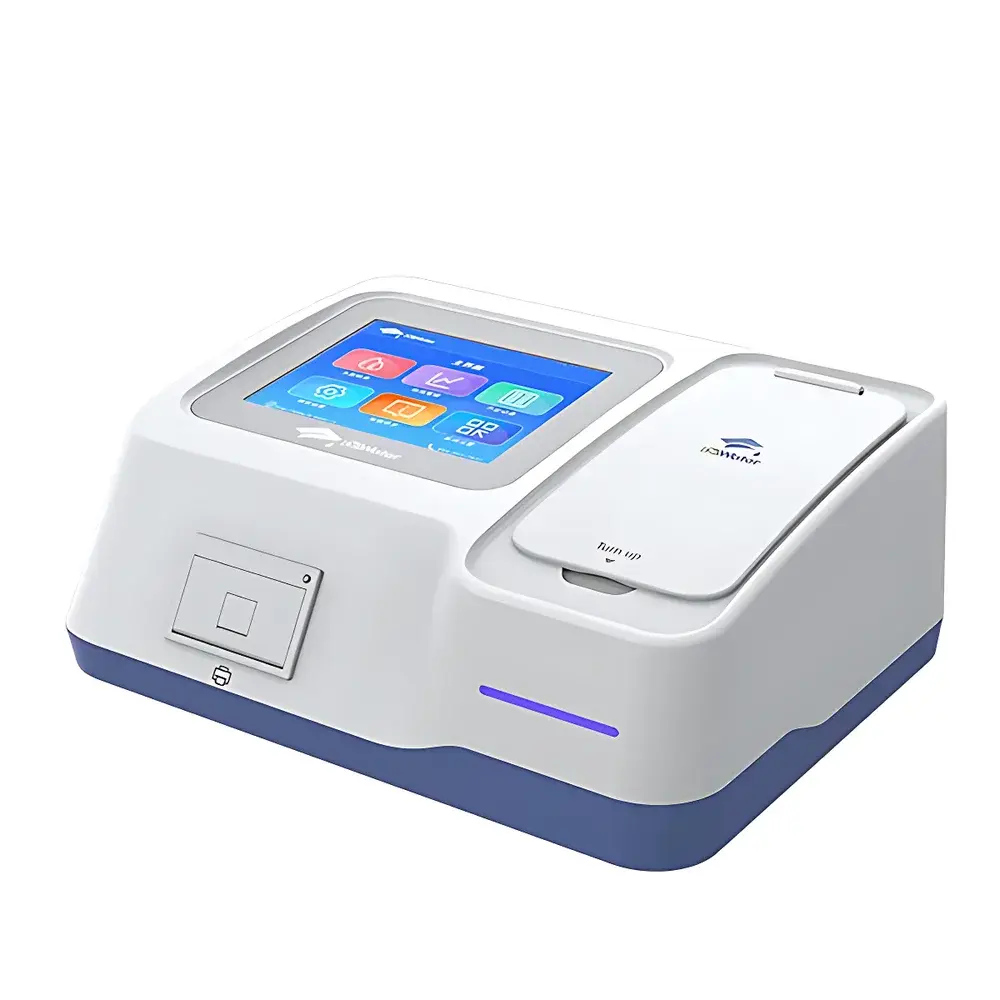
Water Doctor Dr-T9001 COD Analyzer
| Brand | Water Doctor |
|---|---|
| Origin | Hunan, China |
| Model | Dr-T9001 |
| Instrument Type | Laboratory Benchtop Analyzer |
| Measurement Principle | Rapid Digestion Spectrophotometry |
| Wavelength Range | 340–900 nm |
| COD Range | 2–20,000 mg/L |
| Accuracy | ≤ ±5% |
| Repeatability | ≤ ±2% |
| LOD | 2 mg/L |
| Digestion & Measurement Time | 10 min |
| Wavelength Accuracy | ±1 nm |
| Wavelength Repeatability | < 0.2 nm |
| Spectral Bandwidth | 4 nm |
| Transmittance Error | ±0.5% |
| Transmittance Repeatability | ≤ 0.2% T |
| Optical Stability | ≤ ±0.005 Abs/20 min |
| Resolution | 0.001 Abs |
| Light Source | Imported Cold LED (Lifespan > 100,000 h) |
| Pre-stored Calibration Curves | 1,000 |
| Data Storage Capacity | 1,000,000 records |
| Display | 8-inch HD capacitive touchscreen |
| Sample Holders | Rotatable 16-mm tube position (360°), dual cuvette positions (10 mm & 30 mm pathlength), and four independent digital electrode ports |
| Detection Modes | Absorbance, Transmittance, Concentration |
| Calibration Options | Single-point and multi-point standard calibration |
| Data Export | USB flash drive |
| Housing Material | Corrosion- and heat-resistant ABS |
| Dimensions | 392 × 315 × 170 mm |
| Weight | 4.4 kg |
Overview
The Water Doctor Dr-T9001 COD Analyzer is a laboratory-grade benchtop spectrophotometric analyzer engineered for precise, rapid quantification of Chemical Oxygen Demand (COD) in aqueous samples using standardized rapid digestion methodology. It operates on the principle of UV-Vis absorbance measurement following dichromate-based oxidation under acidic, high-temperature conditions—fully compliant with internationally referenced protocols including HJ/T 399–2007 (China’s standard for COD determination by spectrophotometry). The instrument integrates a high-stability optical detection system featuring fiber-optic light guidance, a cold LED light source with spectral stability exceeding 100,000 hours, and real-time thermal compensation to minimize drift during sequential analysis. Its dual-path optical architecture supports simultaneous validation across multiple detection geometries—tube, cuvette, and electrochemical—ensuring traceable, reproducible results across heterogeneous sample matrices.
