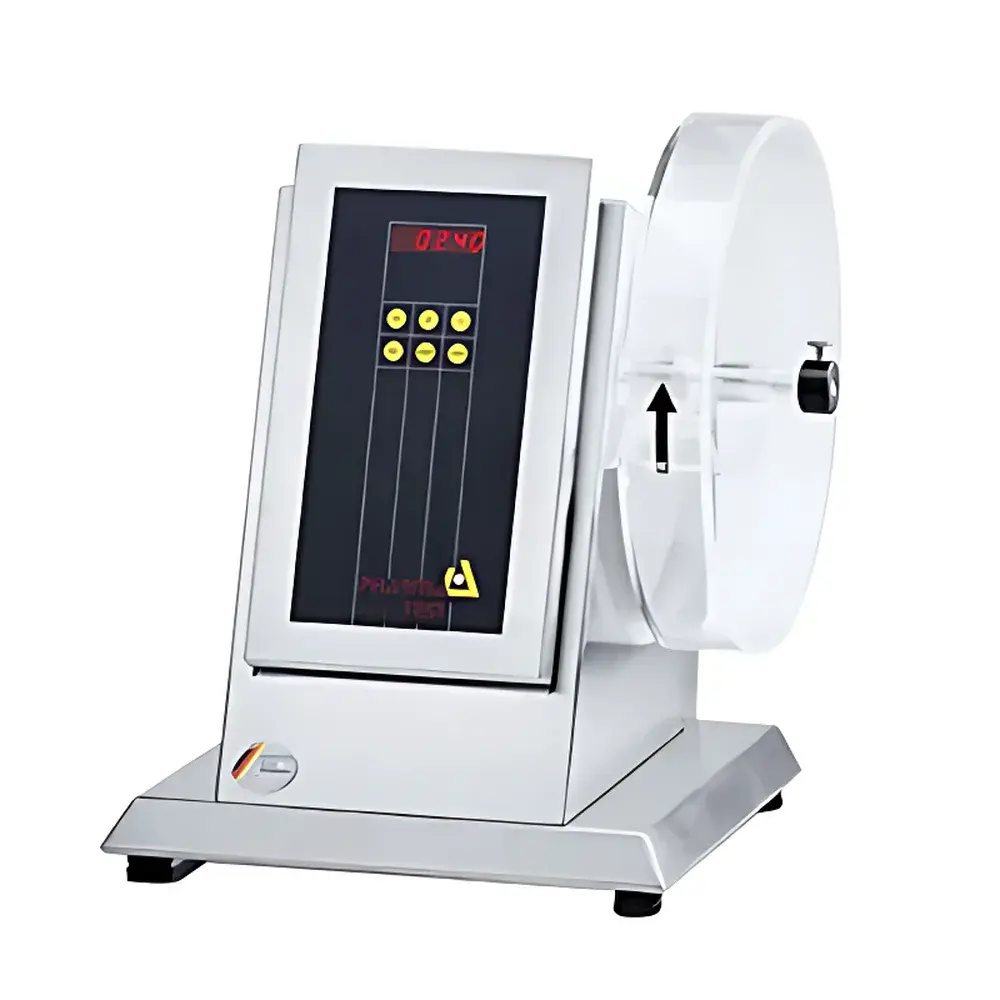
Pharma-test PTF 10E/20E Friability Tester
| Brand | Pharma-test |
|---|---|
| Origin | Germany |
| Model | PTF 10E / PTF 20E |
| Drum Configuration | Single-drum (PTF 10E) or Dual-drum (PTF 20E) |
| Rotation Speed | Fixed at 25 rpm (standard), adjustable 1–9999 rpm (ER variants) |
| Test Duration Range | 1–9999 seconds |
| Rotation Count Range | 1–9999 revolutions |
| Drum Material | Acrylic glass (PMMA), optional anti-static coating |
| Drum Angle | Standard 0° |
| Compliance | USP <1216>, Ph. Eur. <2.9.7>, JP <6.07> |
| Construction | Full stainless-steel housing (AISI 304) |
| Documentation | Factory-supplied IQ/OQ protocols included |
| Interfaces | RS232 serial port, parallel printer interface |
| GMP/GLP Conformance | Designed and validated for regulated pharmaceutical QC environments |
Overview
The Pharma-test PTF 10E/20E Friability Tester is a precision-engineered laboratory instrument designed to quantitatively assess the mechanical robustness of uncoated and coated solid oral dosage forms—primarily tablets—under controlled tumbling conditions. Operating on the principle of rotational attrition, the device subjects samples to repeated collisions against the inner surface of a rotating drum, simulating mechanical stresses encountered during packaging, transportation, and handling. The test measures weight loss (%) after a defined number of revolutions or duration, providing a critical quality attribute for batch release and formulation development. As a fully compliant solution for pharmacopoeial testing, the PTF series implements the standardized methodology outlined in United States Pharmacopeia Chapter , European Pharmacopoeia Chapter , and Japanese Pharmacopoeia Chapter . Its dual configuration—single-drum (PTF 10E) and dual-drum (PTF 20E)—enables flexible throughput scaling without compromising measurement repeatability or regulatory traceability.
