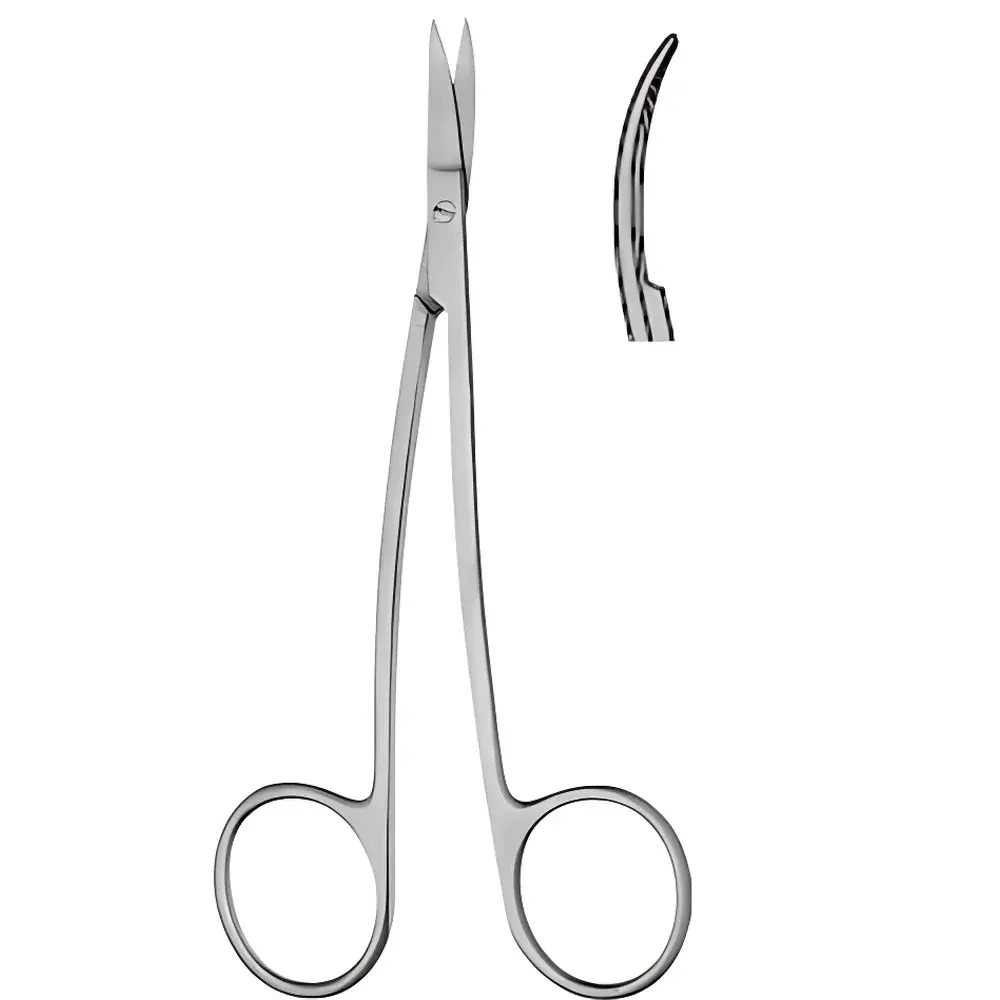
LA GRANGE Precision Scissors – Curved/Tapered Tip & Straight/Tapered Tip, 11.5 cm
| Brand | RWD |
|---|---|
| Origin | Shenzhen, China |
| Manufacturer Type | Direct Manufacturer |
| Origin Classification | Domestic (China) |
| Model | S12033-11 |
| Pricing | Upon Request |
Overview
The LA GRANGE Precision Scissors (Model S12033-11) are high-precision surgical and laboratory-grade cutting instruments engineered for demanding micro-manipulation tasks in life science research, histology, electrophysiology, and preclinical surgery. Designed under RWD’s stringent quality control protocols in Shenzhen, China, these scissors integrate ergonomic stainless steel construction with micron-level blade alignment to ensure consistent cut fidelity across repeated use. The instrument operates on mechanical shear principle—relying on precise blade edge geometry, optimal bevel angle, and controlled pivot tension—to deliver clean, non-crushing incisions in delicate biological tissues, fine sutures, neural filaments, and polymer-based lab materials. Unlike general-purpose scissors, LA GRANGE models are calibrated for low-force actuation and minimal tissue displacement, making them suitable for applications where structural integrity of adjacent structures must be preserved.
