COMECAUSE IN-J200 Automated Colony Counter & Image-Based Microbial Analyzer
| Brand | COMECAUSE |
|---|---|
| Origin | Shandong, China |
| Manufacturer Type | Direct Manufacturer |
| Instrument Type | Fully Automated Colony Counter |
| Sample Format | Petri Dish |
| Compatible Dish Diameter | 90–110 mm |
| Counting Accuracy | ≤ ±10% |
| Imaging Resolution | 1920 × 1080 (16 MP Color CMOS Sensor) |
| Key Features | Fluorescence Excitation (366 nm), UV Sterilization (254 nm), Dual-Independent Adjustable Illumination (Top/Bottom), Touchscreen Interface (15.6″, Full HD), Onboard Windows 10 OS (64-bit, 256 GB SSD), Multi-Mode Segmentation Algorithms, Real-Time Threshold Adjustment, ROI Annotation Tools, Size-Based Classification (25 Bins), PDF Export with Immutable Raw Data Archive, Dual-Language UI (EN/CN), Multi-User Account System with Audit-Trail Capable Data Isolation |
Overview
The COMECAUSE IN-J200 Automated Colony Counter is a dedicated image-based microbial enumeration system engineered for precision, reproducibility, and regulatory-compliant operation in quality control and research microbiology laboratories. It employs high-resolution digital imaging coupled with adaptive computer vision algorithms to quantify viable microorganisms grown on solid media—primarily standard Petri dishes (90–110 mm diameter). Unlike manual or semi-automated alternatives, the IN-J200 integrates optical acquisition, real-time segmentation, and statistical classification within a single, self-contained platform. Its measurement principle relies on contrast-based and fluorescence-enhanced pixel intensity analysis under controlled illumination conditions, enabling reliable detection of colonies as small as 0.1 mm in diameter. The instrument operates without external computing hardware, featuring an embedded 15.6-inch capacitive touchscreen, onboard Windows 10 64-bit OS, and local SSD storage—ensuring data sovereignty and minimizing IT infrastructure dependencies.
Key Features
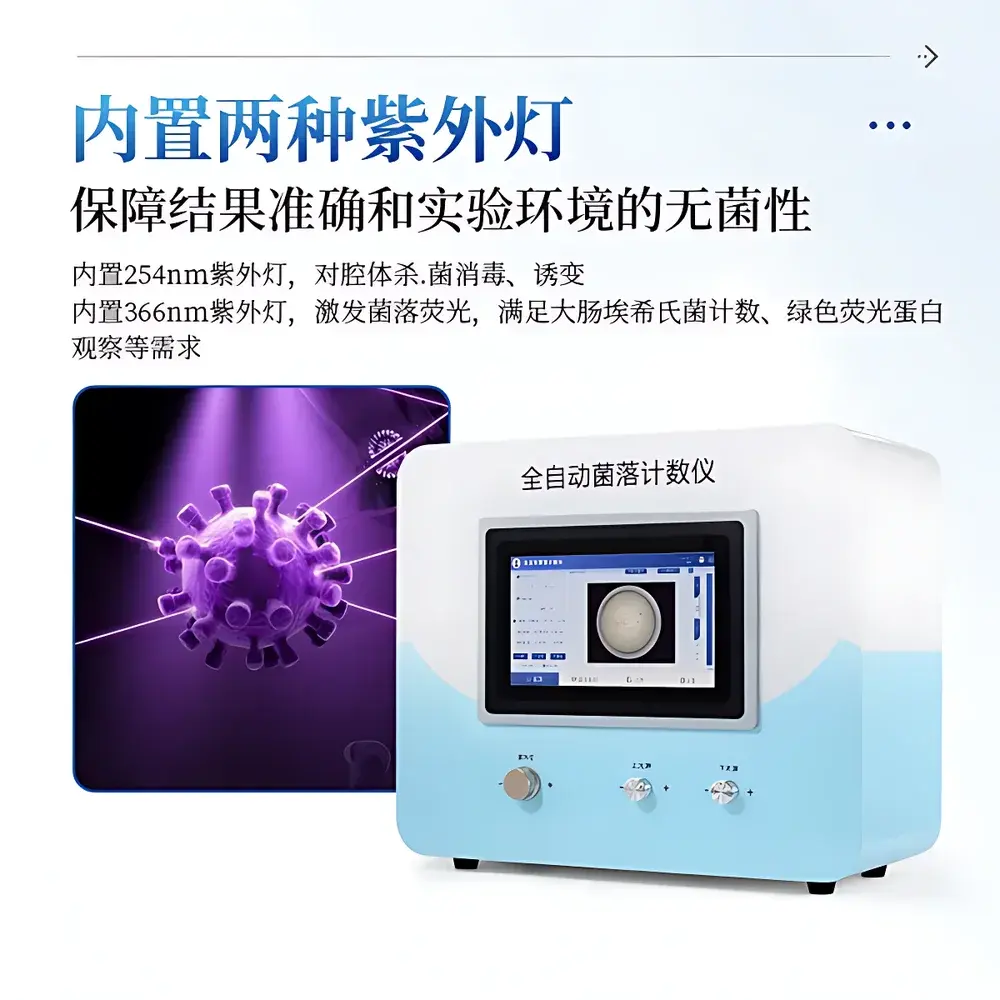
- Sealed dark chamber design eliminates ambient light interference and ensures consistent imaging conditions.
