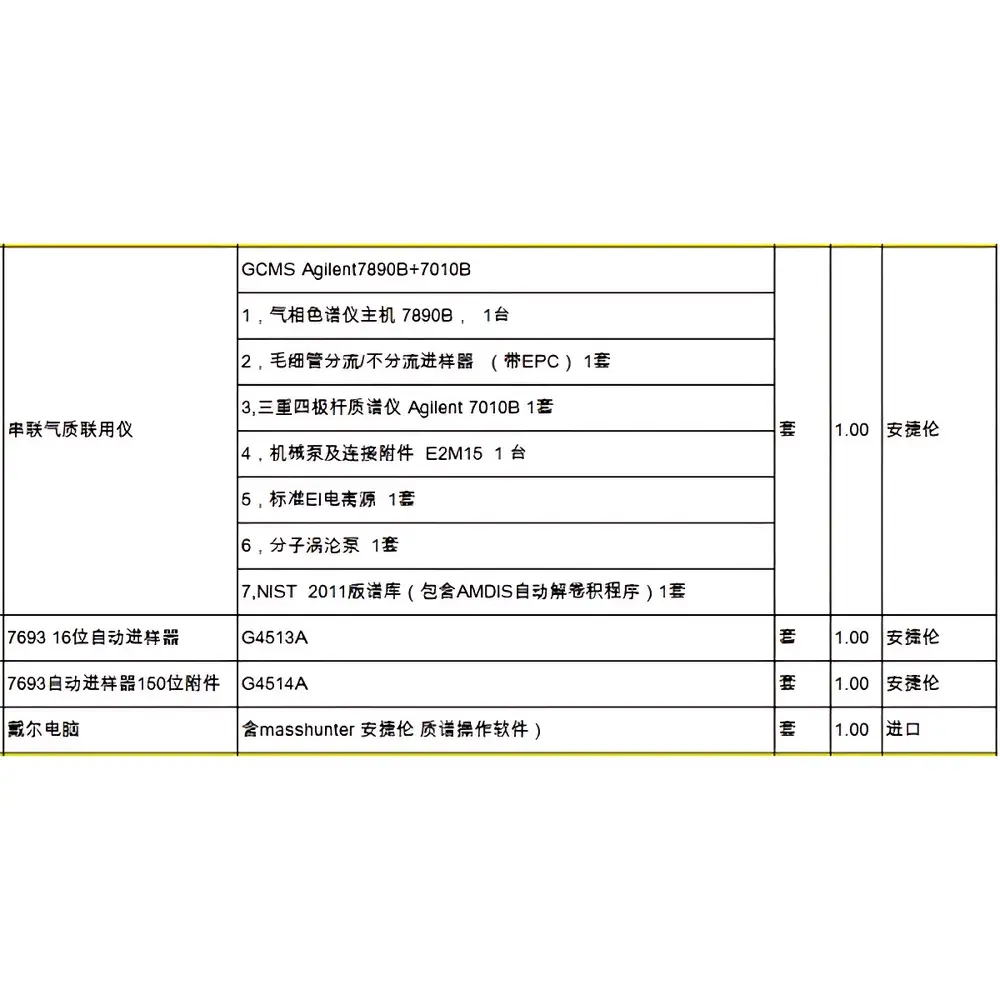
Agilent 7890B GC / 7010B Triple Quadrupole Mass Spectrometer System
| Brand | Agilent Technologies |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Instrument Origin | Imported |
| Model | 7890B + 7010B |
| Price | USD 95,000 (FOB) |
Overview
The Agilent 7890B Gas Chromatograph coupled with the 7010B Triple Quadrupole Mass Spectrometer (GC/MS/MS) is a high-performance, research-grade analytical platform engineered for trace-level quantitation and confirmatory analysis in regulated and non-regulated laboratories. This system operates on electron ionization (EI) and chemical ionization (CI) modes, delivering enhanced selectivity and sensitivity through tandem mass spectrometry (MS/MS) fragmentation—enabling reliable detection and quantification of target analytes in complex matrices such as environmental extracts, food residues, pharmaceutical impurities, and clinical biosamples. The 7890B GC features advanced oven temperature control (±0.01 °C), split/splitless injection with programmable temperature vaporizing (PTV) capability, and integrated carrier gas optimization via Electronic Pressure Control (EPC). The 7010B MS employs three quadrupole mass filters (Q1 → q2 → Q3) to isolate precursor ions, induce collision-induced dissociation (CID) in the RF-only q2 cell, and detect specific product ions—minimizing chemical noise and matrix interferences.
