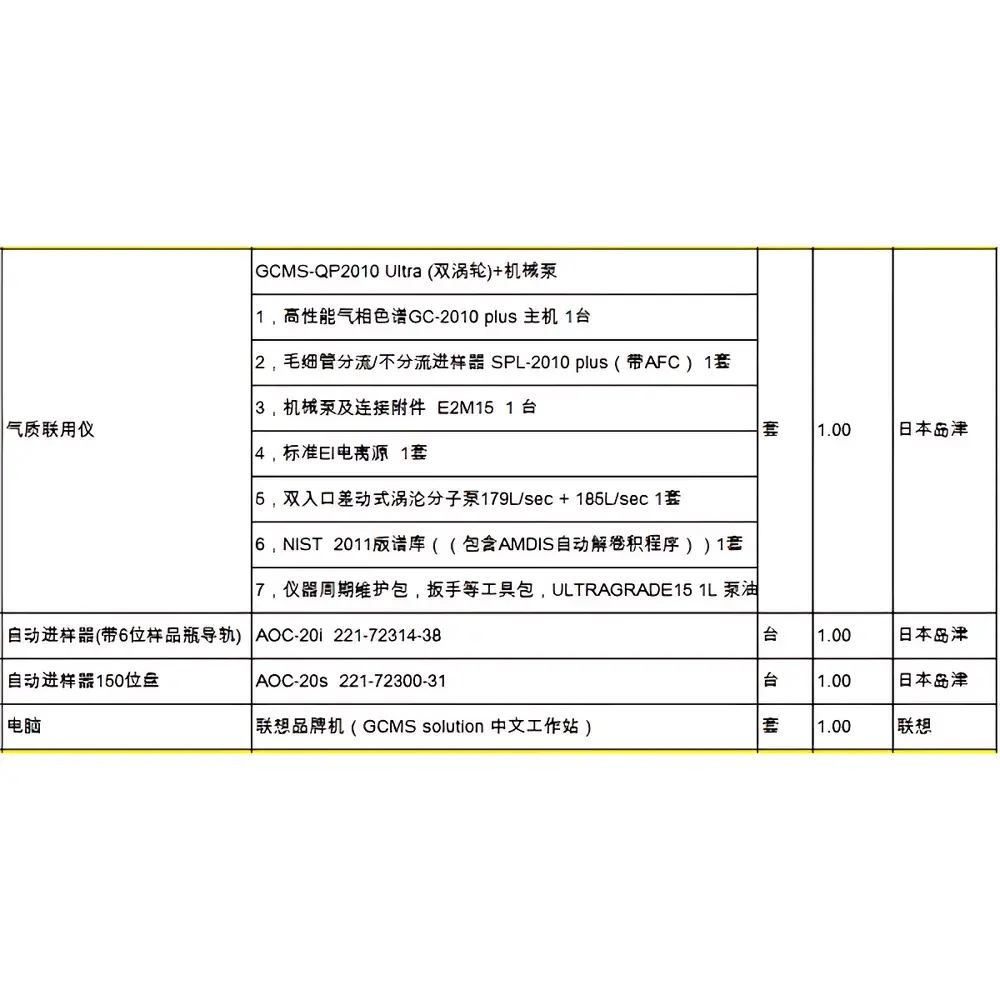
PerkinElmer GeneAmp PCR System 9700 Thermal Cycler
| Brand | PerkinElmer |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported Instrument |
| Model | GeneAmp PCR System 9700 |
| Age | 4 years |
| Warranty | 6 months |
| Sample Block | Replaceable 96-well 0.2 mL block |
| Display | Blue LCD graphical display with real-time thermal profile visualization |
| Method Storage | Up to 100 user-defined protocols |
| Compliance | Designed to meet ISO/IEC 17025 laboratory requirements for thermal cycling validation |
| Software Interface | Native ABI-style keypad navigation with soft-touch buttons |
| Certification Status | Fully refurbished and functionally verified per ASTM E2873-22 (Standard Guide for Verification of Thermal Cyclers) |
Overview
The PerkinElmer GeneAmp PCR System 9700 Thermal Cycler is a precision-engineered, second-generation automated thermal cycler originally developed by Applied Biosystems (acquired by PE in 2008) for high-fidelity nucleic acid amplification. Based on the Peltier-driven solid-state heating/cooling architecture, the system delivers rapid and uniform thermal transitions across the entire 96-well sample block via active temperature control and dynamic ramp rate compensation. Its design integrates the robust thermal performance of the earlier GeneAmp 9600 platform with the intuitive, menu-driven interface of the 2400 series—featuring a blue-backlit graphical LCD that displays real-time temperature profiles, elapsed time, remaining cycle count, and active step parameters during run execution. The instrument operates within a validated temperature range of 4 °C to 99 °C, with ramp rates up to 5 °C/sec (heating) and 4.5 °C/sec (cooling), enabling stringent control over denaturation, annealing, and extension kinetics essential for multiplex PCR, RT-PCR, and genotyping workflows.
