COMECAUSE IN-CL3 Multi-Channel Airborne Particle Counter with 6-Sized Channel Detection and FDA 21 CFR Part 11 Compliance
| Brand | COMECAUSE |
|---|---|
| Origin | Shandong, China |
| Manufacturer Type | OEM Manufacturer |
| Country of Origin | China |
| Model | IN-CL3 |
| Sampling Flow Rate | 28.3 L/min (1 cfm) |
| Particle Size Channels | 0.3, 0.5, 1.0, 3.0, 5.0, 10.0 µm |
| Self-Cleaning Time | ≤10 min |
| Size Accuracy (0.5 & 5.0 µm) | ±30% |
| Concentration Accuracy (0.5 µm) | ±30% |
| Repeatability | ≤±10% |
| Operating Temperature | 15–35 °C |
| Relative Humidity Range | 10–85% RH |
| Data Storage Capacity | 10,000 records |
| Battery Life | ≥6 h (continuous sampling) |
| Power Supply | AC/DC dual-mode, 16.8 V / 15 Ah Li-ion battery |
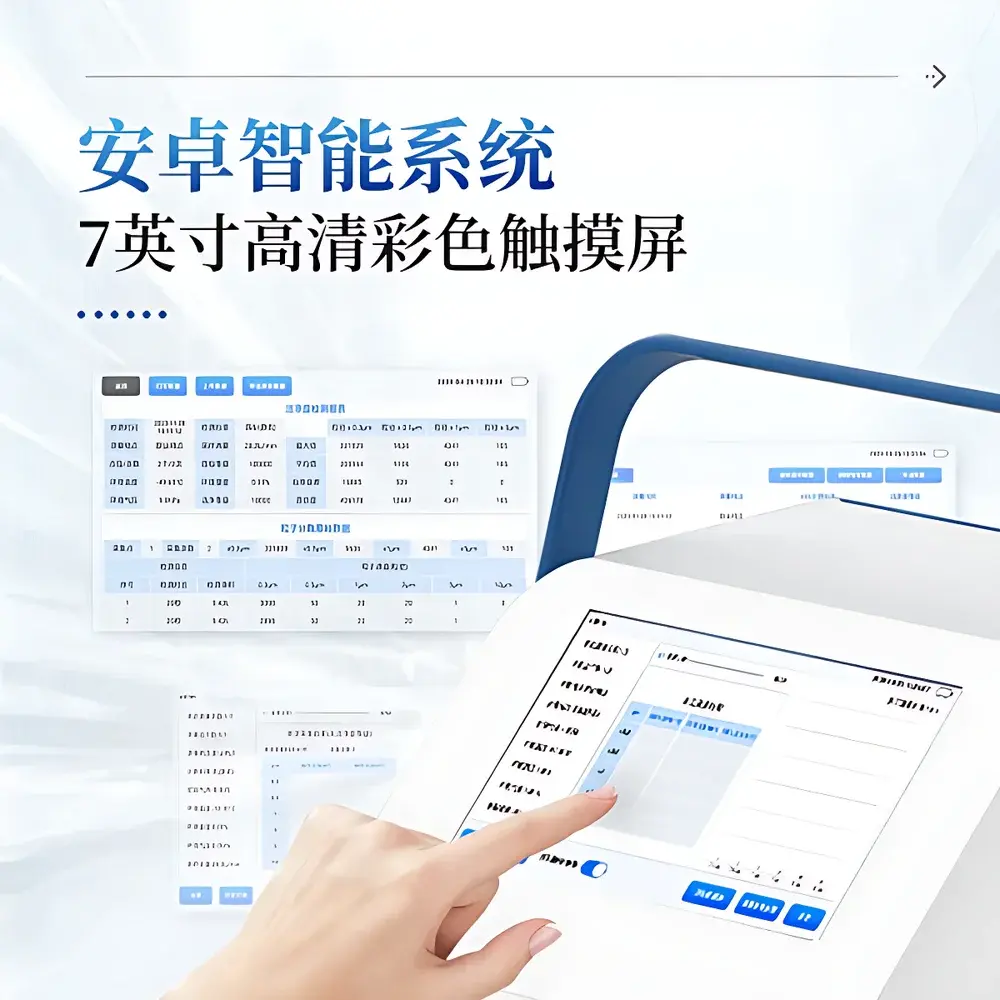
| Display | 7-inch color capacitive touchscreen |
| Connectivity | Wi-Fi, Bluetooth, USB flash drive support |
| Calibration Standard | JJF 1190–2008 |
| Performance Verification Standard | GB/T 6167–2007 |
| Cleanroom Classification Support | ISO 14644-1, FS209E, GMP (static/dynamic) |
Overview
The COMECAUSE IN-CL3 is a high-precision, multi-channel airborne particle counter engineered for quantitative real-time monitoring of suspended particulate matter in controlled environments. It operates on the principle of light scattering photometry: an imported semiconductor laser diode (rated lifetime >30,000 hours) illuminates particles drawn through a precisely defined sensing zone at a constant volumetric flow rate of 28.3 L/min (1 cfm). Scattered light intensity—proportional to particle cross-sectional area—is detected by a high-sensitivity photodiode array. Signal processing algorithms convert pulse amplitude into optical equivalent diameter, enabling simultaneous classification and enumeration across six discrete size channels (0.3, 0.5, 1.0, 3.0, 5.0, and 10.0 µm). The instrument delivers traceable, statistically validated measurements aligned with international cleanroom performance verification protocols—including ISO 14644-1, Federal Standard 209E, and current Good Manufacturing Practice (cGMP) requirements for static and dynamic conditions.
