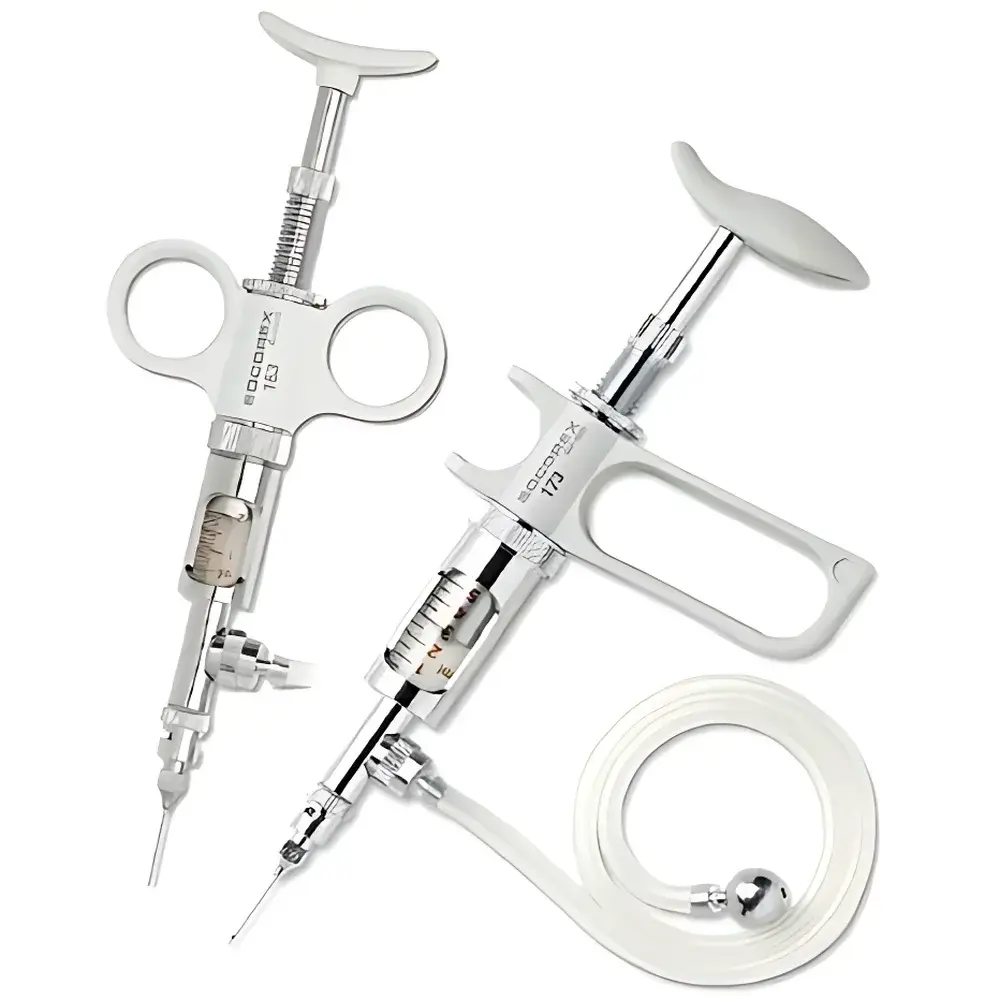
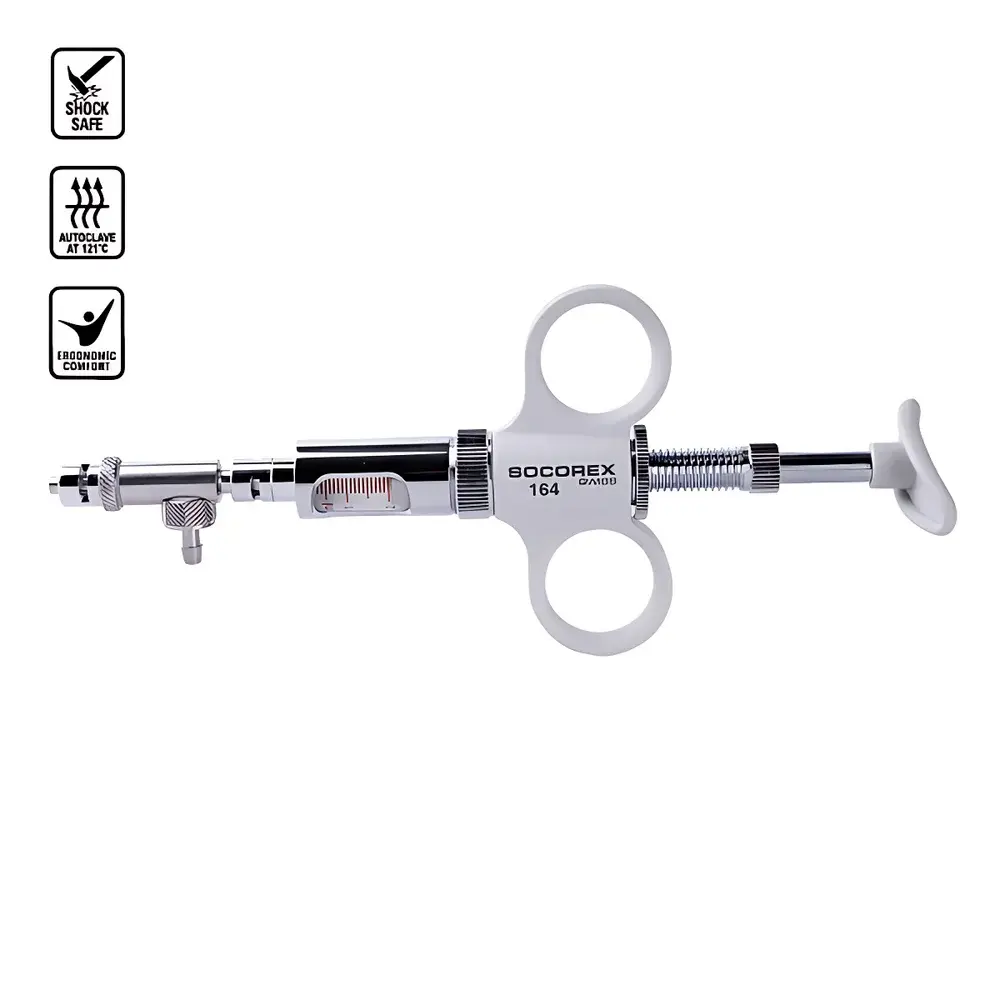
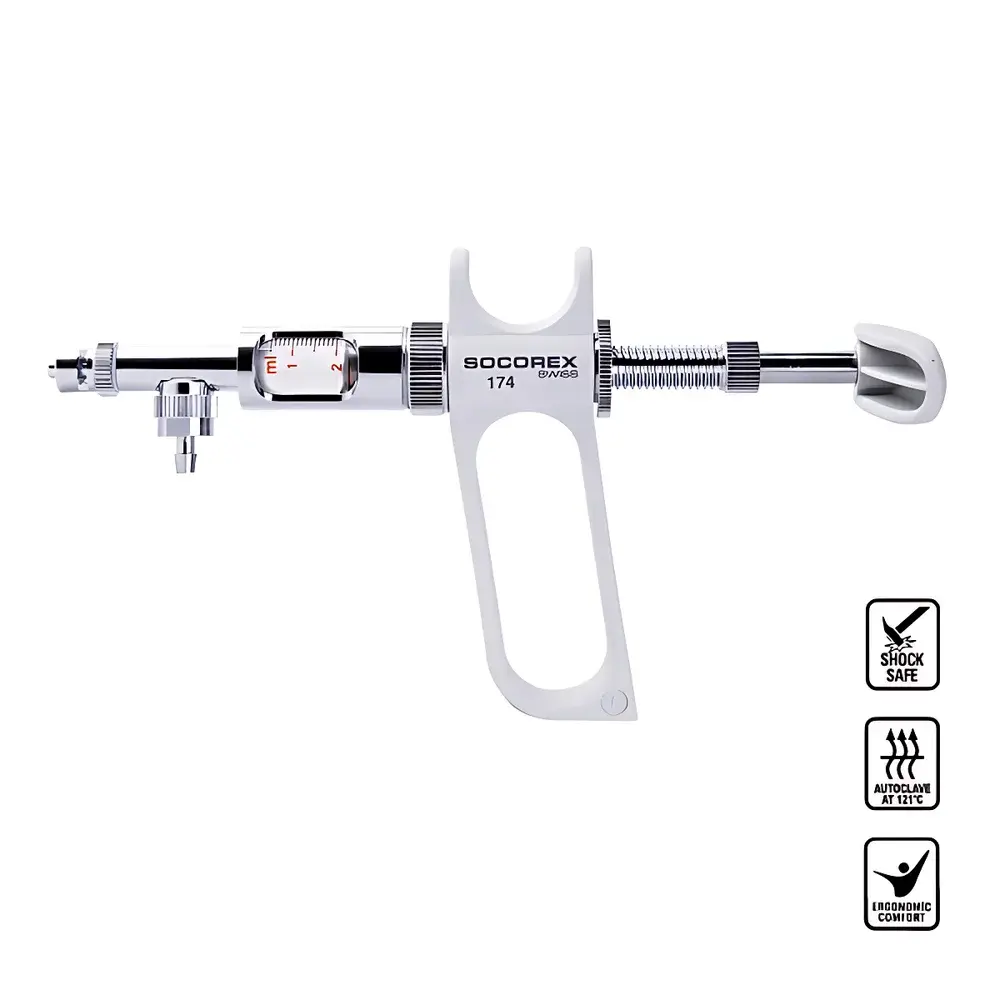
SOCOREX Dosys™ 164-174 Dual-Ring Manual Continuous Dispensing Syringe
| Brand | SOCOREX |
|---|---|
| Origin | Switzerland |
| Model Range | 162–163–164–172–173–174 |
| Type | Single-Channel Manual Dispenser |
| Volume Range | 0.1–10 mL |
| Maximum Capacity | 10 mL |
| Accuracy | ±0.001 mL |
| Sterilization | Autoclavable at 121 °C for 20 min |
| Interface | Luer Lock |
| Construction | Glass barrel with PVC protective sleeve (non-sterilizable) |
| Sealing System | O-ring-free precision-ground piston-cylinder assembly |
| Ergonomics | Dual-ring or pistol-grip handle options |
Overview
The SOCOREX Dosys™ 164–174 is a high-precision, manually operated continuous dispensing syringe engineered for reproducible, contamination-controlled liquid handling in regulated laboratory environments. Unlike conventional piston-driven pipettes, the Dosys™ series employs a positive-displacement, syringe-based fluidic architecture—where volume is defined by mechanical displacement of a calibrated glass barrel and a tightly fitted, O-ring-free piston. This design eliminates seal degradation, hysteresis, and carryover, ensuring long-term volumetric stability across repeated cycles. The 164 (dual-ring handle) and 174 (pistol-grip handle) models are optimized for extended use in applications requiring frequent, low-volume dispensing—such as assay preparation, cell culture reagent distribution, sterile media filling, and preclinical dosing in animal studies. All units comply with ISO 8655-3 for piston-operated volumetric apparatus and are manufactured under strict Swiss quality control standards.
