Invitrogen EVOS XL Core Transmitted-Light Cell Imaging System
| Brand | Invitrogen |
|---|---|
| Origin | USA |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Import Status | Imported |
| Model | EVOS XL Core |
| Pricing | Upon Request |
Overview
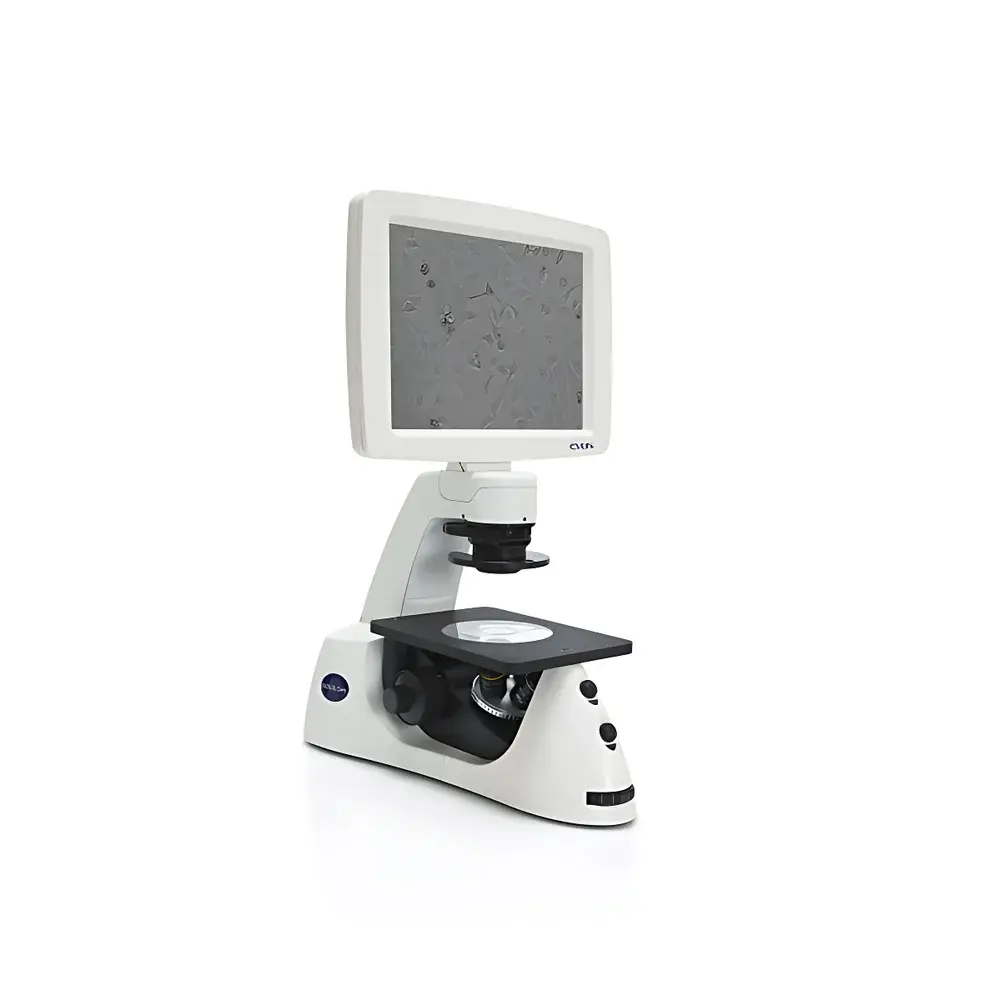
The Invitrogen EVOS XL Core Transmitted-Light Cell Imaging System is an integrated, benchtop-grade inverted microscope platform engineered for routine phase contrast and brightfield imaging of live cells under sterile conditions. Built upon a fixed-optics, LED-illuminated transmitted-light architecture, the system eliminates the need for external computers, complex alignment procedures, or optical path realignment—enabling rapid visual assessment and documentation directly within biosafety cabinets or laminar flow hoods. Its compact footprint (W × D × H ≈ 28 × 30 × 35 cm) and lightweight design (~10 kg) allow seamless integration into Class II biosafety cabinets without compromising workspace ergonomics or airflow integrity. The system operates on Köhler illumination principles with a fixed magnification objective turret (typically 4×, 10×, and 20× phase contrast objectives), delivering consistent contrast and resolution across daily cell culture monitoring, confluency estimation, and morphological validation tasks.
