DHS iSC Series Cell Imaging and Counting System
| Brand | DHS |
|---|---|
| Model | iSC / iSC-PRO |
| Origin | Tianjin, China |
| Instrument Type | Integrated Microscopic Image Analysis System with Touchscreen Display and Built-in CMOS Camera |
| Optical Modes | Brightfield & Phase Contrast |
| Objective Options | Standard 4×, 10×, 20×, 40× (optional) |
| Illumination | Long-life LED (50,000 h rated lifetime, intensity stability ±2% over 8 h) |
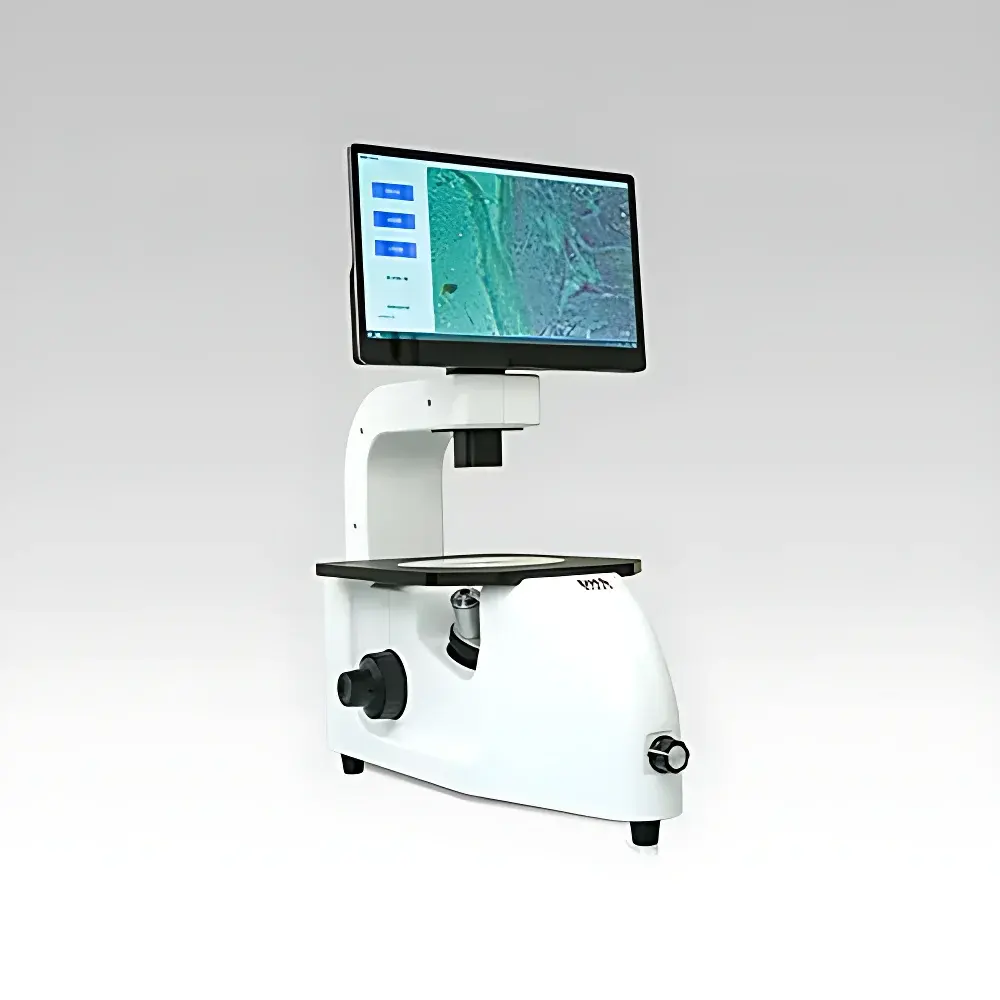
| Image Sensor | Color CMOS, 5 MP resolution (2592 × 1944 pixels), 12-bit dynamic range |
| Display | 10.1″ HD IPS touchscreen (1280 × 800), adjustable tilt (−15° to +60°) |
| Environmental Compatibility | Certified for operation inside Class II Biological Safety Cabinets (BSCs) |
| Software Features | Real-time image freeze, contrast/saturation adjustment, automated cell counting algorithm (based on circularity/size thresholding), export of CSV/Excel reports, audit trail logging (GLP-compliant metadata tagging) |
| Compliance | Meets ISO 13485 design control principles for in vitro diagnostic support equipment |
Overview
The DHS iSC Series Cell Imaging and Counting System is an integrated, benchtop optical instrument engineered for quantitative brightfield and phase contrast microscopy in cell culture quality control, bioprocessing monitoring, and pre-transfection viability assessment. Unlike conventional upright microscopes requiring ocular observation, the iSC platform replaces eyepieces with a calibrated 10.1″ HD touchscreen display—eliminating observer-induced visual fatigue during extended sessions while maintaining optical path integrity. Its compact, sealed chassis (W × D × H: 285 × 310 × 370 mm) and low electromagnetic emission profile enable safe deployment directly within Class II biological safety cabinets, minimizing aerosol exposure and cross-contamination risk during live-cell handling. The system employs a Köhler-illuminated LED light engine with thermally stabilized driver circuitry, ensuring photometric consistency (< ±2% intensity drift over 8 hours) critical for longitudinal image comparison. Optical performance is maintained across interchangeable objectives (4×, 10×, 20×, 40×), with parfocal alignment and standardized DIN 1900/2000 mounting threads.
