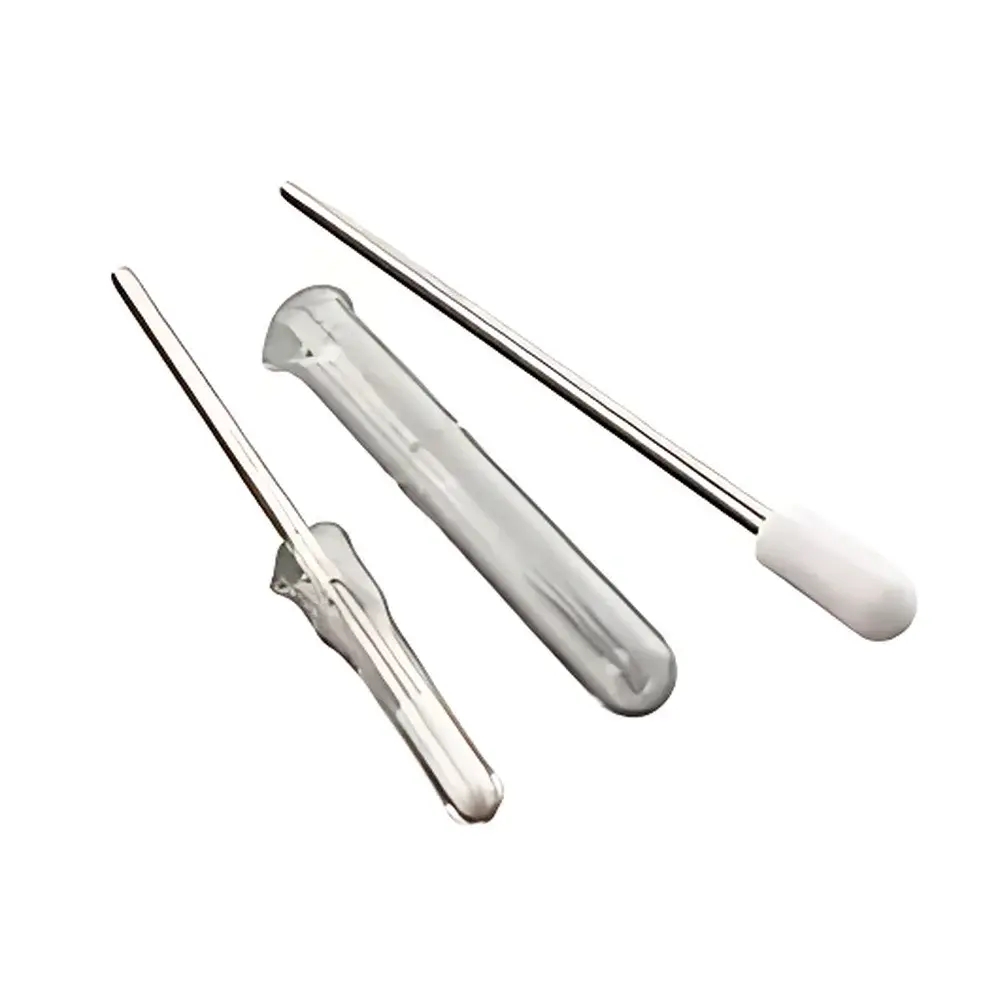
WHEATON Potter-Elvehjem Tissue Homogenizer
| Brand | SOCOREX |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Import Status | Imported |
| Model | Potter-Elvehjem Type |
| Ultrasonic Power | 25 W |
| Operating Frequency | 50 Hz |
| Max Temperature | 100 °C |
| Sample Volume Range | 0.1–55 mL |
| Number of Samples per Run | 1 |
| Grinding Clearance | 0.1–0.15 mm |
| Material | Wheaton 33 Low-Extractable Borosilicate Glass Body with PTFE or Stainless Steel Pestles |
| Sterilization | Autoclavable (121 °C, 20 min) |
| Compliance | USP <661>, ISO 8536-1, ASTM E438 Type I, Class A |
Overview
The WHEATON Potter-Elvehjem Tissue Homogenizer is a precision-engineered manual or motor-driven mechanical homogenization system designed for gentle yet effective disruption of soft biological tissues—including brain, liver, spleen, thymus, and cultured cells—while preserving subcellular integrity and enzymatic activity. Based on the classical Couette flow principle, it operates via controlled shearing between a precisely fitted pestle and a calibrated glass mortar. The clearance gap (0.1–0.15 mm) is maintained within tight tolerances to ensure reproducible shear stress profiles across sample volumes ranging from 0.1 mL (micro-scale) to 55 mL (benchtop scale). Unlike ultrasonic or bead-based disruptors, this device delivers minimal heat generation and no cavitation-induced nucleic acid degradation—making it ideal for preparing native lysates for enzyme assays, organelle isolation, protein extraction, and RNA stabilization workflows.