Key Features
- Integrated all-in-one design: Embedded CMOS digital camera, high-transmission optical train, 15.6-inch full-HD LCD display, and onboard USB storage—no external PC required for image acquisition or review.
- Sterile workflow compatibility: Fully functional inside laminar flow hoods or biosafety cabinets; transparent acrylic viewing window enables real-time observation without opening the cabinet sash.
- Instant-on LED illumination: Cold-white LED light source with >25,000-hour lifetime; zero warm-up time, stable intensity output, and minimal thermal load on samples.
- Maintenance-free operation: Factory-aligned optics, fixed focus calibration, and sealed optical housing eliminate routine recalibration, cleaning, or mechanical adjustment.
- Ergonomic usability: Intuitive touchscreen interface with one-touch capture, auto-exposure optimization, and on-screen measurement tools (e.g., scale bar overlay, ROI annotation).
Sample Compatibility & Compliance
The EVOS XL Core supports standard tissue-culture vessels including 6–96-well plates, T-flasks (T25–T225), Petri dishes, and chamber slides—all imaged in situ without sample transfer. It complies with ISO 13485 design control requirements for medical device manufacturing environments and meets general laboratory safety standards per IEC 61010-1. While not FDA-cleared as a diagnostic instrument, its hardware architecture aligns with GLP/GMP documentation practices when paired with controlled file naming, timestamped metadata export (EXIF-compliant JPEG/TIFF), and audit-ready USB storage logs. The system’s closed optical path and absence of mercury vapor lamps reduce hazardous material handling concerns, supporting institutional biosafety office (BSO) compliance protocols.
Software & Data Management
Imaging workflows are managed via the embedded EVOS Software Suite, which provides native support for lossless TIFF and compressed JPEG export, multi-frame time-lapse sequencing (with user-defined intervals), and basic quantitative analysis—including confluency estimation algorithms based on pixel-intensity thresholding. All images retain embedded metadata (date/time, objective used, exposure settings, user ID if configured). Exported files conform to FAIR data principles (Findable, Accessible, Interoperable, Reusable) and integrate natively with common LIMS platforms via standardized folder-based ingestion. No software licensing or subscription fees apply; firmware updates are delivered via USB drive and validated against SHA-256 checksums for integrity verification.
Applications
- Monitoring pluripotent stem cell colonies (e.g., iPSCs) during passaging, clonal expansion, and morphology-based selection prior to picking.
- Assessing transfection efficiency and reporter gene expression in adherent mammalian lines using brightfield-only workflows.
- Validating monolayer integrity and detecting early signs of contamination (e.g., mycoplasma-induced granularity, fungal hyphae) during routine QC checks.
- Documenting primary cell isolation outcomes (e.g., PBMC adherence, neural rosette formation) with timestamped, version-controlled image archives.
- Supporting teaching laboratories where ease-of-use, reproducibility, and minimal IT dependency are critical success factors.
FAQ
Can the EVOS XL Core be used inside a CO2 incubator?
No—it is designed for ambient-air environments only; temperature and humidity control inside CO2 incubators exceed operational specifications.
Does it support fluorescence imaging?
No—this model is limited to transmitted-light modalities (phase contrast and brightfield); fluorescence capability requires the EVOS FL Auto or EVOS M5000 platforms.
Is remote access or network connectivity supported?
Not natively; however, exported USB media can be ingested into institutional PACS or ELN systems via secure transfer protocols.
What objective magnifications are included by default?
Standard configuration includes 4×, 10×, and 20× phase contrast objectives; additional objectives require separate ordering and mechanical compatibility verification.
How is image calibration performed for quantitative measurements?
Calibration is factory-set using NIST-traceable stage micrometers; users may re-validate using the built-in calibration utility and reference slide—no physical tools required.