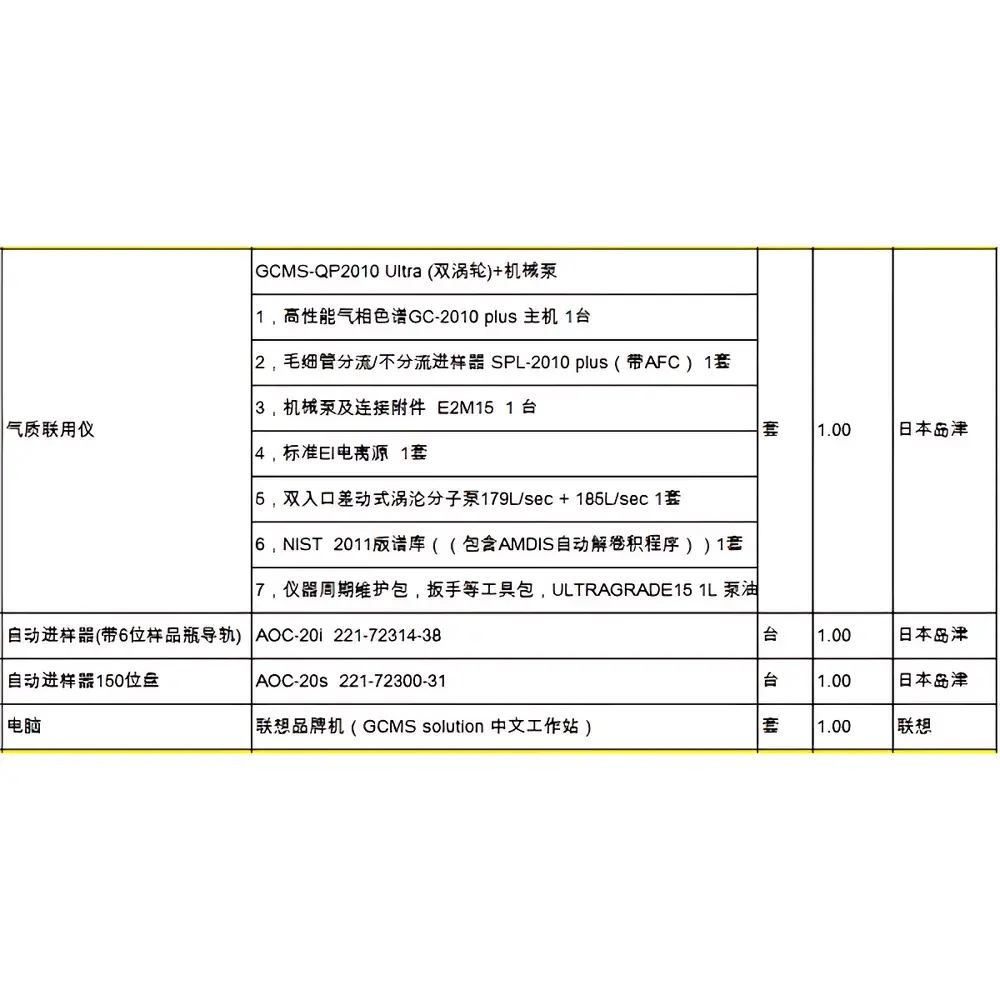
Shimadzu GCMS-QP2010S Used Gas Chromatograph-Mass Spectrometer System
| Brand | Shimadzu |
|---|---|
| Origin | Japan |
| Model | GCMS-QP2010S |
| Mass Spectrometer Type | Single Quadrupole |
| Scan Speed | Up to 10,000 u/sec |
| Ion Source | Front-Opening EI/CI Dual-Mode Ion Source |
| Autosampler | AOC-20i with 100-Position Tray (AOC-20s) |
| Gas Chromatograph | GC-2010 |
| Software | LabSolutions GCMS (vX.X, compliant with FDA 21 CFR Part 11 audit trail requirements) |
| Operational Age | 3–4 years (Total Service Life: ~7 years) |
| Warranty | 6 months limited hardware warranty |
| Vacuum System | Integrated turbomolecular pump assembly |
| Sample Introduction Options | Capillary inlet, Direct Insertion Probe (DIP), SPME, Headspace, Purge-and-Trap, Pyrolysis (PY-GCMS-ready) |
Overview
The Shimadzu GCMS-QP2010S is a high-performance, single-quadrupole gas chromatography–mass spectrometry (GC-MS) system engineered for robust routine analysis in environmental, pharmaceutical, food safety, and petrochemical laboratories. Built on Shimadzu’s proven GC-2010 gas chromatograph platform and integrated with the QP2010S mass spectrometer, this system delivers reliable electron ionization (EI) and optional chemical ionization (CI) capabilities with real-time spectral acquisition at up to 10,000 atomic mass units per second. Its front-opening ion source architecture eliminates the need to vent the vacuum chamber during routine maintenance—enabling rapid cleaning of ion optics, replacement of filaments, or source alignment without compromising system integrity or requiring extended pump-down cycles. The instrument operates under high-vacuum conditions maintained by a dedicated turbomolecular pumping station, ensuring stable sensitivity and long-term signal reproducibility across multi-day analytical sequences.