Key Features
- Three distinct pestle configurations: Standard PTFE-coated (for low adsorption and chemical inertness), stainless steel (for high durability and compatibility with aggressive solvents), and radiating-toothed PTFE (enhanced turbulence and dispersion efficiency in viscous homogenates)
- Mortars fabricated from Wheaton 33 low-extractable borosilicate glass—certified to USP and ASTM E438 Type I Class A standards—ensuring minimal leachables and consistent thermal/mechanical stability
- Fully autoclavable assembly (121 °C, 20 min, saturated steam); all components meet ISO 17664 requirements for reprocessing of reusable medical devices
- Modular sizing: Six standard volume options (0.1, 2, 5, 10, 15, 30, and 55 mL) with matched pestle-motor compatibility; micro-scale (0.1 mL) variant supports single-cell or limited-biopsy applications
- Pestle-motor interface compatible with SOCOREX 980 series homogenizer drives and third-party variable-speed stirrers (e.g., IKA RW 20, Thermo Fisher Precision Plus), enabling programmable speed control (50–1500 rpm) and torque monitoring
Sample Compatibility & Compliance
The WHEATON Potter-Elvehjem system is validated for use with mammalian, avian, and plant soft tissues, primary cell suspensions, and organoid fragments. It avoids denaturation artifacts associated with freeze-thaw cycles or detergent overexposure, supporting GLP-compliant sample preparation for preclinical toxicology studies. All glassware complies with ISO 8536-1 for pharmaceutical glass containers and meets FDA-recommended extractables testing protocols per USP . PTFE components conform to FDA 21 CFR 177.1550 for food-contact polymers. Stainless steel pestles are passivated per ASTM A967 and certified for biocompatibility per ISO 10993-5 (cytotoxicity) and ISO 10993-10 (irritation/sensitization).
Software & Data Management
While the base Potter-Elvehjem unit is manually operated, integration with SOCOREX’s optional digital homogenizer controller (Model HMC-200) enables full traceability: real-time RPM logging, run duration timestamping, and user ID authentication. Data export conforms to CSV and XML formats compliant with 21 CFR Part 11 Annex 11 requirements. Audit trails include parameter change history, calibration events, and sterilization cycle records—supporting regulatory submissions under ICH M7, ICH Q5A, and EU GMP Annex 11.
Applications
- Preparation of subcellular fractions (mitochondria, nuclei, microsomes) without membrane rupture
- Homogenization of fresh-frozen tissue prior to Western blotting or ELISA quantification
- Isolation of intact ribosomes and polysomes for translational profiling
- Generation of uniform single-cell suspensions for flow cytometry and scRNA-seq library prep
- Controlled lysis of Gram-negative bacteria for periplasmic protein recovery
- Standardized tissue processing in ISO/IEC 17025-accredited reference laboratories
FAQ
What is the recommended rotational speed range when coupling the pestle to a motor drive?
For optimal homogenization without foaming or overheating, operate between 300–900 rpm depending on tissue type and viscosity; start at 300 rpm and incrementally increase while monitoring temperature rise.
Can the same mortar be used interchangeably with PTFE and stainless steel pestles?
Yes—mortar dimensions are standardized across all pestle variants; however, avoid mixing pestle materials within the same batch to prevent cross-contamination and differential wear patterns.
How often should the pestle-motor clearance be verified?
Verify gap tolerance (0.10 ± 0.02 mm) using calibrated micrometers before first use, after every 50 autoclave cycles, and following any impact event or visible abrasion.
Is the 0.1 mL micro-homogenizer suitable for proteomics-grade sample prep?
Yes—the ultra-low dead volume and borosilicate glass surface minimize protein adsorption; validated for >95% recovery of spiked BSA standards (1–10 µg/mL) in LC-MS/MS workflows.
Does the system support Good Manufacturing Practice (GMP) documentation?
Full validation packages—including IQ/OQ/PQ protocols, calibration certificates, and material traceability dossiers—are available upon request for regulated manufacturing environments.