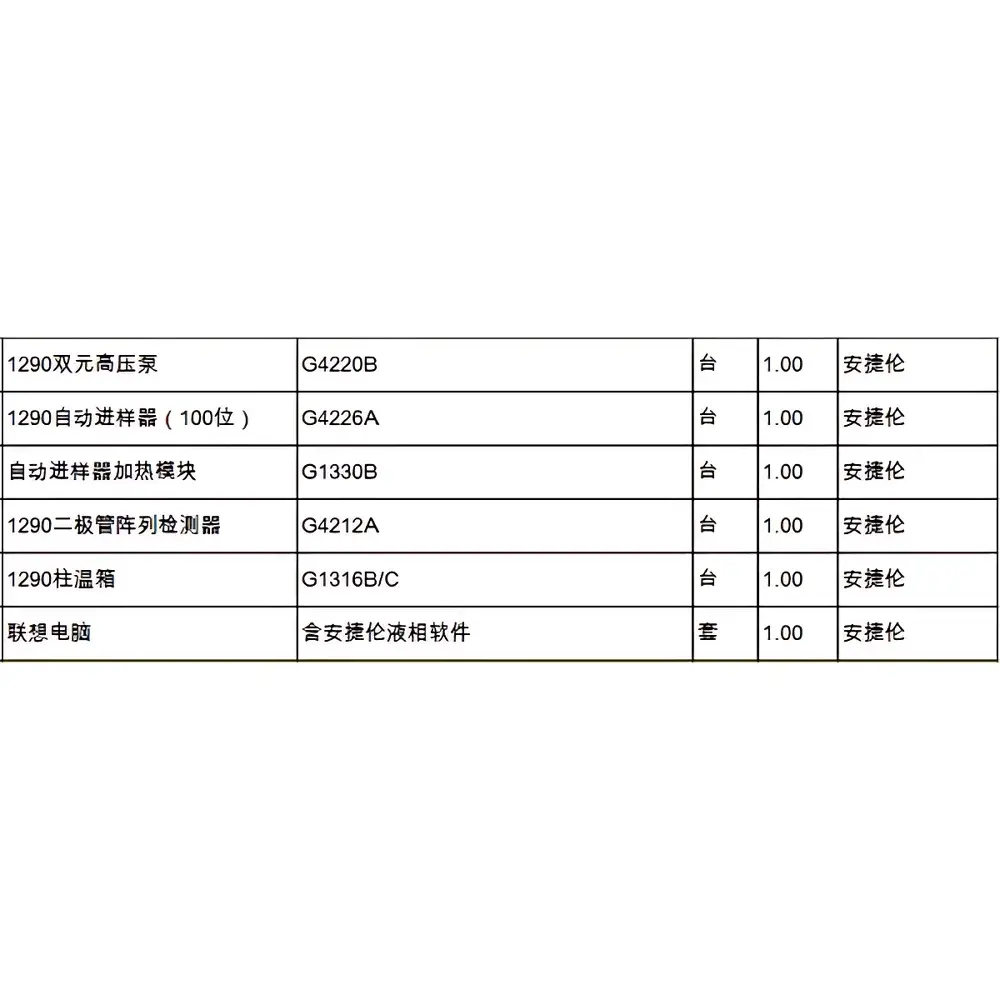
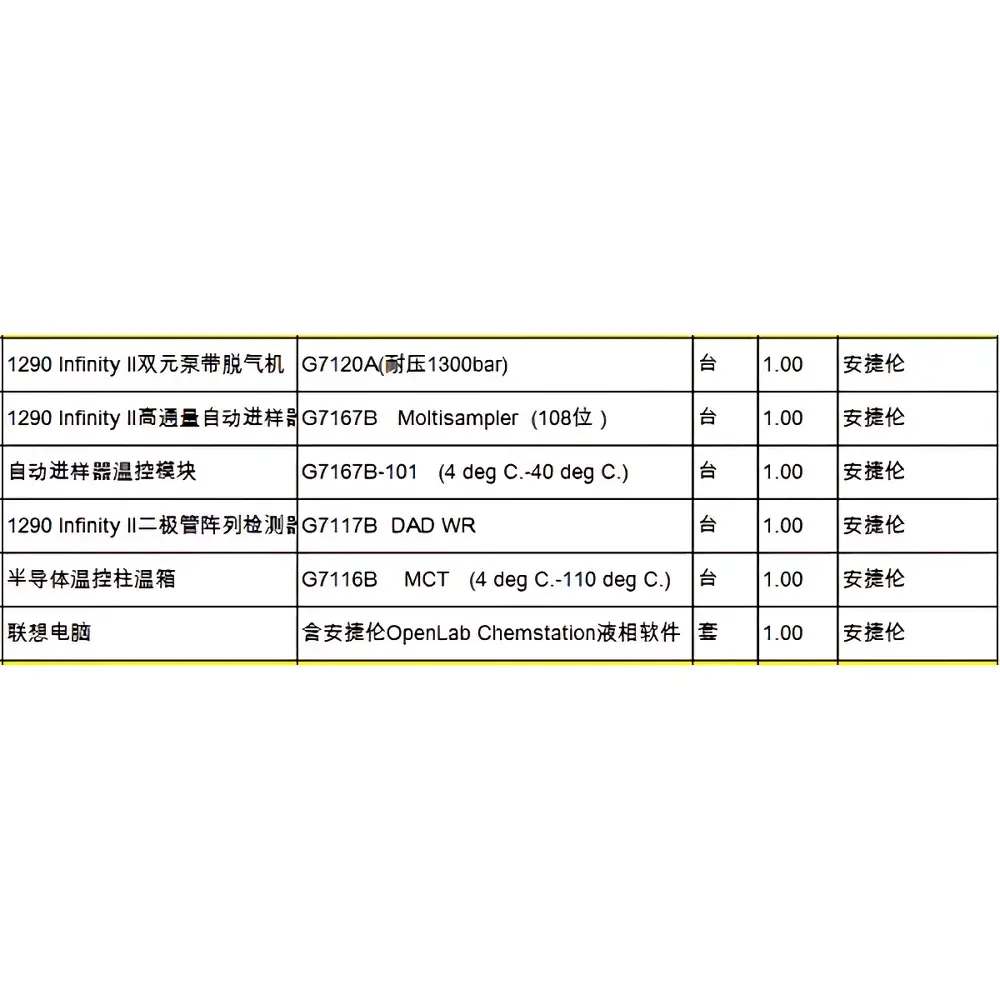
Agilent 1290 Infinity II Binary UHPLC System (Refurbished)
| Brand | Agilent Technologies |
|---|---|
| Origin | USA |
| Configuration | Binary High-Pressure Pump (G4220A), Autosampler (G4226A) with Thermostatted Tray (G1330B), High-Capacity Column Compartment (G1316C, MCT, −20 °C to 110 °C), DAD (G4212A), VWD (G7114B) |
| Maximum System Pressure | 1300 bar |
| Gradient Delay Volume | < 100 µL |
| DAD Sampling Rate | Up to 240 Hz |
| Baseline Noise (DAD, 60-mm flow cell) | ±0.6 µAU/cm |
| Column Capacity | Up to 8 columns |
| Sample Capacity | Up to 6144 vials (with optional stack expansion) |
| Compliance | ASTM D5503, ISO 17025, USP <621>, FDA 21 CFR Part 11 (when operated with compliant CDS) |
Overview
The Agilent 1290 Infinity II Binary UHPLC System (refurbished) is a fully validated, factory-reconditioned ultra-high-performance liquid chromatography platform engineered for laboratories requiring robust, high-resolution separations at pressures up to 1300 bar. Based on Agilent’s InfinityLab architecture, this system implements core UHPLC principles—including minimized system dispersion, low dwell volume gradient delivery, and thermally stabilized fluidic pathways—to deliver exceptional peak capacity, reproducible retention time stability, and sub-ppm carryover performance. Unlike legacy HPLC systems, the 1290 Infinity II employs a dual-piston, actively damped binary pump (G4220A) with embedded firmware-driven flow compensation, reducing pulsation-induced baseline noise and enabling stable gradient elution even with narrow-bore (e.g., 2.1 mm ID) or sub-2-µm particle columns. Its modular design supports both one-dimensional UHPLC and comprehensive two-dimensional LC (LC×LC) configurations without hardware modification—making it suitable for method development, QC release testing, impurity profiling, and regulatory submissions under GLP/GMP environments.
