LEI-TECH RX51 Series Whole Slide Scanner
| Brand | LEI-TECH |
|---|---|
| Origin | Zhejiang, China |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Country of Manufacture | China |
| Model | RX51 Series |
| Pricing | Upon Request |
Overview
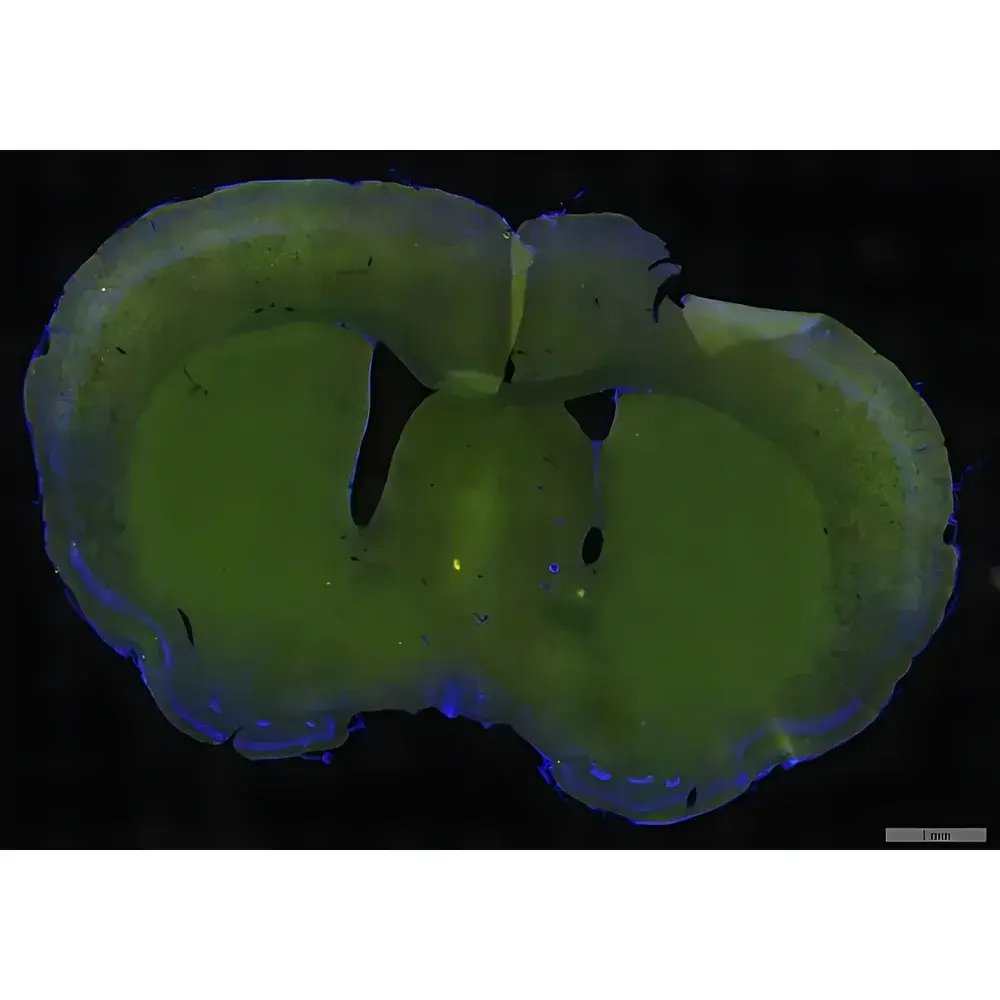
The LEI-TECH RX51 Series Whole Slide Scanner is a high-precision, automated digital pathology imaging platform engineered for quantitative morphological analysis in clinical diagnostics, translational research, and pharmaceutical development. Based on transmitted-light brightfield and epi-fluorescence microscopy principles, the system employs motorized XYZ stages, multi-objective turret optics, and synchronized fluorescence filter wheels to acquire gigapixel-resolution whole-slide images (WSIs) with sub-micron spatial fidelity. Its optical architecture integrates infinity-corrected, plan-apochromatic objectives (SAPO) with field number FN26.5 mm, enabling wide-field, uniform illumination and minimal chromatic aberration across visible to near-infrared spectra (400–1000 nm). Designed for GLP-compliant laboratory environments, the RX51 supports dual-mode acquisition—brightfield and multi-channel fluorescence—within a single scan session, eliminating manual repositioning or instrument switching.
