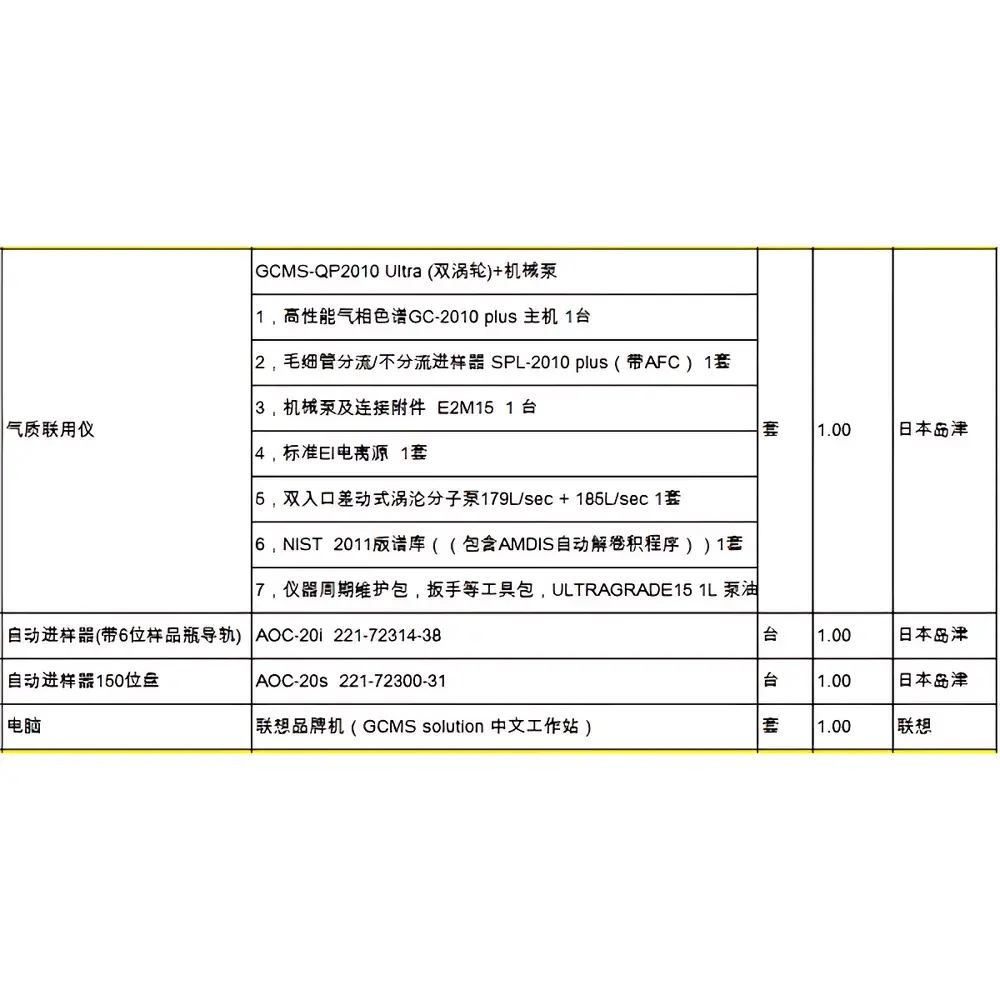
Shimadzu GCMS-QP2010SE Used Gas Chromatography-Mass Spectrometry System
| Brand | Shimadzu |
|---|---|
| Origin | Japan |
| Model | GCMS-QP2010SE |
| Configuration | GC-2010 Plus GC + QP2010SE MS + Auto-sampler (Headspace or Liquid, selectable) + Vacuum Pump + LabSolutions Workstation PC + Essential Tubing/Cabling |
| Warranty | 90-day comprehensive hardware warranty |
| Condition | Fully tested and verified operational status |
Overview
The Shimadzu GCMS-QP2010SE is a benchtop single-quadrupole gas chromatography-mass spectrometry (GC-MS) system engineered for routine qualitative and quantitative analysis in environmental, food safety, pharmaceutical, and forensic laboratories. It integrates the GC-2010 Plus capillary gas chromatograph with the QP2010SE mass spectrometer using electron ionization (EI) and optional chemical ionization (CI) sources. The system operates on the principle of compound separation by retention time in the GC column followed by mass-selective detection via quadrupole mass filtering and electron multiplier detection. Its architecture supports robust method transfer from legacy Shimadzu platforms—including GCMS-QP5000 series—enabling continuity in regulated environments without revalidation overhead. Designed for operational sustainability, the QP2010SE incorporates Eco Mode, an energy- and carrier-gas–optimized standby protocol that reduces power consumption by up to 40% during unattended overnight runs while maintaining system readiness for next-sequence initiation.
