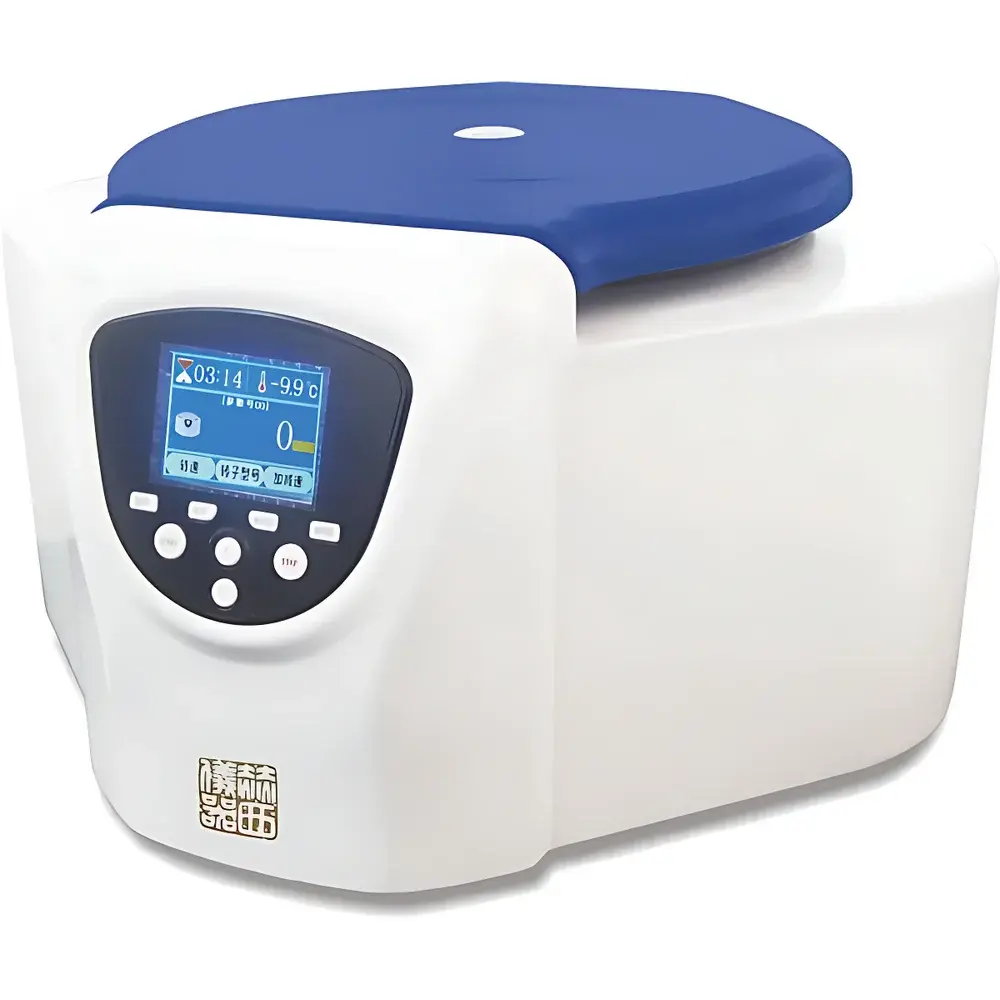
Hershey TDZ4 Benchtop Low-Speed Centrifuge
| Brand | Hershey |
|---|---|
| Origin | Hunan, China |
| Model | TDZ4 |
| Instrument Type | Benchtop Centrifuge |
| Speed Range | Up to 4000 rpm |
| Max RCF | 2600 ×g |
| Max Capacity | 24 × 10 mL |
| Acceleration/Deceleration | 10-step control |
| Display | TFT-LCD with touch + physical keys |
| Safety Certifications | CFDA Registered, ISO 9001:2015 & ISO 13485:2016 compliant |
| Compliance | RoHS 2015/863 (silicone O-ring seal) |
| Noise Level | ≤60 dB(A) |
| Power Supply | AC 220 V, 50 Hz |
| Weight (with rotor) | 20 kg |
| Dimensions (L×W×H) | 500 × 450 × 280 mm |
Overview
The Hershey TDZ4 Benchtop Low-Speed Centrifuge is a CE-marked, regulatory-compliant centrifugation system engineered for routine separation tasks in clinical diagnostics laboratories, hospital clinical chemistry departments, blood banks, and biomedical research facilities. Operating on the principle of sedimentation under controlled centrifugal force, the TDZ4 delivers reproducible separation of biological suspensions—including whole blood, serum, plasma, urine sediments, and cell culture supernatants—based on differences in particle density and size. Its design prioritizes operational safety, user ergonomics, and compliance with international quality management standards applicable to in vitro diagnostic (IVD) support equipment. With a maximum rotational speed of 4000 rpm and a peak relative centrifugal force (RCF) of 2600 ×g, the TDZ4 is optimized for low-speed applications where gentle pelleting or phase separation is required without compromising sample integrity—particularly critical for fragile analytes such as coagulation factors, extracellular vesicles, or temperature-sensitive enzymes.
