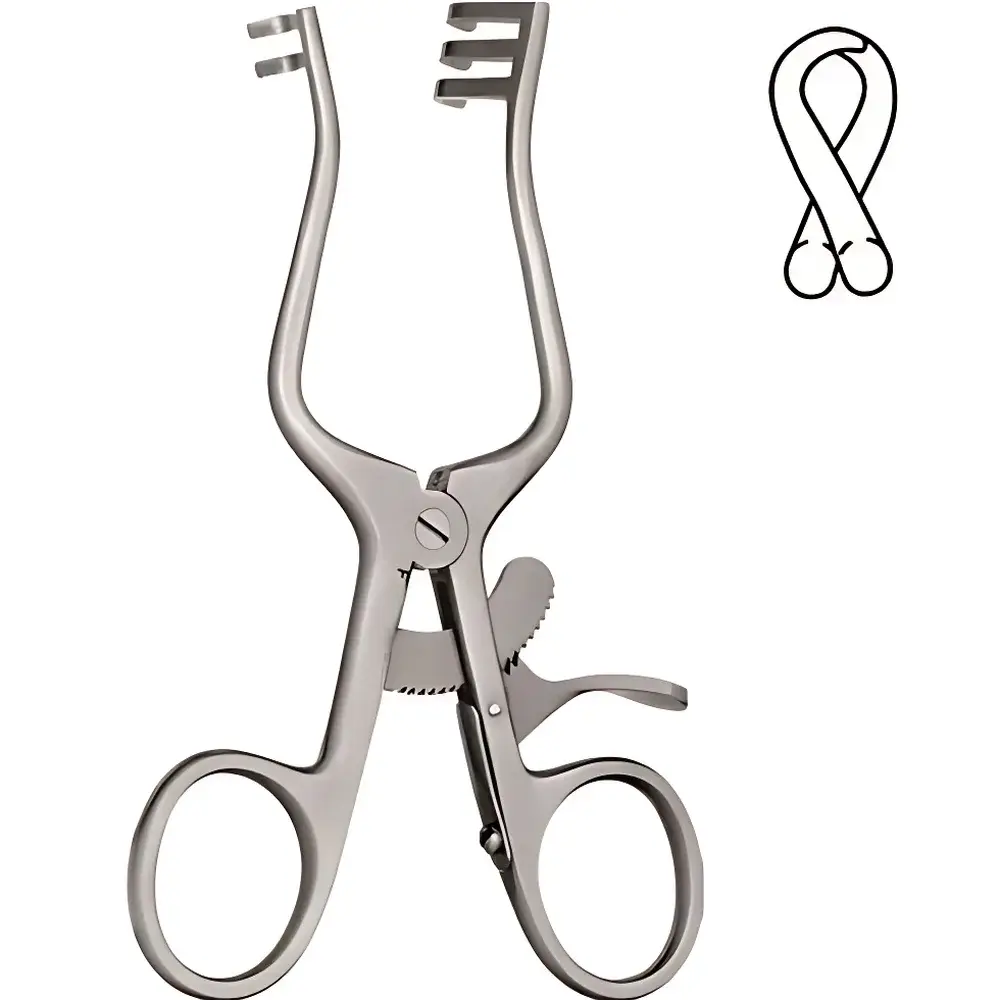
RWD R22013-11 Weitlaner Self-Retaining Retractor (2×3 Tooth, Blunt Tips)
| Brand | RWD |
|---|---|
| Origin | Shenzhen, China |
| Manufacturer Type | Original Equipment Manufacturer (OEM/ODM) |
| Country of Origin | China |
| Model | R22013-11 |
| Pricing | Upon Request |
Overview
The RWD R22013-11 Weitlaner Self-Retaining Retractor is a precision-engineered surgical instrument designed for controlled, hands-free tissue retraction during open surgical procedures. Based on the classic Weitlaner retractor configuration, this model features a 2×3 tooth arrangement with blunt, atraumatic tips to minimize soft tissue trauma while ensuring secure grip and stable exposure of the operative field. Its ratchet-based self-retaining mechanism allows incremental adjustment and reliable locking at multiple opening widths, eliminating the need for continuous manual holding and thereby reducing surgeon fatigue and improving procedural ergonomics. Constructed from high-grade, corrosion-resistant stainless steel (AISI 420 or equivalent), the retractor meets international standards for biocompatibility, sterilizability, and mechanical durability—ensuring consistent performance across repeated autoclave cycles (134 °C, 2–3 bar saturated steam, per ISO 17664 and EN 285).