Key Features
- Android 7.1 operating system with intuitive graphical interface and responsive 7-inch capacitive touchscreen
- Integrated environmental sensors: calibrated temperature, relative humidity, and barometric pressure measurement
- Optimized optical chamber design minimizing stray light; enhanced signal-to-noise ratio for improved counting accuracy and reproducibility
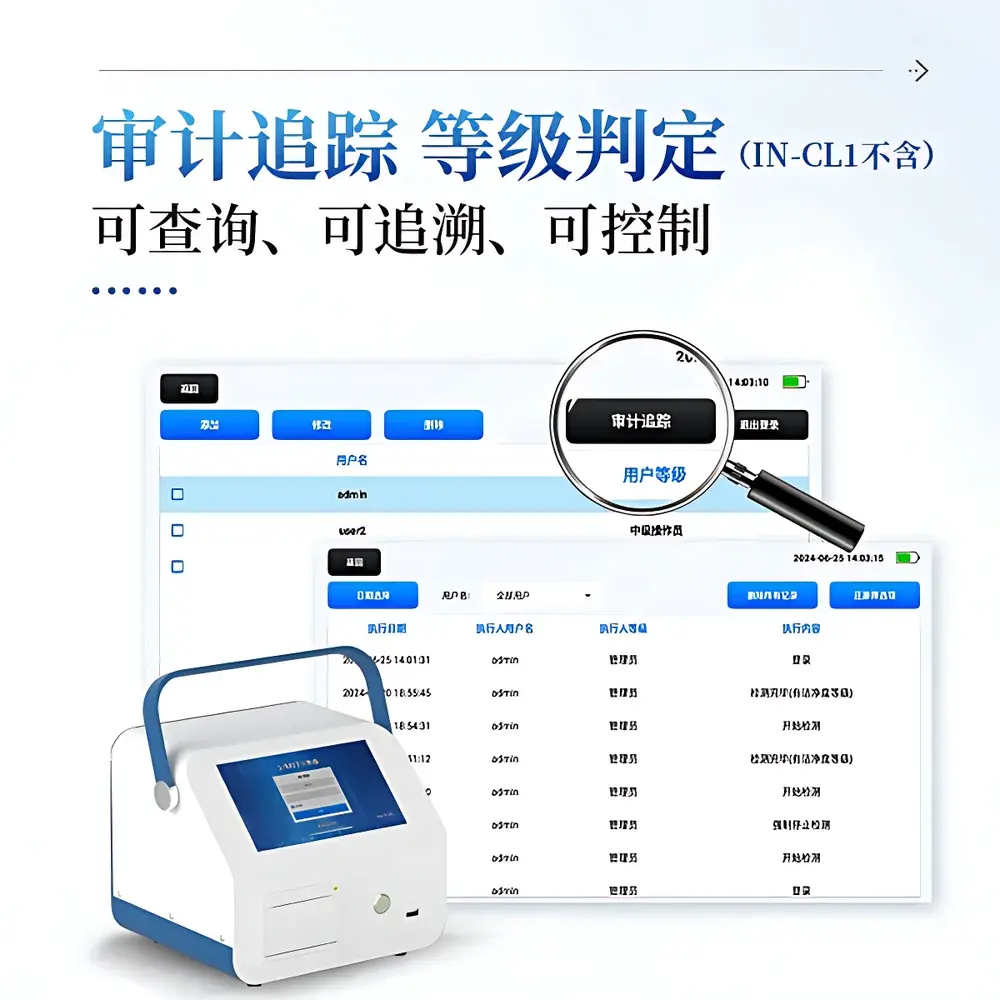
- Automated UCL (Upper Confidence Limit) calculation per ISO 14644-1 Annex B, supporting real-time cleanroom class determination (ISO Class 1–9)
- FDA 21 CFR Part 11-compliant audit trail: time-stamped, user-identified, immutable records of all configuration changes, calibration events, and data exports
- Wireless data transmission via Wi-Fi or Bluetooth; local export to USB flash drives without driver installation
- Cloud-ready architecture: seamless integration with centralized regulatory monitoring platforms for long-term trend analysis and compliance reporting
- Onboard thermal printer with customizable channel selection; supports immediate hardcopy documentation for audits and SOP adherence
- Portable form factor with telescoping tripod mount; designed for rapid deployment in ISO-certified cleanrooms, isolators, and laminar flow hoods
- Dual-power operation: AC mains or rechargeable lithium-ion battery (16.8 V / 15 Ah), delivering ≥6 hours of continuous sampling per charge
Sample Compatibility & Compliance
The IN-CL3 is validated for use in non-hazardous, non-corrosive ambient air sampling across regulated and industrial settings. Its performance meets the metrological requirements specified in JJF 1190–2008 (Calibration Specification for Airborne Particle Counters) and GB/T 6167–2007 (Test Methods for Performance Evaluation of Airborne Particle Counters). It supports classification assessments under ISO 14644-1:2015, US Federal Standard 209E, and EU and US cGMP Annex 1 (2022) for sterile manufacturing environments. The device is routinely deployed in Grade A–D cleanrooms per EU GMP, ISO Class 1–8 spaces, and critical process areas where particle control directly impacts product sterility, yield, and regulatory clearance.
Software & Data Management
