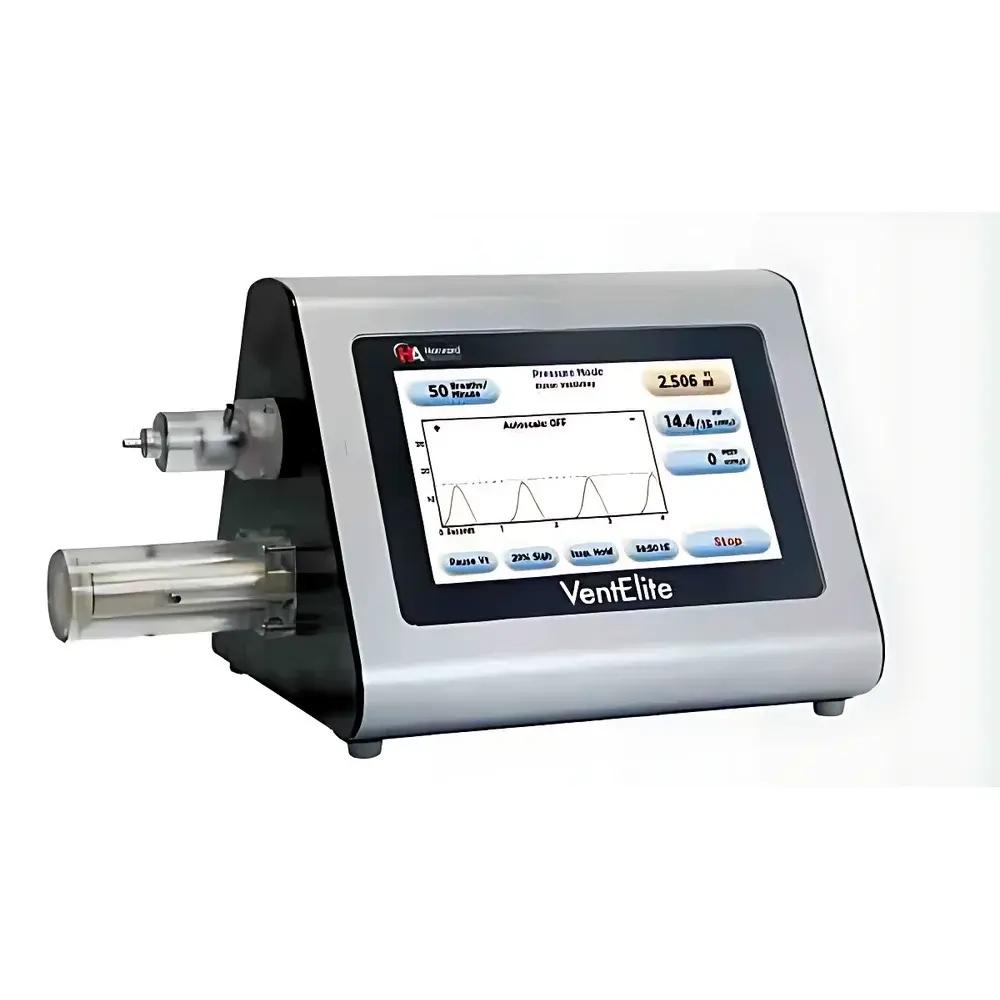
Harvard Apparatus VentElite Small Animal Ventilator
| Brand | Harvard Apparatus |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | VentElite |
| Price Range | USD 1,400 – 7,000 |
| Weight Range | 10 g – 1 kg |
| Species Compatibility | Mice to Guinea Pigs |
| Airway Connection | Inlet 3.6–3.8 mm (0.14–0.15 in), Outlet 4.0 mm (0.16 in) |
| Ventilation Modes | Volume-Controlled and Pressure-Controlled |
| Tidal Volume Range | 50 µL – 5 mL |
| Tidal Volume Resolution | 1 µL |
| Tidal Volume Accuracy | ±0.1 µL |
| Peak Inspiratory Pressure (PIP) Range | 0–125 cmH₂O |
| PIP Accuracy | ±1.75 cmH₂O |
| PIP Resolution | 2.5 cmH₂O |
| Respiratory Rate | 10–300 bpm |
| I | E Ratio: 20%–80% |
| Sigh Frequency | 1 per 10–999 breaths (adjustable) |
| Sigh Volume/Pressure | 0–20% of set tidal volume (VC mode) or PIP (PC mode) |
| Gas Supply | Medical air or non-flammable gas mixtures |
| Alarms | High/low pressure, system fault |
| Input/Output Signals | TTL-compatible (0–5 VDC, 5 mA) |
| Communication Interface | RS-485 |
| Display | 7-inch resistive touchscreen LCD |
| Power Supply | 100–240 VAC, 50/60 Hz |
| Dimensions (W × D × H) | 31.8 × 20.3 × 11.3 cm (12.5 × 8.0 × 4.45 in) |
| Weight | 3.4 kg (7.6 lbs) |
| Warranty | 2 years |
Overview
The Harvard Apparatus VentElite is a microprocessor-controlled small animal ventilator engineered for precision respiratory support in preclinical research. It employs a high-fidelity piston-cylinder actuation system coupled with digitally regulated solenoid valves to deliver reproducible, physiologically relevant ventilation across a broad weight range—from neonatal mice (10 g) to adult guinea pigs (1 kg). Unlike pressure-limited or time-cycled analog devices, the VentElite implements closed-loop feedback control in both volume-controlled (VC) and pressure-controlled (PC) modes, enabling rigorous experimental standardization required for longitudinal studies, pharmacological challenge models, and mechanistic physiology investigations. Its design adheres to core principles of pulmonary mechanics—compliance-driven tidal delivery in VC mode and pressure-limited flow dynamics in PC mode—ensuring alignment with established respiratory physiology frameworks used in peer-reviewed rodent research.