Key Features
- Modular scanning platform supporting 1-, 2-, and 4-inch slide formats via interchangeable tray carriers—software-recognized and auto-configured per batch
- Motorized 3–5 position objective turret with standard magnifications: 10×, 20×, 40×, and 100× oil immersion; optional 5-objective configuration with automated oil dispensing system
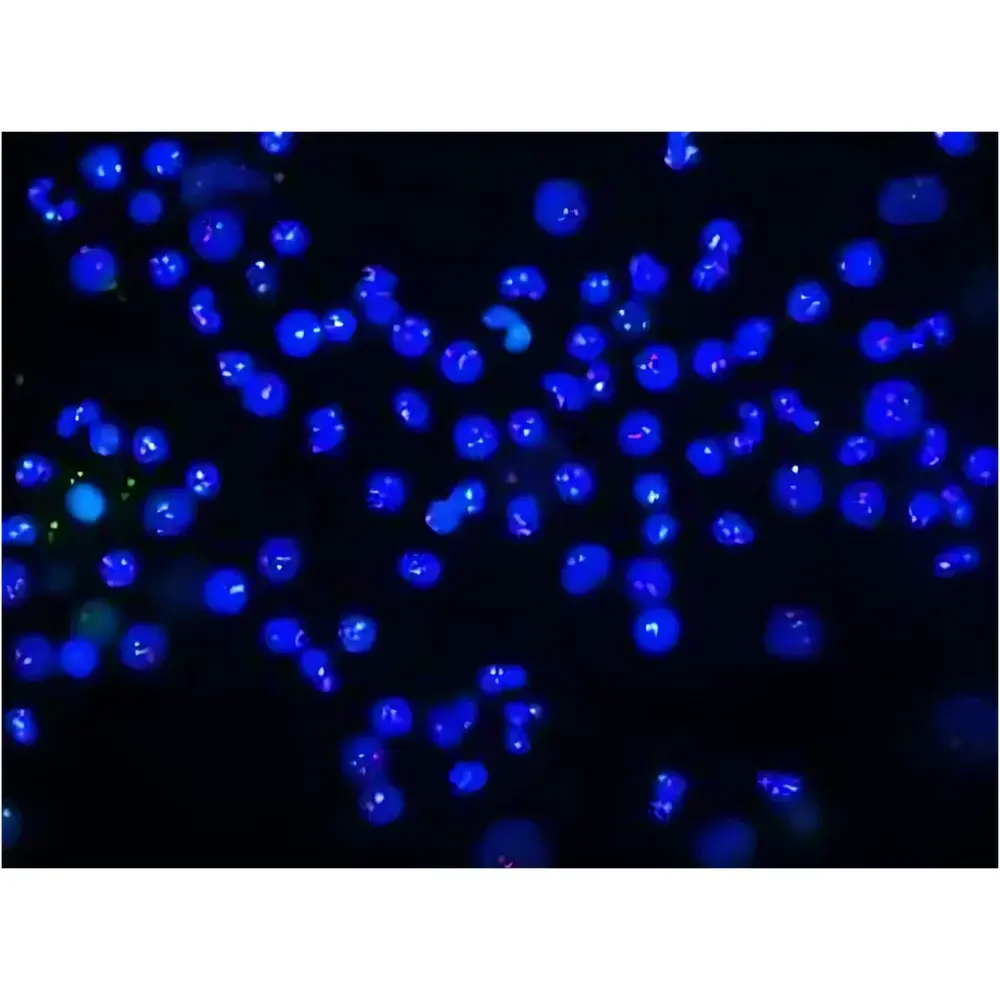
- 6-position fluorescence filter wheel (expandable to 10), each slot accommodating excitation/emission filters and dichroic mirrors for multiplexed IHC, IF, and FISH applications
- Intelligent autofocus with ≤20 ms/µm response time and Z-stack fusion for extended-depth-of-field imaging of thick or uneven tissue sections
- Barcode recognition engine supporting QR Code, DataMatrix, Code 39/128, and PDF417—auto-linking metadata to WSI filenames with zero manual entry
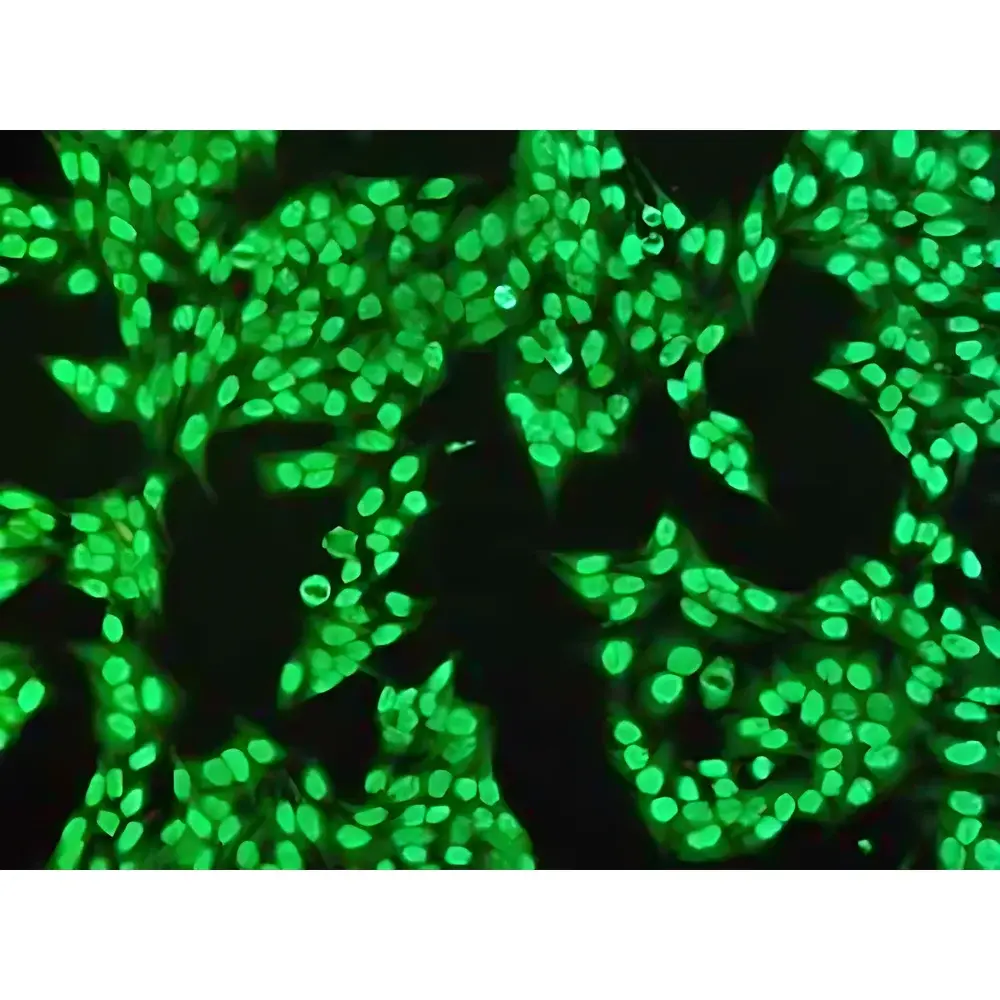
- True-color LED illumination (CRI R9 ≥ 90) ensuring accurate color reproduction for H&E, trichrome, and special stains critical to diagnostic interpretation
- Low-autofluorescence optical train and SAPO objectives with NA up to 1.45, optimized for high signal-to-noise ratio in low-abundance fluorophore detection
- High-speed linear stage with positioning accuracy ≤0.2 µm and travel velocity ≥60 mm/s, enabling rapid whole-slide acquisition at 20× and 40×
Sample Compatibility & Compliance
The RX51 accommodates standard 1″ × 3″ glass slides as well as custom-sized specimens—including cytology smears, tissue microarrays (TMAs), and large-format histology sections—via adaptable tray geometry. All optical components comply with ISO 10934-1 (microscope nomenclature) and ISO 8578 (objective lens specifications). The system meets essential requirements for diagnostic use under CLIA and CAP guidelines when deployed in validated workflows. Data integrity is reinforced through audit-trail-enabled software adhering to FDA 21 CFR Part 11 principles, including electronic signatures, user access controls, and immutable log histories. Mechanical design incorporates Panasonic reflective sensors and magnetic linear encoders to eliminate slide fragmentation—a documented failure mode in high-throughput scanning—ensuring specimen integrity throughout the digitization process.
