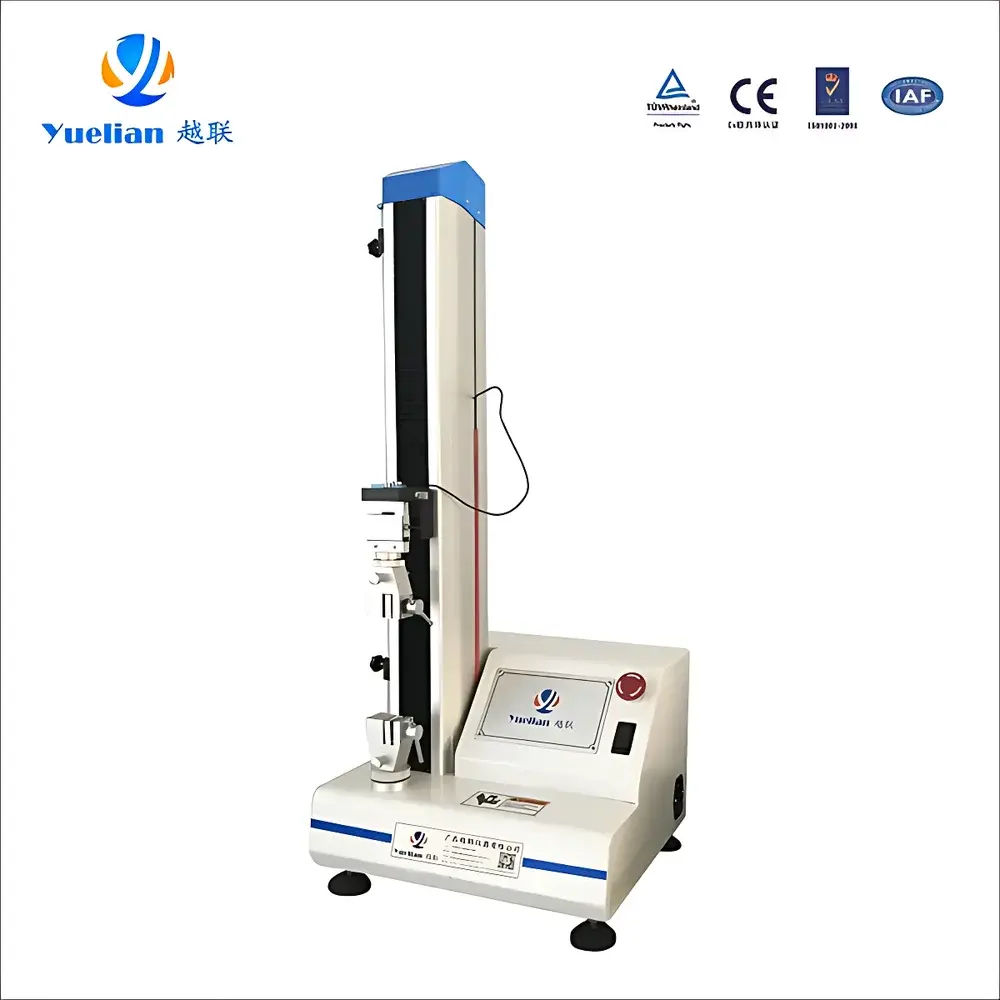
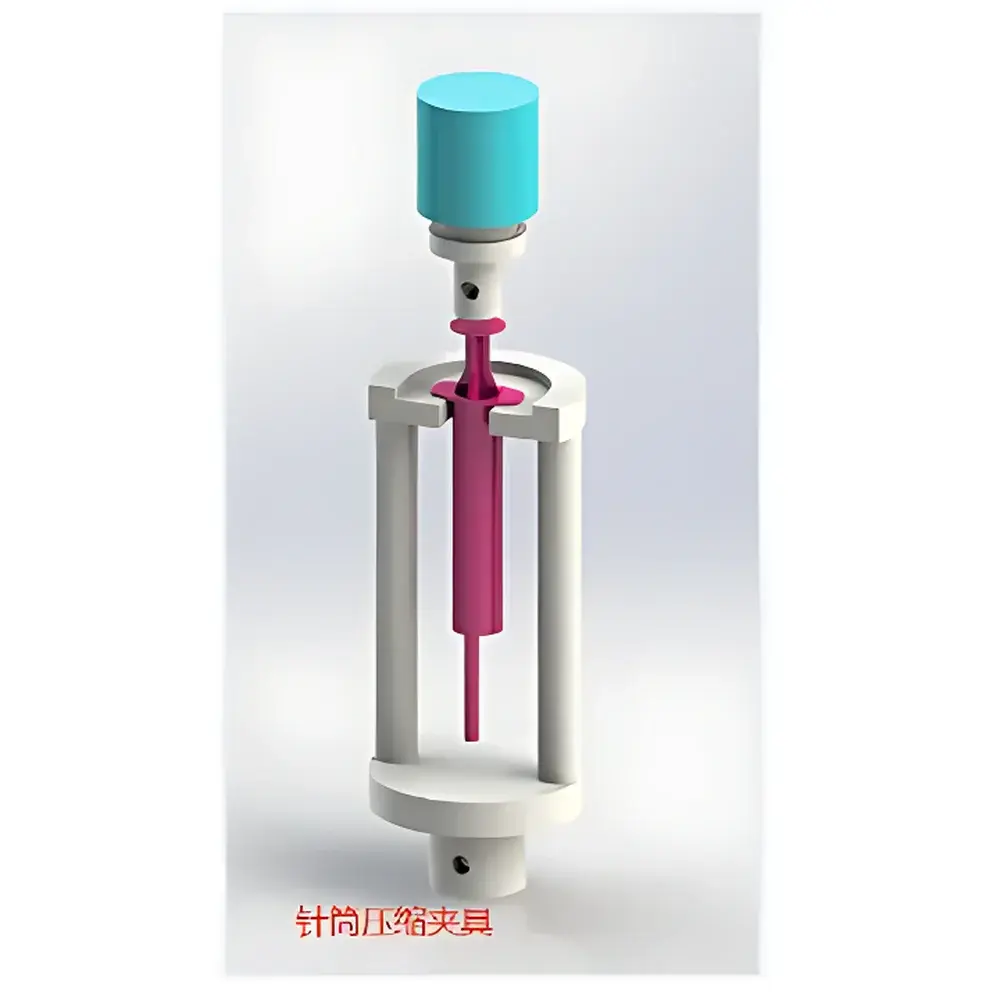
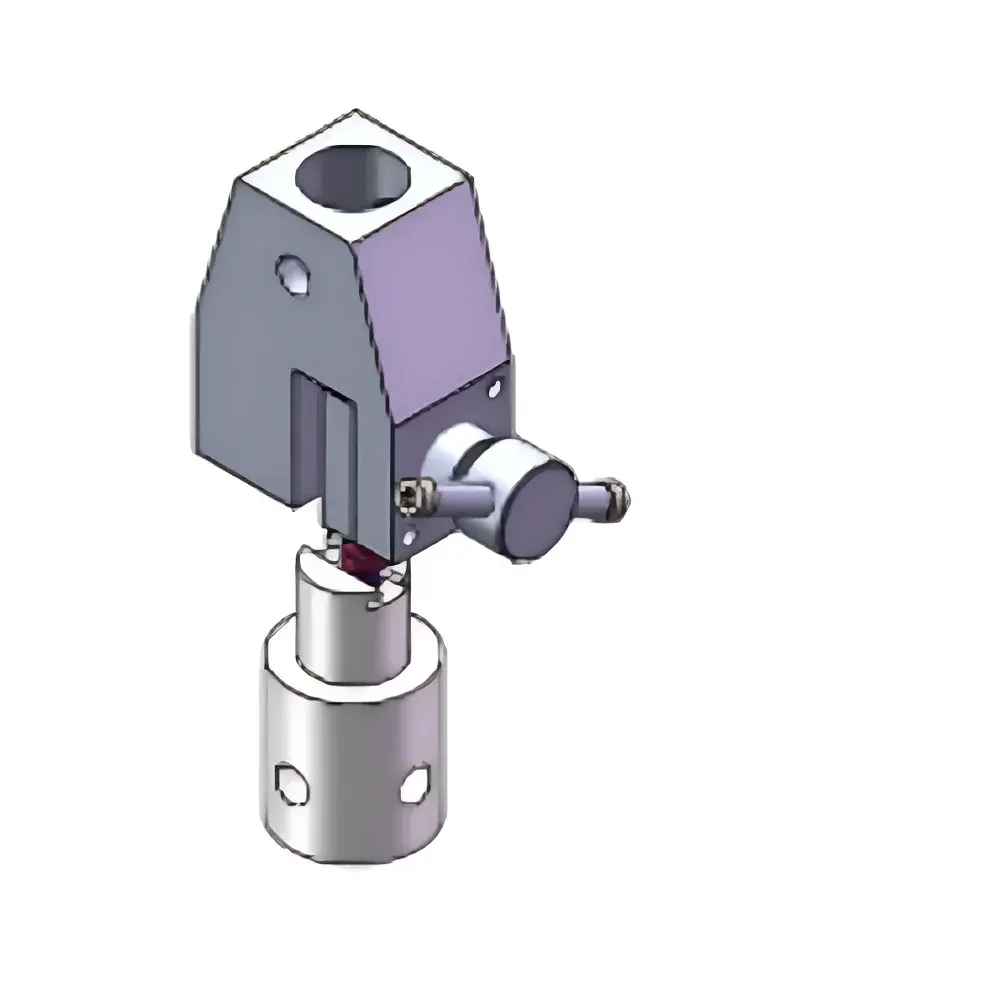
YUELIAN PW-US90 Syringe Plunger Force Tester
| Brand | YUELIAN / PW |
|---|---|
| Origin | Guangdong, China |
| Manufacturer Type | Authorized Distributor |
| Product Category | Domestic |
| Model | PW-US90 |
| Instrument Type | Electromechanical Universal Testing Machine |
| Force Capacity Options | 10 N, 20 N, 50 N, 100 N, 200 N, 500 N, 1 kN (dual-range configuration available) |
| Force Resolution | 1/500,000 full scale |
| Force Accuracy | ≤ ±0.5% of reading |
| Displacement Resolution | 0.005 mm |
| Test Stroke | 500 mm |
| Test Width | Ø55 mm |
| Speed Range | 5–500 mm/min (software-programmable) |
| Load Unit Conversion | kgf, N, lbf, g, ton, MPa |