Key Features
- Triple quadrupole architecture with unit-mass resolution across m/z 10–1050, supporting both targeted quantitative analysis (e.g., MRM mode) and qualitative screening (e.g., SIM, full-scan)
- High-efficiency EI/CI source with dual-mode operation, rapid source exchange (<5 min), and bake-out capability up to 350 °C
- Agilent JetClean self-cleaning ion source reduces downtime and maintains consistent sensitivity over extended run sequences
- Integrated GC inert flow path with low-bleed DB-5ms or equivalent capillary columns (0.18–0.25 mm ID, 10–60 m length)
- MassHunter Workstation software v10.x or later, supporting method development, acquisition, processing, and reporting compliant with 21 CFR Part 11 requirements (audit trail, electronic signatures, user access levels)
- Robust vacuum system: turbomolecular pump (260 L/s) backed by a dry scroll pump; base pressure <5 × 10−7 Torr
Sample Compatibility & Compliance
This GC/MS/MS system accommodates liquid, solid, and gaseous samples following standard preparation protocols—including solvent extraction, solid-phase extraction (SPE), QuEChERS, purge-and-trap, and thermal desorption. It supports regulatory compliance workflows aligned with EPA Methods 525.3, 8270, and 1613; ISO 17025-accredited laboratories routinely validate its performance per ICH Q2(R2) guidelines. All hardware and software components meet CE marking requirements and are compatible with GLP/GMP environments when configured with appropriate documentation packages and instrument qualification (IQ/OQ/PQ) support files.
Software & Data Management
Data acquisition and processing are managed exclusively through Agilent MassHunter Workstation software, which provides automated calibration, retention time locking (RTL), spectral deconvolution, and compound identification using NIST/EPA/NIH libraries. Raw data files (.D format) are stored in a secure, timestamped structure with built-in checksum verification. Audit trails record all user actions—including method edits, integration parameter changes, and report generation—with immutable timestamps and operator IDs. Optional extensions include MassHunter Quantitative Analysis with AutoQuant for high-throughput batch processing and MassHunter Unknowns Analysis for non-targeted profiling.
Applications
- Environmental monitoring: pesticide residue analysis in soil/water, dioxin/furan congener profiling, PCB congeners in sediment
- Food safety: mycotoxin quantitation (aflatoxins, ochratoxin A), veterinary drug residues (beta-agonists, sulfonamides), packaging migrants (BADGE, NOGE)
- Pharmaceutical quality control: genotoxic impurity screening (ICH M7), extractables/leachables studies, stability-indicating assays
- Clinical toxicology: confirmation of drugs of abuse (opioids, stimulants, synthetic cannabinoids) in urine and blood matrices
- Forensic chemistry: arson accelerant identification, ignitable liquid residue (ILR) classification per ASTM E1618
FAQ
Is this system fully refurbished and performance-verified?
Yes. Each unit undergoes comprehensive refurbishment at an Agilent-authorized service center, including detector recalibration, vacuum integrity testing, GC inlet/septum replacement, and full system validation against Agilent Performance Verification Test (PVT) standards.
Does it include original Agilent software licenses?
The system ships with perpetual MassHunter Workstation licenses (Quantitative and Qualitative Analysis modules), verified and reactivated under the purchaser’s Agilent contract number.
What documentation accompanies the instrument?
Delivery includes factory service records, PVT reports, IQ/OQ protocol templates, electronic copies of user manuals, and a Certificate of Conformance signed by the authorized distributor.
Can it be integrated into an existing LIMS environment?
Yes. MassHunter supports ASTM E1578-compliant data export formats (CSV, XML, mzML) and offers optional LIMS connectivity via Agilent OpenLab CDS or third-party middleware adapters.
What warranty and service options are available?
Standard coverage includes a 12-month parts-and-labor warranty, remote diagnostics support, and priority access to Agilent-certified field engineers across North America and Europe. Extended service agreements (ESA) are available upon request.