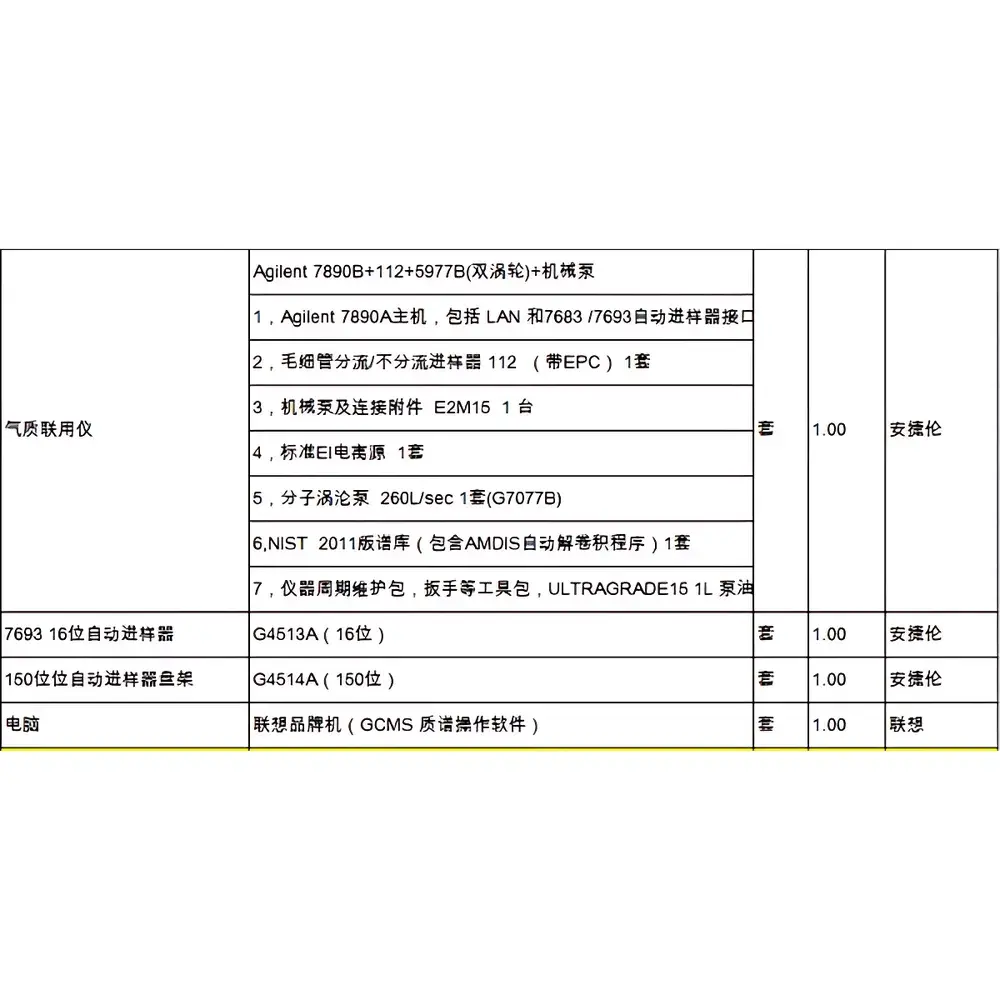
Thermo Fisher Scientific Corona Ultra CAD Charged Aerosol Detector
| Brand | Thermo Fisher Scientific |
|---|---|
| Origin | USA |
| Model | Corona Ultra CAD |
| Detection Principle | Charged Aerosol Detection (CAD) |
| Power Supply | 100–240 V AC, 100 VA |
| Serial Number | DU-0136 |
| Age | 3–5 years |
| Warranty | 6 months |
| Compliance | Designed for GLP/GMP-compliant laboratories |
| Software Compatibility | Compatible with Chromeleon CDS (v7.2+), Empower (v3+), and other industry-standard chromatography data systems |
| Regulatory Alignment | Supports 21 CFR Part 11 audit trail requirements when paired with validated CDS configurations |
| Sample Compatibility | Non-volatile and semi-volatile analytes across small molecules to macromolecules |
Overview
The Thermo Fisher Scientific Corona Ultra CAD Charged Aerosol Detector is a high-performance, universal mass-sensitive detector engineered for liquid chromatography (LC) systems. Unlike optical or ionization-based detectors, the Corona Ultra CAD operates on the principle of charged aerosol detection: analyte-containing eluent is nebulized into fine droplets, dried to form non-volatile particles, and then uniformly charged in a controlled corona discharge field. The resulting charge—proportional to analyte mass—is measured via a highly sensitive electrometer. This physical detection mechanism delivers near-uniform response factors across diverse compound classes, eliminating the need for compound-specific calibration curves. As a result, the Corona Ultra CAD serves as a robust, quantitative alternative to refractive index (RI), low-wavelength UV, and evaporative light scattering detection (ELSD), particularly where analytes lack chromophores or ionizable groups.