Software & Data Management
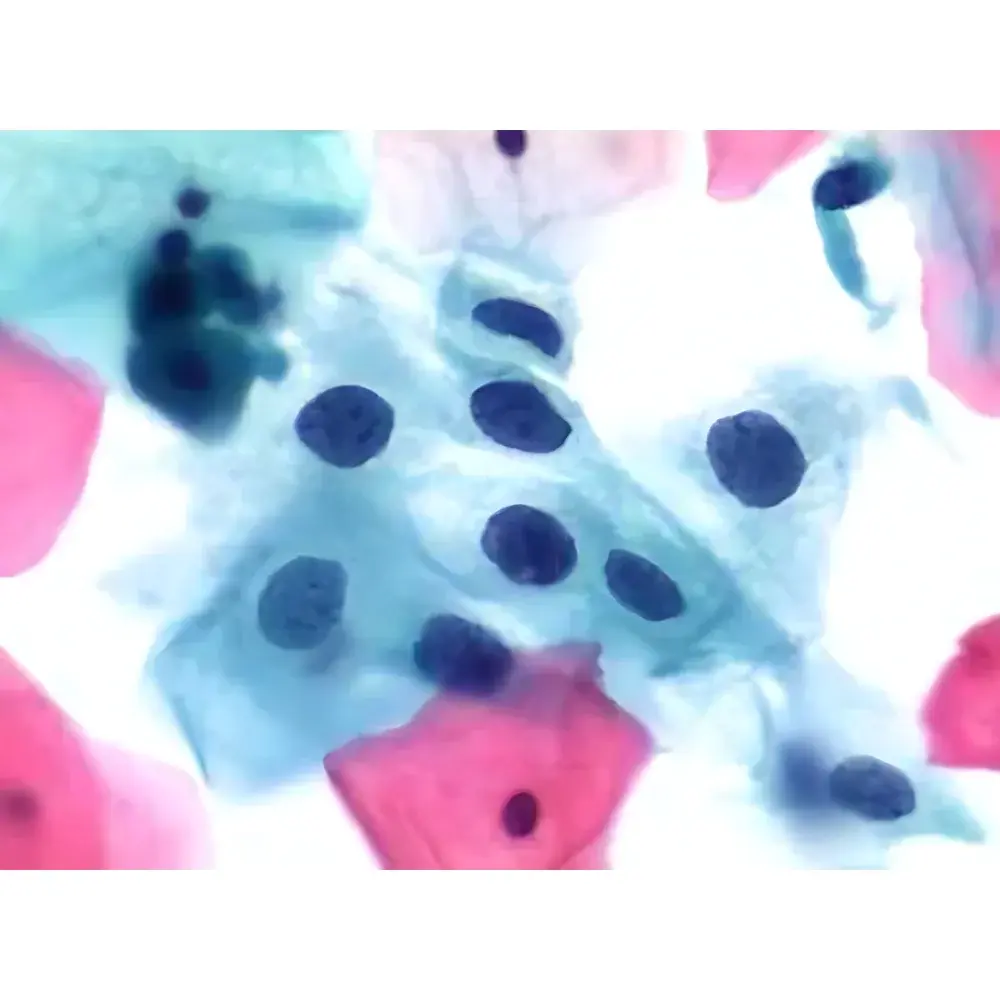
The RX51 operates on a browser-based (B/S) architecture, requiring no local client installation. Scanning, viewing, annotation, and collaborative review occur within a unified web interface compatible with Windows, macOS, iOS, and Android. Image data are stored in DICOM-SR compliant format with embedded acquisition metadata (objective, exposure, gain, focus map, barcode ID). The integrated TPS (ThinPrep Processing System) module performs AI-assisted primary screening of cervical cytology slides, highlighting morphologically atypical regions for pathologist review. Quantitative IHC analysis tools compute H-score, percentage positivity, and subcellular localization metrics using calibrated intensity thresholds. All operations—including remote expert consultation, multi-user annotation, and version-controlled slide comparison—are logged with timestamped, role-based access records to support GxP traceability.
Applications
- Clinical pathology: Routine digitization of surgical pathology and cytology specimens for primary diagnosis, second opinion, and telepathology networks
- Pharmaceutical R&D: High-content analysis of preclinical tissue sections for biomarker validation, toxicopathology assessment, and drug mechanism studies
- Academic research: Multiplexed fluorescence imaging for spatial transcriptomics correlation, tumor microenvironment mapping, and longitudinal cohort studies
- Medical education: On-demand virtual slide libraries with synchronized annotation layers, zoom-locked multi-slide comparison, and curriculum-integrated case repositories
- Quality assurance: Automated QC reporting on focus uniformity, illumination flatness, barcode read success rate, and file integrity checksums
FAQ
Does the RX51 support FDA-cleared diagnostic use?
The RX51 is intended for research use and clinical laboratory use under laboratory-developed test (LDT) frameworks. Diagnostic clearance requires site-specific validation per local regulatory authority requirements.
Can the system perform simultaneous brightfield and fluorescence scanning?
Yes—multi-modal acquisition is supported in a single workflow, with independent exposure control and channel registration aligned to pixel-level precision.
What image formats are exported?
WSIs are saved in pyramidal TIFF and DICOM-SR formats; thumbnails and region-of-interest exports are available in JPEG2000 and PNG.
Is cloud storage integration available?
The platform supports secure S3-compatible object storage and on-premise NAS deployment; HIPAA-compliant encryption and TLS 1.3 transport are enforced by default.
How is calibration maintained across instruments?
Each unit ships with NIST-traceable calibration slides; periodic verification is performed using built-in photometric and geometric reference targets embedded in the optical path.