- Dual-wavelength UV system: 254 nm lamp for internal cavity sterilization and mutagenesis control; 366 nm lamp for fluorescence excitation—validated for Escherichia coli enumeration using GFP-expressing strains and chromogenic substrates.
- 16-megapixel color CMOS sensor with fixed-focus macro optics optimized for Petri dish flat-field imaging at working distances compliant with ISO 4833-2:2013 and AOAC Official Method 2012.02.
- Independent top and bottom illumination channels, each with continuous brightness adjustment via physical rotary dials—enabling optimization for translucent, opaque, or pigmented media (e.g., VRBA, PCA, ChromAgar).
- Six specialized counting modes: Dispersed Colonies, Aggregated Colonies, Microcolonies (0.1–0.5 mm), 3M Petrifilm™, Compact Dry™ (black-grid), RIDA™ membrane filters—each applying proprietary thresholding and watershed segmentation logic calibrated per substrate reflectivity and spectral response.
- Real-time correction suite: interactive ROI selection (circle, rectangle, freehand brush), dynamic threshold tuning, and post-count size binning across 25 discrete diameter intervals (0.1–5.0 mm).
Sample Compatibility & Compliance
The IN-J200 accommodates standard polystyrene and polypropylene Petri dishes (90 mm, 100 mm, and 110 mm diameters), with centering guides ensuring repeatable positioning. It supports agar-based, chromogenic, and selective media—including those used in pharmaceutical sterility testing (USP ), food safety validation (ISO 6579-1, ISO 4833-1), and environmental monitoring (ISO 11731). While not certified to IEC 61000-4 or FDA 21 CFR Part 11 out-of-the-box, its multi-user authentication, immutable PDF reporting, and segregated data storage architecture align with GLP and GMP documentation requirements. Audit trail functionality (user login timestamps, parameter change logs, and export history) can be enabled via firmware update to meet internal QA protocols.
Software & Data Management
The embedded software provides a validated workflow from acquisition to report generation. All image files (RAW TIFF + processed JPEG) and metadata (timestamp, operator ID, dish ID, illumination settings, algorithm parameters) are stored locally with SHA-256 hash integrity verification. PDF reports include embedded thumbnails, colony distribution histograms, size-frequency tables, and configurable headers (lab name, SOP number, analyst signature field). Data export supports CSV for LIMS integration. The bilingual interface (English/Chinese) allows seamless transition in multinational R&D environments. No cloud upload or telemetry is active by default—ensuring full compliance with GDPR and HIPAA data residency policies when deployed on-premises.
Applications
- Pharmaceutical QC labs performing aerobic plate counts per USP and EP 2.6.12.
- FDA-regulated food processors conducting Listeria monocytogenes, Salmonella, and total viable count assays on chromogenic membranes.
- Cosmetic safety testing per ISO 17516 and CPSC guidelines for preservative efficacy.
- Environmental monitoring in cleanrooms (ISO 14644-1 Class 5–8) using settle plates and contact plates.
- Academic microbiology teaching labs requiring rapid turnaround for student-generated culture data.
- Biotech process development teams quantifying colony-forming units (CFU) from transformation efficiency assays and mutant screening plates.
FAQ
Does the IN-J200 comply with FDA 21 CFR Part 11?
The system supports core Part 11 elements—including electronic signatures, audit trails, and record retention—but requires site-specific validation documentation and optional firmware activation for full compliance.
Can it count colonies on non-standard media such as blood agar or MacConkey agar?
Yes—the dual illumination system and multi-mode segmentation engine accommodate high-contrast and low-contrast backgrounds through real-time brightness and threshold optimization.
Is calibration required before each use?
No. The system performs automatic geometric calibration at startup using factory-stored lens distortion profiles and dish-diameter presets. Manual recalibration is only needed after physical sensor relocation.
What file formats are supported for data export?
PDF (final report), TIFF (raw image), JPEG (processed preview), and CSV (numerical colony metrics with metadata).
How is user access controlled?
Role-based accounts (Admin, Analyst, Viewer) enforce password-protected login, session timeout, and isolated workspace directories—preventing cross-user data access.