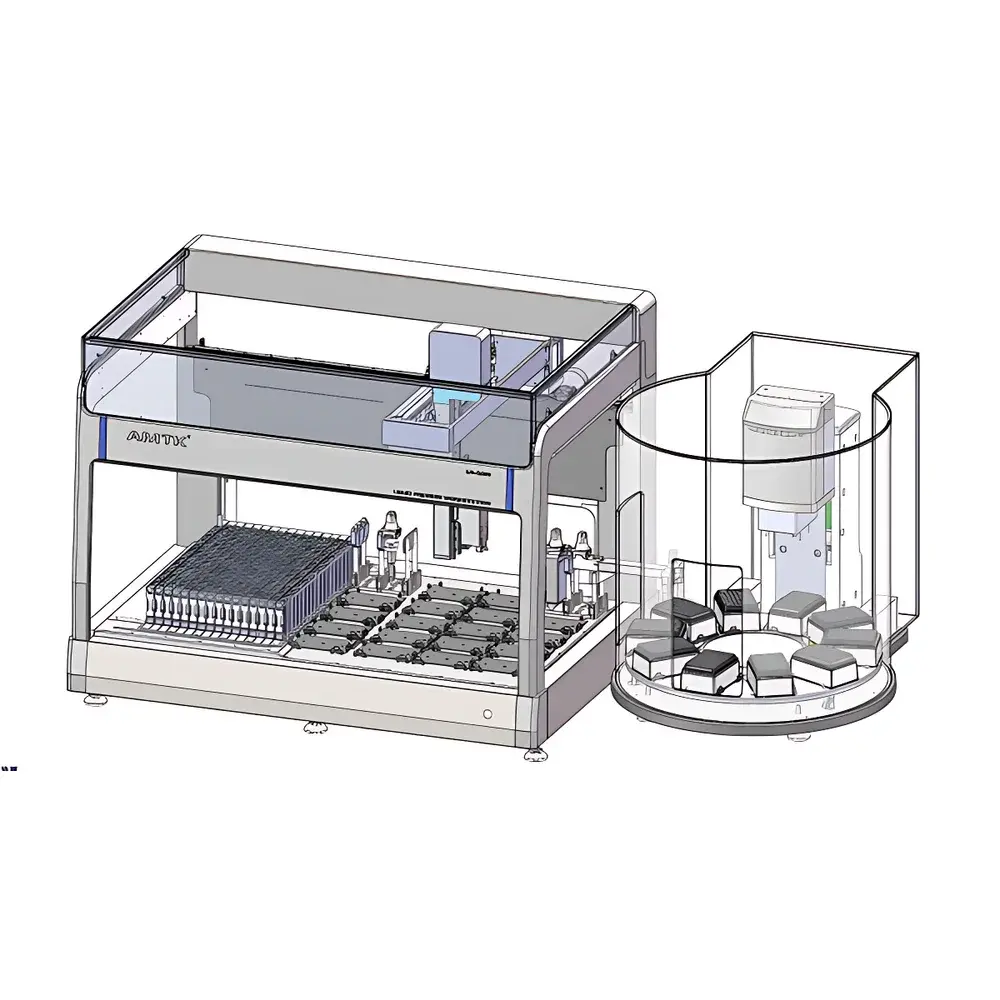
AMTK LH1808 Fully Automated Liquid Handling Workstation
| Brand | AMTK |
|---|---|
| Origin | Beijing, China |
| Model | LH1808 |
| Deck Capacity | 30 + 10 (expandable) |
| Pipetting Accuracy | CV < 5% at 2 µL |
| Pipetting Range | 0.5–1000 µL |
| Sample Throughput | 384 samples |
| Dimensions (Main Deck) | 110 × 80 × 90 cm (L×D×H) |
| Dimensions (Rotary Carousel) | 60 × 80 × 70 cm (L×D×H) |
| Power Supply | AC 220 V, 50 Hz, 450 W |
| Weight | 150 kg |
| Compatible Tips | 10 µL, 30 µL, 50 µL, 250 µL, 1 mL |
| SBS-Compliant Vessels | 24-, 96-, and 384-well plates, centrifuge tubes, collection tubes |
| Barcode Scan Time per Tube | < 2.3 s |
| Liquid Level Detection Error | ±0.5 mm |
| Pipetting Accuracy & Precision | 250 µL tip: ±8.0% accuracy / ≤4.0% CV at 10 µL |
| 1000 µL tip | ±2.0% / ≤1.0% at 100 µL |
Overview
The AMTK LH1808 Fully Automated Liquid Handling Workstation is an integrated benchtop platform engineered for high-precision, high-throughput liquid manipulation in molecular diagnostics, NGS library preparation, and clinical qPCR workflows. It operates on a dual-motion architecture—combining a large fixed-deck Cartesian coordinate system with an independent rotary carousel module—to enable concurrent execution of multiple processing steps without manual intervention. The system employs capacitive liquid level detection (LLD) with real-time Z-axis compensation, delivering superior sensitivity and reproducibility compared to traditional conductive or optical LLD methods. All pipetting operations are performed using air-displacement technology with temperature-stabilized tip racks and pressure-sensing feedback to verify aspiration status and prevent dry-tip errors. Designed for compliance-critical environments, the LH1808 supports audit-ready operation through timestamped event logging, user-access controls, and full traceability from raw sample tube to final assay plate.