| Stop Modes | Overload shutdown, emergency stop, specimen rupture auto-stop, upper/lower limit trigger, automatic return |
| Drive System | Stepper motor + timing belt + precision ball screw |
| Power Input | 220 V, 50/60 Hz, 10 A |
| Rated Power | 180 W |
| Dimensions (W×D×H) | 40 × 30 × 70 cm |
| Weight | ~28 kg |
| Standard Accessories | One set of syringe-specific fixtures, PC-based control software, USB interface cable |
Overview
The YUELIAN PW-US90 Syringe Plunger Force Tester is a dedicated electromechanical universal testing system engineered for precise quantification of mechanical force parameters associated with medical syringes and related drug delivery devices. It operates on the principle of controlled axial displacement under closed-loop load monitoring, enabling high-reproducibility measurement of plunger actuation force, needle penetration resistance, barrel detachment force, and syringe seal integrity. Designed in strict alignment with regulatory test requirements, the system delivers traceable, audit-ready data essential for quality assurance in ISO 13485-certified manufacturing environments and pre-market validation per FDA QSR and EU MDR Annex I criteria.
Key Features
- Modular fixture architecture supporting standardized syringe evaluation: plunger push-out, needle puncture, barrel pull-off, and cap removal tests per ISO 11608-3:2022 and GB 155810–2019.
- Dual-range force transducer configuration option (e.g., 10 N / 1 kN) enables high-resolution measurement across low-force sealing events and high-force structural failure thresholds without sensor swapping.