Key Features
- Pharmacopoeia-compliant drum geometry and rotation dynamics: fixed 25 rpm speed per USP and Ph. Eur. , with optional ER variants supporting programmable speed (1–9999 rpm) for method development and non-standard investigations.
- Drum construction from high-transparency, impact-resistant acrylic glass (PMMA), available with certified anti-static coating to prevent electrostatic adhesion of fine particulates—critical for low-dose or high-surface-area formulations.
- Optional 10° drum tilt fixture enables reliable friability assessment of oversized tablets (diameter >13 mm), addressing a documented limitation in standard drum configurations.
- Integrated LED display provides real-time feedback of set parameters—including elapsed time (1–9999 s), total revolutions (1–9999), and active drum status—ensuring immediate operator verification prior to test initiation.
- Full stainless-steel (AISI 304) enclosure meets stringent hygiene and corrosion-resistance requirements for cleanroom-integrated QC laboratories operating under cGMP and GLP frameworks.
- Built-in RS232 serial interface and parallel printer port support direct data export to LIMS, electronic lab notebooks (ELN), or local thermal printers—facilitating audit-ready documentation.
Sample Compatibility & Compliance
The PTF 10E/20E accommodates standard tablet sizes up to 13 mm diameter in its default horizontal drum configuration; the optional 10° tilt accessory extends compatibility to larger formats without altering drum volume or rotational inertia. Each drum holds up to 20 g of sample mass (typical for 60–80 tablets depending on density), satisfying pharmacopoeial minimum loading requirements. All hardware and firmware are validated against ISO/IEC 17025:2017 principles for testing laboratories. Factory-supplied Installation Qualification (IQ) and Operational Qualification (OQ) documentation includes calibration certificates, performance verification protocols, and traceable reference standards—fully aligned with FDA 21 CFR Part 11 expectations for electronic records and signatures when paired with compliant software systems.
Software & Data Management
While the base PTF 10E/20E operates via embedded microcontroller logic with no proprietary PC software dependency, its RS232 interface supports integration with third-party laboratory data management platforms. Exported data streams include timestamp, drum ID, runtime, revolution count, and pass/fail status flags—structured for automated parsing into CSV or XML formats. When deployed within regulated environments, the instrument’s deterministic behavior, non-volatile parameter storage, and hardware-based timer ensure full compliance with ALCOA+ data integrity principles (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available). Audit trails—when generated externally via connected systems—can satisfy 21 CFR Part 11 requirements for electronic records retention and reviewer access control.
Applications
- Batch release testing of compressed tablets in pharmaceutical manufacturing facilities.
- Formulation optimization studies evaluating binder efficacy, granulation method impact, and coating integrity.
- Stability program assessments tracking mechanical degradation over accelerated or real-time storage conditions.
- Supplier qualification of excipients where particle hardness and flow-induced attrition influence final tablet robustness.
- Educational use in university pharmacy and pharmaceutics programs for hands-on pharmacopoeial method training.
FAQ
What pharmacopoeial standards does the PTF 10E/20E comply with?
It fully satisfies USP , Ph. Eur. , and JP requirements for apparatus design, rotational speed tolerance (±1 rpm), drum dimensions, and test reporting conventions.
Can the PTF 10E be upgraded to dual-drum operation?
No—PTF 10E and PTF 20E are mechanically distinct platforms; dual-drum functionality requires the PTF 20E chassis and associated drive electronics.
Is the anti-static drum coating validated for GMP use?
Yes—the coating is chemically stable, non-leaching, and supplied with CoA confirming compatibility with ICH Q5C biocompatibility guidelines for contact surfaces.
Does the instrument include calibration tools or reference weights?
A certified reference weight set (10 g, 20 g, 50 g) and digital tachometer are recommended accessories—not included standard—but supported by Pharma-test’s validation service package.
How is cleaning and maintenance performed under GMP conditions?
The drum assembly detaches without tools; all wetted surfaces are autoclavable (121°C, 20 min) or compatible with validated alcohol-based sanitizers per facility SOPs.