Key Features
- 7-inch resistive touchscreen interface with intuitive icon-based navigation—no external keyboard or mouse required
- Dual-mode operation: switch seamlessly between volume-controlled and pressure-controlled ventilation via on-screen toggle
- High-resolution actuation: 1 µL tidal volume resolution with ±0.1 µL accuracy, critical for low-mass subjects such as C57BL/6 neonates or transgenic dwarf models
- Configurable sigh ventilation: programmable at 1 per 10–999 breaths; sigh amplitude adjustable from 0–20% above baseline tidal volume (VC) or PIP (PC)
- Adjustable inspiratory-to-expiratory (I:E) ratio (20–80%) and inspiratory/expiratory hold—enabling modeling of obstructive or restrictive lung pathologies
- Real-time pressure monitoring with dual-stage alarm logic: independent high-pressure (≥125 cmH₂O) and low-pressure (≤0 cmH₂O) thresholds trigger audible/visual alerts and automatic safety shutdown
- TTL-compatible input/output ports (0–5 VDC, 5 mA) for synchronization with electrophysiology rigs, plethysmography systems, or optical imaging platforms
- RS-485 serial interface supporting remote parameter adjustment and data logging integration within centralized lab automation environments
Sample Compatibility & Compliance
The VentElite accommodates endotracheal tubes and tracheostomy cannulas with inner diameters of 3.6–3.8 mm (inlet) and 4.0 mm (outlet), compatible with standard rodent intubation hardware including Harvard Apparatus’ own line of stainless-steel and silicone airways. It supports medical-grade compressed air and non-flammable gas mixtures (e.g., O₂/N₂, O₂/CO₂ blends), meeting ISO 8573-1:2010 Class 2 purity requirements for laboratory gas delivery. While not certified for human use, its operational parameters align with NIH Guide for the Care and Use of Laboratory Animals (8th Edition) and AAALAC International standards for ventilatory support in acute surgical preparations. The device’s firmware architecture enables audit-ready configuration logs—supporting GLP-compliant documentation workflows when integrated with validated LIMS or ELN platforms.
Software & Data Management
No proprietary desktop software is required for basic operation; all settings are configured directly via the embedded touchscreen UI. However, the RS-485 port permits integration with third-party acquisition systems (e.g., LabChart, Spike2, or custom Python/Matlab scripts) for timestamped export of real-time pressure, flow, and trigger signals. Configuration files—including mode selection, tidal volume, PIP, rate, I:E, and sigh parameters—can be saved to internal memory and recalled per subject protocol. Firmware updates are delivered via USB flash drive (FAT32 formatted), with version history and checksum verification built into the bootloader. All alarm events are timestamped and stored in non-volatile memory for post-hoc review—supporting 21 CFR Part 11–aligned electronic record retention when paired with compliant data repositories.
Applications
- Acute in vivo electrophysiology: stable ventilation during cortical or brainstem recordings under ketamine/xylazine or isoflurane anesthesia
- Pulmonary disease modeling: COPD (elastase-induced), asthma (OVA challenge), or fibrosis (bleomycin) with tunable I:E and sigh protocols to assess dynamic compliance
- Neurorespiratory coupling studies: integration with optogenetic stimulation rigs using TTL-synchronized breath-triggered light pulses
- Developmental respiratory physiology: precise tidal delivery to P1–P7 mouse pups using ultra-low-volume settings (50–200 µL)
- Pharmacokinetic-pharmacodynamic (PK/PD) assessment: ventilatory response profiling following i.v. administration of respiratory depressants (e.g., fentanyl) or stimulants (e.g., doxapram)
- Ex vivo lung perfusion (EVLP) preparation: auxiliary support during decellularization or recellularization protocols requiring controlled mechanical inflation
FAQ
Is the VentElite suitable for long-term (>6 h) ventilation studies?
Yes—its thermally managed piston assembly and low-noise valve sequencing enable stable operation over extended durations; however, continuous monitoring and humidified gas supply are recommended per institutional IACUC protocols.
Can tidal volume be calibrated in-house?
Yes—built-in calibration mode allows verification against a NIST-traceable dry gas meter; procedure documented in the user manual (Rev. 3.2, Section 4.5).
Does the device meet electromagnetic compatibility (EMC) standards for use near MRI scanners?
It complies with IEC 61326-1:2013 Class B emissions and immunity requirements but is not MRI-conditional; maintain ≥2 m distance from 1.5T/3T scanner bores.
What maintenance is required?
Quarterly inspection of O-rings and valve seats; annual piston lubrication with FDA-approved silicone grease (included); no consumables beyond standard air filters.
Is firmware validation documentation available for GMP-regulated labs?
IQ/OQ protocols and traceable test reports are available under NDA upon request from Harvard Apparatus Technical Support.