Key Features
- Universal detection capability for non-volatile and semi-volatile compounds without structural bias
- Wide dynamic range (>10⁴) with high reproducibility (RSD < 3% for repeated injections)
- Enhanced sensitivity compared to ELSD—typically 5–10× lower detection limits for many pharmaceutical actives
- Stable baseline performance under gradient elution conditions due to optimized drying gas flow control and real-time background compensation
- Modular design supporting integration with UHPLC, HPLC, and preparative LC systems
- Self-diagnostic firmware with event logging and operational parameter monitoring for preventive maintenance planning
- Compliance-ready architecture: supports electronic signatures, audit trails, and user access controls when deployed with validated chromatography data systems (e.g., Chromeleon CDS or Waters Empower)
Sample Compatibility & Compliance
The Corona Ultra CAD is routinely applied to analytes spanning molecular weights from ~200 Da to >200 kDa—including APIs, excipients, carbohydrates, lipids, steroids, peptides, proteins, and synthetic polymers. It is widely adopted in pharmaceutical QC/QA laboratories for stability-indicating assays, impurity profiling, and formulation analysis per ICH Q2(R2) and USP . The detector meets essential hardware prerequisites for GLP and GMP environments: traceable calibration records, secure configuration management, and deterministic signal processing. While the instrument itself does not carry FDA clearance, its operational parameters and output behavior are fully documentable for regulatory submissions and internal validation protocols (e.g., IQ/OQ/PQ).
Software & Data Management
The Corona Ultra CAD interfaces natively with Thermo’s Chromeleon Chromatography Data System (CDS) v7.2 or later, enabling full method control, real-time signal visualization, and automated calibration routines. It also supports analog/digital output modes compatible with third-party CDS platforms including Waters Empower, Agilent OpenLab CDS, and Shimadzu LabSolutions. All raw detector signals are timestamped and stored in vendor-neutral formats (e.g., .cdf, .axd). When integrated into a validated CDS environment, the system supports 21 CFR Part 11 compliance features—including electronic signatures, role-based access control, and immutable audit trails for all acquisition and processing events.
Applications
- Quantitative analysis of non-UV-absorbing excipients (e.g., polysorbates, PEGs, cyclodextrins) in biopharmaceutical formulations
- Stability-indicating assays for degradation products lacking chromophores
- Carbohydrate profiling in nutraceuticals and biologics (e.g., glycan mapping of monoclonal antibodies)
- Lipidomics workflows requiring consistent response across phospholipids, triglycerides, and sphingolipids
- Polymers characterization—including molecular weight distribution estimation via SEC-CAD coupling
- Method development support for orthogonal detection strategies in regulatory filings
FAQ
Is the Corona Ultra CAD compatible with UHPLC systems?
Yes—it supports flow rates from 0.1 to 3.0 mL/min and operates with backpressures up to 1000 psi, making it suitable for both conventional HPLC and UHPLC applications.
Does this unit include original manufacturer documentation?
All units shipped by SpectraLab Scientific include scanned copies of the original Thermo Fisher installation qualification (IQ) checklist, operation manual, and factory test report.
Can the detector be validated for GMP use?
Yes—the hardware design supports full IQ/OQ/PQ execution; SpectraLab provides optional validation support packages including protocol templates and on-site assistance.
What maintenance is required between calibrations?
Routine maintenance includes weekly nebulizer cleaning, quarterly capillary inspection, and annual corona needle replacement—full procedures are documented in the operator manual.
Is firmware upgrade support available post-purchase?
Firmware updates are distributed freely via Thermo Fisher’s official support portal; SpectraLab provides technical guidance for safe installation and version verification.