Key Features
- Dual-deck architecture: Main deck (30+10 configurable positions) plus dedicated rotary carousel for parallel reagent staging, plate rotation, and thermal conditioning.
- Integrated barcode scanning module: Reads 1D/2D tube barcodes in <2.3 seconds with auto-registration into local LIMS-compatible metadata fields.
- Intelligent liquid level detection: Capacitive sensing with ±0.5 mm vertical resolution and dynamic Z-height adjustment during aspiration and dispense cycles.
- Modular functional integration: Supports optional modules including magnetic bead separation units, Peltier-based heating/cooling blocks (4–95 °C), orbital shakers (200–1500 rpm), UV-C decontamination, and robotic plate grippers.
- Multi-tip flexibility: Swappable single-channel, 4-channel, and 8-channel pipetting heads compatible with standard SBS-format tips (10–1000 µL), enabling seamless transition between low-volume precision (<1 µL CV-controlled dispensing) and high-volume transfer (up to 5 mL via extended-range syringe pumps).
- Autonomous tip exchange: On-deck tip rack management with collision-aware path planning and tip ejection verification.
Sample Compatibility & Compliance
The LH1808 accommodates a broad range of primary and secondary sample containers compliant with ANSI/SBS standards—including 24-, 96-, and 384-well microplates, 0.2–15 mL conical tubes, EDTA/K2EDTA blood collection tubes, and custom OEM consumables. Its open-deck design allows third-party module integration while maintaining mechanical stability under continuous operation. From a regulatory standpoint, the workstation meets essential requirements for ISO 13485-aligned IVD manufacturing environments and supports GLP/GMP data integrity practices through secure user authentication, electronic signatures, and immutable activity logs. While not pre-certified for FDA 21 CFR Part 11, its software architecture includes built-in features necessary for validation: audit trails with operator ID, action timestamps, and change history for all protocol edits and run executions.
Software & Data Management
Controlled via AMTK’s proprietary LH-Studio software suite, the LH1808 provides a graphical workflow builder with drag-and-drop protocol design, real-time simulation, and error-prevention logic (e.g., volume conflict checks, cross-contamination avoidance). Protocols can be exported as XML or JSON for version control and shared across instrument fleets. Raw run data—including pipette pressure curves, motor encoder positions, and barcode scan results—are stored in structured SQLite databases with optional export to CSV or direct API push to LIMS or ELN systems. Software updates are delivered over HTTPS with SHA-256 signature verification, and configuration backups include cryptographic hash validation to ensure integrity.
Applications
- NGS sample prep: Direct tube-to-plate plasma aliquoting, automated nucleic acid extraction (including magnetic bead-based purification), library fragmentation, adapter ligation, and index PCR setup.
- qPCR assay assembly: High-fidelity serial dilution series generation, master mix dispensing, template addition across 384-well formats, and pooling of indexed libraries prior to sequencing.
- Clinical screening workflows: End-to-end automation of blood-borne pathogen testing (e.g., HBV/HCV/HPV), including primary tube decapping, nucleic acid isolation, and RT-qPCR reaction assembly—all traceable to original specimen IDs.
- Tumor profiling pipelines: FFPE tissue lysate processing, DNA quantification, targeted amplicon panel setup, and QC plate generation for fragment analysis.
- Biobanking operations: Long-term sample inventory management with auto-generated storage maps, viability testing schedules, and re-aliquoting triggers based on freeze-thaw cycle counts.
FAQ
Does the LH1808 support integration with third-party LIMS or ELN systems?
Yes—via RESTful API endpoints and configurable CSV/JSON export templates. Authentication uses OAuth 2.0 or basic HTTP auth with role-based access control.
What is the maximum number of simultaneous protocols that can be queued?
Up to 16 protocols may be scheduled in the job queue, with priority-based execution and dependency chaining (e.g., “run Protocol B only after Protocol A completes successfully”).
Is remote monitoring and troubleshooting supported?
The system includes embedded VNC server functionality with TLS-encrypted session initiation, allowing secure remote desktop access for service engineers and lab managers.
How is calibration verified and documented?
Gravimetric calibration is performed using NIST-traceable balances and certified reference liquids. Calibration reports include uncertainty budgets per ISO/IEC 17025 and are archived with digital signatures.
Can the LH1808 operate unattended overnight?
Yes—equipped with internal UPS buffering (optional), door-open safety interlocks, thermal runaway protection, and automatic shutdown upon critical error detection or scheduled time window expiration.