Key Features
- Front-access ion source design for tool-free, vacuum-intact maintenance of EI/CI sources, filaments, and quadrupole assemblies
- High-speed scanning capability (up to 10,000 u/sec) optimized for fast GC gradients and complex mixture analysis (e.g., essential oils, biofuels, polymer extracts)
- Integrated LabSolutions GCMS software with MS Navigator—a guided diagnostics and maintenance assistant providing step-by-step visual protocols for source cleaning, column conditioning, and calibration verification
- Dual-mode ionization (EI/CI) support via interchangeable ion source components; CI mode enables enhanced molecular weight confirmation for thermally labile or low-ionization-efficiency compounds
- Modular sample introduction compatibility: capillary split/splitless injection, direct insertion probe (DIP) for non-volatile solids/liquids, SPME, headspace, purge-and-trap, and pyrolysis (PY-GCMS-ready with optional accessories)
- Factory-calibrated mass axis stability (<±0.1 u over 48 h) and detector linearity (10⁴ dynamic range) validated per ASTM D5845 and ISO 17025 internal reference protocols
Sample Compatibility & Compliance
The GCMS-QP2010S accommodates a broad spectrum of sample types—from volatile organics in water (EPA Method 524/8260-compliant when paired with purge-and-trap) to semi-volatile residues in food matrices (EU SANTE/11312/2021 screening workflows) and polymer additives analyzed via thermal desorption or pyrolysis. Its hardware and software architecture supports GLP/GMP-aligned operation: LabSolutions GCMS includes full 21 CFR Part 11 compliance features—including electronic signatures, role-based access control, immutable audit trails, and secure data archiving. All refurbished units undergo comprehensive functional validation against Shimadzu’s original factory specifications, including retention time reproducibility (RSD <0.05% n=10), sensitivity (1 pg OFN S/N ≥100:1), and mass accuracy (≤0.2 u at m/z 502). Calibration certificates and IQ/OQ documentation packages are available upon request.
Software & Data Management
LabSolutions GCMS vX.X serves as the unified control, acquisition, and processing environment. It provides method templates for USP , ASTM D3699 (gasoline oxygenates), and EPA 8270D workflows, along with customizable report generators supporting PDF/A-1b archival compliance. Raw data files (.qgd) are stored in vendor-neutral formats compatible with third-party spectral libraries (NIST, Wiley, MassBank) and open-source processing tools (e.g., MZmine, OpenMS). Audit trail logs record all user actions—including method edits, peak integration changes, and reprocessing events—with timestamps, operator IDs, and IP addresses. Data backup utilities support scheduled export to network-attached storage (NAS) or LTO tape systems meeting ISO/IEC 27001 information security standards.
Applications
- Environmental monitoring: Quantification of PAHs, PCBs, chlorinated pesticides, and VOCs in soil, water, and air samples
- Pharmaceutical impurity profiling: Identification and structural elucidation of genotoxic impurities per ICH M7 guidelines
- Food authenticity and adulteration detection: Fatty acid methyl ester (FAME) profiling, flavor compound fingerprinting, and pesticide multiresidue screening
- Polymer characterization: Additive identification (antioxidants, plasticizers) via PY-GCMS and thermal desorption–GC-MS
- Clinical toxicology: Rapid screening of drugs of abuse and metabolites in urine and blood using automated library search and retention index matching
FAQ
Is this unit fully refurbished and performance-verified?
Yes. Each GCMS-QP2010S undergoes full electrical, vacuum, and spectral validation at our ISO 17025-accredited refurbishment facility. Full test reports—including sensitivity, resolution, mass accuracy, and retention time stability—are provided with delivery.
Does the system include original Shimadzu documentation and software licenses?
All units ship with original LabSolutions GCMS installation media, electronic license keys, and digitized operation manuals. Extended software maintenance contracts are available.
Can the system be configured for regulatory-compliant environments (e.g., FDA-regulated labs)?
Yes. LabSolutions GCMS supports 21 CFR Part 11 compliance out-of-the-box. We provide IQ/OQ documentation templates and can assist with site-specific validation protocols.
What consumables and spare parts are included?
Standard shipment includes one set of GC inlet liners, septa, gold-plated ferrules, a new ion source gasket kit, and a 6-month supply of high-purity helium carrier gas filters. Optional extended spares kits are available.
Is technical support available post-purchase?
Yes. Our team of Shimadzu-certified application scientists provides remote troubleshooting, method optimization, and on-site service contracts globally.