Key Features
- Touchscreen-native interface with dual input support (capacitive touch + USB mouse), enabling glove-compatible operation inside biosafety cabinets
- Real-time image freeze functionality decouples analysis from sample motion—essential for non-adherent suspension cultures or viscous media where stage vibration compromises focus stability
- Adjustable contrast and saturation controls permit optimization of refractive index gradients in unstained phase contrast specimens without hardware reconfiguration
- On-device cell counting algorithm applies adaptive thresholding and morphological filtering to distinguish viable cells from debris based on area (10–200 µm²), circularity (>0.6), and edge gradient strength
- Integrated 5 MP color CMOS sensor delivers 12-bit raw data capture with >60 dB signal-to-noise ratio at ISO 400, supporting downstream histogram-based QC metrics
- Modular hardware architecture allows field-upgradeable components—including optional motorized XY stage (Order No. 0401600-1) and advanced counting software license (0401600-2)
Sample Compatibility & Compliance
The iSC system accommodates standard 35 mm, 60 mm, and 100 mm Petri dishes; T25/T75 flasks (with lid removal); and hemocytometer chambers (Neubauer-improved and disposable types). It supports both adherent (e.g., HEK293, CHO-K1) and suspension (e.g., Jurkat, Sf9) cell lines across passage ranges P3–P25. All firmware and application software comply with ISO 13485:2016 Annex C for medical device-related instrumentation used in IVD workflows. Audit trail functionality records user ID, timestamp, parameter changes, and export events—meeting GLP documentation standards per OECD Principles and enabling traceability for internal QA audits. While not classified as a medical device under FDA 510(k), its software architecture implements 21 CFR Part 11 controls including role-based access, electronic signature capture, and immutable log archiving.
Software & Data Management
The embedded operating system runs a Linux-based real-time kernel optimized for deterministic image acquisition latency (<120 ms frame-to-display). Data management includes local storage on 64 GB eMMC flash (expandable via USB 3.0), encrypted CSV exports with embedded metadata (objective magnification, exposure time, gain setting, date/time stamp), and DICOM-SR (Structured Reporting) compatibility for integration into LIMS environments. The counting module generates summary statistics per field-of-view: total count, mean diameter (µm), coefficient of variation (%), and viability percentage (if paired with trypan blue staining protocol). All software updates are delivered via signed OTA packages validated against SHA-256 checksums.
Applications
- Routine confluence estimation prior to subculturing or transfection
- Post-thaw viability assessment of cryopreserved primary cells
- Bioreactor inoculum qualification in upstream bioprocessing
- QC release testing for cell banking operations (per USP <1043> guidelines)
- Educational use in undergraduate cell biology laboratories requiring quantitative imaging literacy
- Support for ISO 20387-accredited biobanks performing donor-derived cell line verification
FAQ
Does the iSC system require external computer connectivity for basic counting functions?
No—core imaging, freeze, and counting operations execute natively on the onboard processor. A PC is only required for advanced batch analysis or LIMS integration.
Can the iSC-PRO model perform automated focus stacking?
No—the iSC-PRO enhances counting accuracy via improved low-light SNR and extended dynamic range but does not include motorized Z-drive or z-stack acquisition capability.
Is calibration traceable to NIST standards?
Yes—each unit ships with a certificate of calibration referencing NIST-traceable stage micrometer (10 µm pitch) and photometric reference tile (CIE LAB L* = 50.0 ± 0.2).
What is the maximum supported file export rate during time-lapse acquisition?
At 1 fps continuous capture (1920 × 1080), sustained write speed exceeds 8 MB/s to internal storage, supporting ≥4-hour unattended recording.
Are software updates provided free of charge during the warranty period?
Yes—DHS provides all firmware and application patches at no cost for 24 months post-purchase, including security hardening and regulatory alignment updates.