Key Features
- Self-retaining ratchet mechanism with 6 distinct locking positions for precise, repeatable blade separation control
- 2×3 tooth configuration—three blunt, rounded teeth on each jaw—optimized for gentle yet firm engagement of fascial and subcutaneous layers
- Full-length, ergonomic ring handles with textured surface for enhanced tactile control and slip resistance under wet conditions
- Monoblock hinge design with zero-play articulation, ensuring long-term mechanical integrity and consistent torque transmission
- Electropolished finish compliant with ASTM F86 and ISO 10993-1 for reduced surface roughness (Ra ≤ 0.4 µm) and minimized protein adhesion
- Compatible with standard instrument tracking systems (e.g., RFID-enabled trays) and reusable instrument management protocols in centralized sterile processing departments (SPDs)
Sample Compatibility & Compliance
The R22013-11 is validated for use across general surgery, orthopedic exposure, plastic reconstructive procedures, and veterinary applications requiring moderate-to-deep wound access. It is not intended for neurosurgical or microsurgical use due to its macro-scale geometry and non-microtip profile. The device conforms to ISO 7153-1:2016 (Surgical Instruments — Metallic Materials — Part 1: Stainless Steels) and carries CE marking under Regulation (EU) 2017/745 (MDR) as a Class I reusable surgical retractor. All manufacturing processes adhere to ISO 13485:2016 quality management system requirements, including traceability of raw materials, dimensional inspection (via coordinate measuring machine), and functional testing of ratchet engagement force (measured per ISO 9626 Annex B methodology). Sterilization validation follows AAMI ST79:2023 guidelines for steam sterilization of reusable devices.
Software & Data Management
As a purely mechanical, non-electronic surgical instrument, the R22013-11 does not incorporate embedded electronics, firmware, or software interfaces. However, it supports digital workflow integration through standardized UDI (Unique Device Identification) labeling per FDA 21 CFR Part 830 and EU MDR Annex VI. Each unit bears a permanent laser-etched UDI-DI code enabling seamless entry into hospital inventory management systems (e.g., McKesson Horizon, Cerner PeriOp Manager) and automated sterilization cycle logging via SPD scanning stations. Full traceability documentation—including batch-specific material certifications, sterilization compatibility reports, and mechanical test records—is available upon request for GLP/GMP-aligned institutions conducting internal audit or regulatory review.
Applications
- Abdominal wall retraction during laparotomy and hernia repair
- Fascial layer separation in orthopedic approaches to the hip or knee joint
- Soft-tissue exposure in mastectomy and flap elevation procedures
- Veterinary surgical access in canine and equine thoracotomy/laparotomy models
- Training and simulation environments where reproducible, low-trauma retraction is required
FAQ
What is the maximum recommended opening width of the R22013-11 retractor?
The fully extended jaw separation measures 275 mm (10.8 inches), corresponding to the outermost ratchet position. This dimension is verified during final quality inspection using calibrated calipers traceable to NIST standards.
Can this retractor be sterilized using low-temperature methods such as hydrogen peroxide plasma?
Yes—the R22013-11 is validated for sterilization via saturated steam (autoclave), ethylene oxide (EtO), and hydrogen peroxide plasma (e.g., STERRAD® systems), provided cycle parameters remain within manufacturer-specified limits for metallic instruments.
Is the instrument supplied with a maintenance or sharpening protocol?
No sharpening is required or recommended, as the blunt-tip design is integral to its function and safety profile. Routine cleaning per ANSI/AAMI ST79 and visual inspection for tooth deformation or hinge wear are sufficient for continued safe use.
Does RWD provide instrument tracking support for large-volume institutional orders?
Yes—custom UDI encoding, serialized packaging, and integration-ready data files (CSV/Excel) for ERP or SPD software are available under OEM supply agreements.