- High-fidelity motion control: stepper motor-driven actuation with precision ball screw transmission ensures displacement linearity ≤ ±0.01% over full 500 mm stroke; speed programmability from 5 to 500 mm/min supports both quasi-static compliance testing and dynamic response profiling.
- Sub-micron displacement resolution (0.005 mm) and 1/500,000 full-scale force resolution ensure detection of subtle mechanical transitions—such as elastomeric seal breakaway or lubricant film shear onset—critical for root cause analysis in process optimization.
- Comprehensive safety and operational safeguards: hardware-integrated emergency stop, overload cutoff, rupture-triggered auto-halt, and configurable upper/lower force limits prevent specimen damage and operator risk during unattended runs.
- Full PC-based operation via USB interface with intuitive GUI; real-time force–displacement curve visualization, statistical summary (mean, SD, CV%), pass/fail flagging against user-defined specification limits, and export-ready CSV/PDF reporting.
Sample Compatibility & Compliance
The PW-US90 accommodates standard Luer-lock and Luer-slip syringes (1 mL to 60 mL), prefilled cartridges, and auto-injector subassemblies. Fixture interchangeability allows rapid reconfiguration between plunger rod engagement, needle tip compression, and barrel grip modes. All test protocols comply with mandatory clauses of ISO 11608-3:2022 (Penetration force of needles), GB 155810–2019 (Technical requirements for syringes for injection), and supplementary references including ISO 7886-1 (Syringe performance), ASTM D3420 (Peel strength of seals), and USP (Package integrity testing). Data acquisition meets ALCOA+ principles, supporting 21 CFR Part 11-compliant electronic records when deployed with validated software configurations and access-controlled user accounts.
Software & Data Management
The embedded Windows-based control suite provides calibrated force–displacement acquisition at ≥100 Hz sampling rate, real-time curve overlay for comparative batch analysis, and automated calculation of peak force, yield point, work-to-failure, and slope-derived stiffness metrics. Audit trails log all parameter changes, test executions, and calibration events with timestamp, operator ID, and instrument serial number. Export functions support raw data (CSV), summary reports (PDF), and image-captured curves (PNG) — fully compatible with LIMS integration via ODBC or REST API extensions. Optional IQ/OQ documentation packages are available for GMP-regulated facilities requiring formal validation.
Applications
- Verification of plunger rod push-out force consistency across production lots to ensure reliable drug delivery and avoid under-dosing or over-pressure incidents.
- Quantification of needle penetration force into simulated skin substrates (e.g., polyurethane membranes per ISO 10993-23) for ergonomic and safety assessment.
- Evaluation of rubber stopper friction coefficient and lubrication efficacy under varying humidity and temperature preconditioning cycles.
- Validation of secondary packaging integrity—e.g., blister peel strength, vial cap torque release, or cartridge holder retention force—using interchangeable fixtures.
- Supporting design transfer activities by correlating material selection (barrel polymer grade, plunger elastomer durometer) with measured mechanical performance envelopes.
FAQ
Does the PW-US90 support GLP/GMP-compliant data archiving?
Yes—when configured with role-based user authentication, electronic signatures, and audit-trail-enabling firmware, the system satisfies core GLP (OECD Principles) and GMP (ICH Q7, Annex 11) data integrity requirements.
Can custom test methods be programmed beyond the built-in ISO/GB templates?
Absolutely—the software permits creation of multi-step sequences with conditional logic (e.g., hold at 50 N for 30 s, then ramp to failure), variable speed profiles, and user-defined pass/fail criteria.
Is calibration traceability provided to national standards?
Each unit ships with a factory calibration certificate traceable to CNAS-accredited reference standards; optional annual recalibration services include NIST-traceable force and displacement verification.
What maintenance intervals are recommended for long-term accuracy stability?
Routine verification using certified check weights and displacement gauges is advised before each shift; full mechanical inspection and transducer recalibration are recommended every 12 months or after 10,000 test cycles.
Are fixtures available for non-standard syringe formats (e.g., insulin pens or wearable injectors)?
Yes—custom fixture design and CNC fabrication services are offered upon submission of dimensional drawings and functional test specifications.