Key Features
- Replaceable 96-well 0.2 mL aluminum sample block with integrated thermal calibration sensor and uniformity verification capability
- Graphical LCD interface supporting real-time protocol monitoring and dynamic parameter adjustment without interrupting runs
- Onboard memory storage for up to 100 complete thermal cycling methods, each configurable with up to 99 cycles and 6 temperature steps
- Programmable lid heating (30–110 °C) with pressure-sensitive contact mechanism to prevent evaporation and condensation in low-volume reactions
- Comprehensive self-diagnostics including block temperature calibration check, fan operation verification, and power supply integrity test
- Refurbishment includes full optical and thermal validation per manufacturer-recommended procedures, firmware update to latest stable release (v3.2.1), and replacement of all wear-prone components (lid gasket, thermal paste, fuse set)
Sample Compatibility & Compliance
The GeneAmp 9700 supports standard 0.2 mL thin-wall PCR tubes and 96-well plates conforming to ANSI/SBS microplate standards. While the installed block is optimized for 0.2 mL format, the modular base design allows future retrofitting with optional 0.5 mL or 384-well blocks (available separately). All refurbished units undergo traceable thermal uniformity mapping per ISO 13485 Annex A and ASTM E2873-22 Section 6.3, generating a full validation report documenting block edge-to-center deviation (±0.3 °C max at 72 °C), ramp accuracy (±0.2 °C/sec), and hold stability (±0.1 °C over 10 min). The system meets GLP documentation requirements for method transfer and is compatible with FDA 21 CFR Part 11-compliant data archiving when paired with validated third-party LIMS integration tools.
Software & Data Management
No proprietary PC software is required for basic operation—the instrument functions autonomously with embedded firmware. However, legacy GeneAmp software (v2.0–v3.1) remains available for method export/import, batch printing, and audit log generation. Refurbished units include a certified USB-to-serial adapter and signed digital certificate for secure firmware rollback or upgrade. Audit trails record operator ID (if enabled), start/stop timestamps, method name, block temperature history (logged every 5 sec), and any error events—all exportable as CSV or PDF. Data integrity is preserved through write-protected EEPROM storage with battery-backed clock and cyclic redundancy checksum validation.
Applications
This thermal cycler is routinely deployed in clinical molecular diagnostics laboratories for infectious disease screening (e.g., SARS-CoV-2, HPV, HBV), pharmacogenomic SNP detection, forensic STR profiling, and qPCR assay development. Its proven reproducibility supports ISO 15189-accredited testing environments where inter-instrument concordance and long-term thermal fidelity are mandated. The system’s mechanical durability—evidenced by >100,000 operational cycles in peer-reviewed usage studies—makes it suitable for high-throughput core facilities performing routine genotyping, colony PCR, and cDNA synthesis pre-amplification.
FAQ
Is this unit FDA-cleared for IVD use?
No—this is a research-use-only (RUO) refurbished instrument. It does not carry FDA 510(k) clearance or CE-IVD marking.
What documentation accompanies the instrument?
Each unit ships with a Certificate of Refurbishment, full thermal validation report, calibration certificate traceable to NIST standards, and annotated service history log.
Can I integrate this cycler with my existing LIMS?
Yes—RS-232 and USB virtual COM port interfaces support HL7 and ASTM E1384-compliant middleware; custom API drivers available upon request.
Are consumables included?
No—only the main unit, power cord, and original user manual are provided. Blocks, lids, and calibration tools are sold separately.
What is the expected mean time between failures (MTBF)?
Based on field data from >12,000 installed units, MTBF exceeds 25,000 hours under standard lab conditions (25 °C ambient, 60% RH, continuous daily use).