Key Features
- Brushless DC motor with high-torque output and microprocessor-based vector control ensures stable rotation, minimal thermal drift, and long service life—critical for repeated daily use in high-throughput clinical labs.
- Integrated dual-mode interface: 4.3-inch TFT-LCD touchscreen paired with tactile physical keys enables intuitive parameter entry, real-time monitoring of setpoint vs. actual rpm/RCF/time, and on-the-fly adjustments without interrupting the run.
- Comprehensive safety architecture includes automatic rotor recognition (via mechanical coding), imbalance detection with auto-shutdown, overspeed and overtemperature protection, and an electromagnetic door lock compliant with IEC 61010-2-020.
- Biological containment certified: angular rotor features a fully encapsulated silicone elastomer gasket meeting EU RoHS Directive 2015/863, preventing aerosol leakage during centrifugation of potentially infectious specimens.
- Robust mechanical construction: 304 stainless steel centrifuge chamber, powder-coated steel housing, reinforced front panel, and triple-layer protective casing meet structural durability requirements per ISO 13485:2016 Annex C for medical device ancillary equipment.
- Programmable operation: 20 user-defined protocols stored in non-volatile memory; recall of last-used method at power-on; adjustable acceleration/deceleration profiles (10 steps each) to minimize resuspension of delicate pellets.
Sample Compatibility & Compliance
The TDZ4 accommodates standard 10 mL conical tubes (e.g., Falcon 14–959E, Sarstedt 62.542.001) in its fixed-angle rotor configuration (24-place, 10° angle). It supports common clinical specimen types including EDTA/K₂EDTA anticoagulated whole blood, serum separator tubes (SST), lithium heparin plasma, cerebrospinal fluid (CSF), and urinary sediment preparations. All materials contacting samples are certified non-cytotoxic and extractables-tested per ISO 10993-5. The device carries CFDA registration (Class II IVD accessory) and adheres to essential requirements outlined in Annex I of the EU In Vitro Diagnostic Medical Devices Regulation (IVDR) 2017/746 for ancillary instrumentation. Documentation includes full traceability of manufacturing processes aligned with ISO 13485:2016, supporting GLP/GMP-aligned laboratory audits.
Software & Data Management
While the TDZ4 operates as a standalone instrument without network connectivity, its embedded firmware provides audit-ready operational logging: each run records timestamp, rotor ID (mechanically encoded), final rpm, actual RCF, elapsed time, and any triggered safety event (e.g., imbalance alert). Logs are retained internally for ≥1000 cycles and exportable via USB port in CSV format for integration into LIMS or QA documentation systems. The interface supports multi-language localization (English, Chinese, Russian, Portuguese), facilitating deployment across multinational healthcare networks. Firmware updates are performed offline using signed binary files, maintaining data integrity and regulatory compliance per FDA 21 CFR Part 11 principles for electronic records.
Applications
- Routine serum/plasma separation from whole blood specimens prior to clinical chemistry or immunoassay analysis.
- Preparation of platelet-poor plasma (PPP) for coagulation testing (PT, APTT, fibrinogen).
- Sediment concentration of urine for microscopic urinalysis per CLSI GP16-A3 guidelines.
- Clarification of bacterial or yeast cultures in microbiology workflows.
- Isolation of peripheral blood mononuclear cells (PBMCs) when used with density gradient media (e.g., Ficoll-Paque PLUS) at sub-2000 ×g conditions.
- Processing of environmental water samples for pathogen concentration in public health surveillance programs.
FAQ
What regulatory certifications does the TDZ4 hold for clinical use?
The TDZ4 is registered with China’s National Medical Products Administration (NMPA, formerly CFDA) as a Class II IVD accessory and conforms to ISO 9001:2015 and ISO 13485:2016 quality management standards.
Is the rotor autoclavable?
No—the fixed-angle rotor is not designed for autoclaving; cleaning is performed with 70% ethanol or neutral detergent followed by air drying. Sterilization is achieved via validated chemical disinfection protocols compatible with stainless steel surfaces.
Can the TDZ4 be operated on unstable power grids?
Yes. Its wide-input voltage range (100–230 V AC, 50/60 Hz) and active power factor correction ensure reliable performance in environments with ±10% voltage fluctuation, common in rural or mobile clinical units.
Does the TDZ4 support rotor calibration verification?
The unit includes built-in speed verification functionality via optical tachometer input; users may perform periodic checks using NIST-traceable handheld tachometers per manufacturer-recommended intervals (every 6 months or after 500 runs).
How is data integrity ensured during power interruption?
All critical runtime parameters and error logs are written to non-volatile flash memory before execution begins, ensuring no loss of audit trail data—even during unexpected shutdown.