Key Features
- O-ring-free sealing system: Precision-ground borosilicate glass barrel and PTFE-coated stainless-steel piston provide leak-tight performance without elastomeric wear or lubricant migration—ideal for aggressive solvents, acids, and bases.
- Autoclavable construction: Fully sterilizable at 121 °C for 20 minutes (PVC protective sleeve must be removed prior to autoclaving); compatible with standard laboratory steam sterilization protocols.
- Dual-ring ergonomic handle (Model 164): Optimized for sustained manual operation; reduces finger fatigue during repetitive dispensing tasks; integrates intuitive volume adjustment via knurled sleeve with laser-etched, parallax-free scale markings.
- Luer Lock interface: Ensures secure, leak-free connection to standard medical-grade needles, filtration units, tubing adapters, or bottle-top dispensing manifolds.
- Modular disassembly: Composed of only six primary components—barrel, piston, handle, locking collar, scale sleeve, and cap—enabling rapid cleaning, visual inspection, and maintenance without tools.
- Volumetric lock mechanism: Integrated mechanical stop prevents unintentional volume drift during dispensing, guaranteeing consistent delivery across operator shifts and usage conditions.
Sample Compatibility & Compliance
The Dosys™ 164–174 accommodates aqueous buffers, organic solvents (e.g., DMSO, ethanol, acetone), viscous reagents (up to ~500 mPa·s), and biological media—including serum-supplemented DMEM and antibiotic-containing solutions. Its chemically inert wetted path (glass, PTFE, stainless steel) ensures minimal adsorption and no extractables—supporting compliance with USP , ISO 10993-5 cytotoxicity screening, and GLP/GMP documentation requirements. Units are supplied with traceable calibration certificates (optional NIST-traceable certification available). The device meets ISO/IEC 17025 calibration traceability frameworks when serviced by authorized SOCOREX service centers.
Software & Data Management
As a fully manual, non-electronic instrument, the Dosys™ 164–174 requires no firmware, drivers, or software integration. However, its deterministic mechanical design supports audit-ready documentation: each unit bears a unique serial number engraved on the barrel; volume settings are physically fixed and verifiable via caliper measurement against the integrated scale; and batch-level calibration records can be archived per FDA 21 CFR Part 11-compliant SOPs for manual instrumentation. Optional calibration verification kits (including gravimetric test protocols and certified reference weights) facilitate in-house performance qualification per ASTM E2758 and ISO 8655-5.
Applications
- Sterile media and supplement dispensing into multiwell plates or bioreactor bags
- Precision dosing of pharmacological agents in rodent IV/SC/IP administration studies
- Controlled addition of catalysts or initiators in polymer synthesis workflows
- Filtration setup priming and vacuum-assisted liquid recovery
- Calibration standard preparation for HPLC, GC, and ICP-MS sample introduction systems
- Low-volume reagent aliquoting in qPCR master mix assembly and CRISPR editing workflows
FAQ
Can the Dosys™ syringe be used with corrosive solvents such as concentrated HCl or NaOH?
Yes—the glass barrel and PTFE-coated piston are resistant to most common inorganic acids and bases below 6 mol/L concentration. Avoid prolonged exposure to hydrofluoric acid or strong oxidizers (e.g., hot nitric acid).
Is the PVC protective sleeve autoclavable?
No—the PVC sleeve must be removed before autoclaving. It serves only as mechanical protection during handling and storage.
How often should the piston seal be inspected or replaced?
Under normal use, the O-ring-free piston requires no scheduled replacement. Visual inspection for scratches or debris is recommended after each cleaning cycle.
What is the recommended cleaning protocol between uses?
Rinse thoroughly with deionized water or appropriate solvent, followed by air-drying in a laminar flow hood. For sterility-critical applications, perform a final rinse with 70% ethanol and dry under nitrogen.
Does SOCOREX provide calibration services for these dispensers?
Yes—authorized service centers offer accredited calibration per ISO/IEC 17025, including full uncertainty budgets and certificate issuance with traceability to national metrology institutes.