Data acquisition, storage, and reporting are governed by a secure embedded software framework compliant with ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available). All measurements include timestamp, location tag (user-defined), operator ID, and environmental metadata. Internal memory retains up to 10,000 sample records with full export capability in CSV and PDF formats. Remote platform integration enables automated alerting, statistical process control (SPC) charting, and deviation tracking—supporting GLP and GMP audit readiness. Audit logs record every login, parameter modification, print command, and data transfer event with cryptographic integrity.
Applications
- Pharmaceutical manufacturing: environmental monitoring in aseptic processing suites, isolators, and lyophilization chambers per FDA and EMA guidance
- Semiconductor fabrication: validation of ISO Class 1–5 cleanrooms during photolithography, wafer handling, and packaging operations
- Medical device production: verification of cleanroom compliance for Class II/III implantables and single-use systems
- Hospital infrastructure: routine assessment of ORs, ICU ventilation systems, and bone marrow transplant units per HTM 03-01 and ISO 14644-2
- HEPA/ULPA filter integrity testing: leak detection and efficiency verification using challenge aerosols
- Food & beverage processing: monitoring of Grade A packaging zones for dairy, infant formula, and ready-to-eat products under SQF and BRCGS standards
- Aerospace component assembly: contamination control in optical coating facilities and precision bearing manufacturing
FAQ
Does the IN-CL3 meet ISO 14644-1:2015 cleanroom certification requirements?
Yes—the instrument complies with the sampling plan, statistical confidence (95% UCL), and instrument performance criteria outlined in ISO 14644-1 Annex B.
Can it be used for both static and dynamic cleanroom qualification?
Yes—it supports both operational (dynamic) and as-built (static) testing modes per EU GMP Annex 1 and ISO 14644-3.
Is the internal battery replaceable in the field?
No—the sealed Li-ion pack is service-replaceable only by authorized COMECAUSE technicians to maintain IP rating and calibration integrity.
What is the minimum detectable particle concentration?
The lower limit of quantitation is 1 particle per cubic foot (35.3 particles/m³) at 0.3 µm, verified per GB/T 6167–2007.
Does it support remote firmware updates?
Firmware updates require authenticated USB media and must be performed under controlled change management procedures per 21 CFR Part 11.