Key Features
- Maximum column flow capacity of 4 mL/min helium or hydrogen, enabling broader selection of analytical columns—including wide-bore, high-flow, and low-bleed phases—without compromising sensitivity or resolution.
- Backward-compatible method migration: GCMS-QP5000-derived acquisition and quantitation methods can be imported directly into LabSolutions software with minimal parameter adjustment.
- Direct inlet capability: Supports manual or automated direct sample introduction without requiring physical relocation of the GC unit—ideal for rapid screening or volatile compound analysis.
- Eco Mode automation: Activates post-batch completion to suspend non-essential subsystems (ion source heater, detector high voltage, GC oven cooling fans, PC display) while preserving vacuum integrity and column temperature stability.
- Integrated LabSolutions GC/MS workstation: Provides unified control of GC parameters, MS acquisition, spectral library search (NIST/Wiley), peak integration, calibration curve generation, and report export compliant with laboratory documentation standards.
Sample Compatibility & Compliance
The GCMS-QP2010SE accommodates liquid, headspace, and solid-phase microextraction (SPME) samples via compatible autosamplers. It meets essential performance criteria outlined in ASTM D5845 (determination of aromatic hydrocarbons in gasoline), EPA Method 8270D (semivolatile organic compounds), and ISO 17025–accredited testing workflows. While not pre-certified for 21 CFR Part 11, the LabSolutions platform supports audit-trail-enabled user authentication, electronic signature logging, and data integrity safeguards required for GLP/GMP-aligned operations when configured with appropriate IT controls and procedural SOPs.
Software & Data Management
LabSolutions GC/MS v5.x (or later) serves as the primary acquisition and processing interface. It features real-time total ion chromatogram (TIC) monitoring, selected ion monitoring (SIM) setup with dwell time optimization, automated tune verification (autotune), and customizable report templates. Raw data (.qgd files) are stored in a structured folder hierarchy with embedded metadata (instrument ID, operator, date/time, method name). Backup and archiving are supported via network drive mapping or external NAS; all processed results—including calibration curves, peak tables, and spectral matches—are exportable in CSV, PDF, or XML formats for LIMS integration.
Applications
This system is routinely deployed for residual solvent analysis in pharmaceutical intermediates (ICH Q3C), pesticide residue screening in agricultural commodities (EU MRLs), polycyclic aromatic hydrocarbon (PAH) profiling in soil extracts, fragrance component identification in cosmetics, and volatile organic compound (VOC) emission testing per ISO 16000-6. Its balance of sensitivity (sub-pg on-column detection for chlorinated benzenes), reproducibility (RSD < 5% for replicate injections), and ease of maintenance makes it suitable for mid-throughput QC labs operating under ISO/IEC 17025 or internal quality management systems.
FAQ
Is this instrument supplied with original Shimadzu documentation and service history?
Yes—each unit includes scanned copies of the original installation certificate, factory acceptance test (FAT) report, and last-known preventive maintenance log. Full documentation is provided upon purchase.
Can the system be validated for GxP use?
The hardware and LabSolutions software support IQ/OQ/PQ execution per vendor-recommended protocols. We provide validation support packages including URS templates, test scripts, and traceability matrices.
What consumables and spare parts are included?
Standard delivery includes one set of GC inlet septa, liner, gold seal, and column nuts; one EI filament; and a 90-day supply of pump oil. Additional consumables are available through our global distribution network.
Is remote diagnostics and technical support available post-installation?
Yes—SpectraLab Scientific offers tiered support plans including remote access-assisted troubleshooting, firmware updates, and priority response SLAs for critical instrument downtime events.