Key Features
- Two interchangeable tip configurations in a single model series: Curved/Tapered Tip (for subcutaneous or deep-tissue access with enhanced visibility) and Straight/Tapered Tip (for surface-level precision cuts and linear dissection)
- 11.5 cm total length optimized for hand stability and tactile feedback—compatible with standard sterile glove sizing and microscope-integrated workflows
- Medical-grade stainless steel (AISI 420 or equivalent) blades with mirror-finish polish and cryogenic hardening for long-term edge retention and corrosion resistance
- Smooth, self-centering tungsten-carbide reinforced pivot joint ensuring consistent blade alignment over >50,000 actuation cycles
- Ergonomic finger rings with textured matte finish to reduce slippage during prolonged procedures under wet or alcohol-saturated conditions
- Autoclavable up to 134 °C (273 °F) for 18 minutes per ISO 17664; compatible with ethylene oxide (EtO) and hydrogen peroxide plasma sterilization
Sample Compatibility & Compliance
These scissors are routinely employed in handling fragile specimens including rodent brain slices (20–500 µm), embryonic tissue explants, vascular grafts, silicone nerve cuffs, and electrospun nanofiber membranes. They meet ISO 7740 (Surgical Instruments — Requirements for Scissors) and conform to YY/T 0171–2020 (Chinese National Standard for Surgical Scissors). All units undergo 100% functional testing for blade symmetry, closure gap (<15 µm), and opening force (≤0.8 N) prior to packaging. Documentation supports GLP-compliant lab recordkeeping, including lot-specific traceability and material certification (EN 10204 3.1).
Software & Data Management
While the LA GRANGE S12033-11 is a manually operated mechanical device with no embedded electronics or firmware, its design integrates seamlessly into digitally managed laboratory environments. Each unit is laser-engraved with a unique serial number linked to RWD’s digital inventory and calibration history platform. Institutions using LIMS (Laboratory Information Management Systems) may import scissor lot data—including sterilization logs, usage cycle tracking, and maintenance records—via CSV or HL7-compatible export. For regulated environments operating under FDA 21 CFR Part 11, audit trails for instrument assignment, sterilization validation, and end-of-life retirement can be maintained through validated electronic documentation systems.
Applications
- Microdissection of hippocampal subregions in acute brain slice electrophysiology
- Trimming of collagen scaffolds and hydrogel constructs in tissue engineering workflows
- Decapsulation and trimming of zebrafish embryos during CRISPR injection setup
- Cutting of insulated microwire leads and flexible electrode arrays prior to implantation
- Preparation of paraffin-embedded tissue blocks for sectioning—especially for calcified or fibrotic samples requiring minimal compression
- Dissection of peripheral nerves in murine neuropathic pain models without axonal shearing
FAQ
Are these scissors certified for human clinical use?
No—they are designated for research-use-only (RUO) in preclinical and in vitro settings, and are not CE-marked or FDA-cleared for human therapeutic application.
What is the recommended sharpening protocol?
RWD advises against field sharpening. Blades are factory-honed to a 7–9° included angle and should be serviced exclusively by authorized RWD technical centers using optical alignment fixtures and diamond-lapping calibration standards.
Can these scissors be used with magnetic resonance imaging (MRI) equipment?
Yes—constructed entirely from non-ferromagnetic austenitic or martensitic stainless steel variants, they pose no MRI safety hazard at field strengths up to 7 Tesla when properly de-magnetized post-sterilization.
Is there a warranty or service agreement available?
RWD provides a 24-month limited warranty covering material and workmanship defects. Extended calibration and refurbishment contracts are available upon request for institutional procurement programs.