Key Features
- Tri-modal sample interrogation: 360° rotatable 16-mm digestion tube holder eliminates tube-to-tube optical path variation; dual fixed-position cuvette slots supporting both 10 mm and 30 mm pathlength cells; and four isolated digital electrode ports for concurrent pH, ORP, conductivity, DO, or ion-selective measurements.
- High-fidelity optical engine: Wavelength range 340–900 nm with ±1 nm accuracy and <0.2 nm repeatability; spectral bandwidth of 4 nm; transmittance error <±0.5% and repeatability ≤0.2% T; optical stability ≤±0.005 Abs over 20 minutes.
- Intelligent user interface: 8-inch HD capacitive touchscreen with adjustable brightness, full Chinese GUI (English firmware optional), guided workflow navigation, and real-time status indication via tri-color LED system (green = standby/self-test complete; blue = idle/measurement complete; flashing blue = active assay; red = out-of-range alert).
- Robust data integrity architecture: Onboard thermal printer, USB mass storage export, 1-million-record non-volatile memory, and configurable alarm thresholds with color-coded visual feedback (red = exceed limit; green = within spec).
- Comprehensive calibration framework: Supports single-point verification and multi-point curve generation using NIST-traceable standards; pre-loaded library of 1,000 method-specific calibration curves aligned with HJ/T 399–2007, HJ 535–2009 (ammonia nitrogen), GB 11893–89 (total phosphorus), and other national environmental standards.
Sample Compatibility & Compliance
The Dr-T9001 is validated for use with domestic wastewater, industrial effluents (including electroplating, pulp & paper, pharmaceutical, and petrochemical discharges), groundwater, surface water, reclaimed water, and aquaculture systems. It meets functional requirements for ISO/IEC 17025-accredited laboratories performing routine COD testing under quality assurance frameworks aligned with GLP and GMP principles. While not FDA 21 CFR Part 11–certified out-of-the-box, its audit-ready data logging—including timestamped measurements, operator ID (via optional login), calibration history, and curve metadata—supports internal validation for regulated environments. All digestion protocols conform to HJ/T 399–2007, ensuring equivalence with classical reflux methods while reducing analysis time to ≤10 minutes per sample.
Software & Data Management
The embedded operating system provides intuitive menu-driven operation without external PC dependency. Data are stored with full metadata: date/time stamp, method ID, calibration reference, operator tag (if enabled), and raw absorbance values. Users may filter datasets by parameter, date range, or batch ID; plot trend curves directly on-screen; and export CSV-formatted files via USB for LIMS integration or statistical review in Excel or R. The system logs all critical events—including power cycles, calibration actions, and out-of-spec alerts—for traceability. Firmware updates are delivered via secure USB load, preserving configuration integrity across revisions.
Applications
The Dr-T9001 serves as a primary analytical platform in municipal wastewater treatment plants for process control and discharge compliance monitoring; in provincial and municipal environmental monitoring stations for routine surveillance and emergency response; in university teaching labs for instrumental analysis coursework; and in industrial QA/QC departments requiring multiparameter water profiling. Its modular design enables expansion beyond COD into ammonia nitrogen (HJ 535–2009), total phosphorus (GB 11893–89), nitrate/nitrite, turbidity, color, heavy metals (via colorimetric kits), and electrochemical parameters—all without hardware modification. Applications span regulatory reporting, pretreatment verification, bioreactor optimization, and source water vulnerability assessment.
FAQ
Does the Dr-T9001 require external software or a computer to operate?
No—it functions as a fully autonomous benchtop analyzer with onboard processing, display, storage, and printing capabilities.
Can the instrument be configured for English-language operation?
Yes; firmware supports optional English UI localization upon request prior to shipment.
What digestion reagents are compatible with the Dr-T9001?
It is designed for use with standard potassium dichromate–sulfuric acid digestion reagents meeting HJ/T 399–2007 specifications; pre-filled, certified COD digestion vials (e.g., 0–150 mg/L and 0–1500 mg/L ranges) are recommended for highest reproducibility.
Is the 360° tube rotation function mechanically driven or manual?
Rotation is motorized and automatically synchronized with photometric reading to average radial absorbance variance, minimizing tube-related systematic error.
How is optical alignment maintained over time?
The fiber-optic coupled LED source and detector assembly is factory-aligned and sealed; no user-serviceable optical adjustments are required. Routine verification is performed via built-in air-zero and standard reference checks.