Key Features
- Binary high-pressure pumping system (G4220A) with <100 µL gradient delay volume and real-time pressure feedback control for precise solvent mixing and rapid gradient re-equilibration
- Agilent 1290 Infinity II Multisampler (G4226A) with dual-needle injection, programmable needle wash (including deep-cleaning cycles), and temperature-controlled tray (G1330B) maintaining 4–40 °C stability for thermolabile analytes
- High-capacity thermostatted column compartment (G1316C, MCT) offering −20 °C to 110 °C operation, 8-column switching capability via 1300-bar valves, and active air circulation for uniform thermal distribution
- Dual-wavelength detection architecture: G4212A DAD with 60-mm waveguide flow cell (±0.6 µAU/cm noise, 240 Hz spectral acquisition) and G7114B VWD supporting time-programmed wavelength switching and dual-channel quantitation
- Instrument Control Framework (ICF) compatibility with third-party chromatography data systems including Waters Empower 3/4, Thermo Chromeleon 7.3+, and Shimadzu LabSolutions, ensuring audit-trail-ready operation per FDA 21 CFR Part 11 when configured with electronic signatures
- Smart System Emulation Technology (ISET) enables retention time transfer across Agilent, Waters, and Shimadzu platforms—critical for method migration in multi-vendor labs or contract research organizations
Sample Compatibility & Compliance
The system accommodates standard 1.5–2 mL vials, 96-well and 384-well microplates, and custom sample containers via configurable tray adapters. It supports reversed-phase, HILIC, ion-exchange, size-exclusion, and chiral separations using columns from 1.0 mm to 4.6 mm internal diameter and particle sizes ranging from 1.7 µm to 5 µm. All refurbished units undergo full functional verification per Agilent’s Certified Refurbished Instrument Protocol—including leak testing at maximum pressure, gradient accuracy validation (per ASTM D5503 Annex A3), UV absorbance linearity assessment (NIST-traceable standards), and system suitability testing with USP tailing factor, resolution, and precision criteria. Documentation includes IQ/OQ protocols, calibration certificates, and a 12-month limited warranty covering parts and labor. Units are pre-configured for compliance with ISO/IEC 17025:2017 analytical validation requirements and support full audit trail generation when paired with validated CDS software.
Software & Data Management
The system operates natively with OpenLab CDS (v2.5 or later), providing full instrument control, sequence management, peak integration, and report generation with customizable templates. For labs using alternative CDS platforms, Agilent’s ICF driver suite ensures deterministic command-response timing, secure parameter locking, and synchronized data acquisition—even during high-speed DAD scanning. All raw data files (.agilent) are stored in vendor-neutral HDF5 format, enabling long-term archival integrity and third-party reprocessing. Electronic signatures, user role-based access control, and automatic event logging meet FDA 21 CFR Part 11 requirements for regulated environments. Optional add-ons include MassHunter Quant/Qual for hyphenated LC-MS workflows and ChemStation Legacy Mode for backward-compatible method execution.
Applications
This platform is routinely deployed in pharmaceutical QC labs for assay, dissolution, and stability-indicating methods per ICH Q2(R2); in environmental testing for EPA Method 8330B (PAHs) and 8082A (PCBs); in food safety for mycotoxin screening (AOAC 2012.01); and in biopharma for monoclonal antibody charge variant analysis using cation-exchange UHPLC. Its low system volume and high-pressure tolerance enable rapid method development for complex natural product extracts, polymer additive profiling, and oligonucleotide purity assessment. The dual-detector configuration supports simultaneous UV/VIS and photodiode array spectral confirmation—essential for identification confidence in forensic toxicology and doping control laboratories accredited to ISO 17025.
FAQ
Is this a factory-refurbished unit or dealer-refurbished?
All instruments are Agilent Certified Refurbished—reconditioned at Agilent’s authorized service centers in the USA using OEM parts, original firmware, and full functional testing against factory specifications.
Does the 1-year warranty cover preventive maintenance?
The warranty covers defects in materials and workmanship; scheduled PM services (e.g., pump seal replacement, lamp calibration) are available under separate service agreements.
Can I integrate this system with my existing Empower 4 installation?
Yes—Agilent’s ICF driver is certified for Empower 4.8+ and supports full remote control, real-time status monitoring, and seamless result import into Empower’s processing modules.
What documentation is provided upon delivery?
Each unit ships with a Certificate of Conformance, IQ/OQ execution records, calibration reports (pressure, flow, UV absorbance), and a complete set of electronic manuals in English.
Are consumables and spare parts included?
No—consumables (seals, frits, lamps) and spare modules are ordered separately; however, Agilent’s global parts network guarantees ≤5-business-day lead time for critical spares.