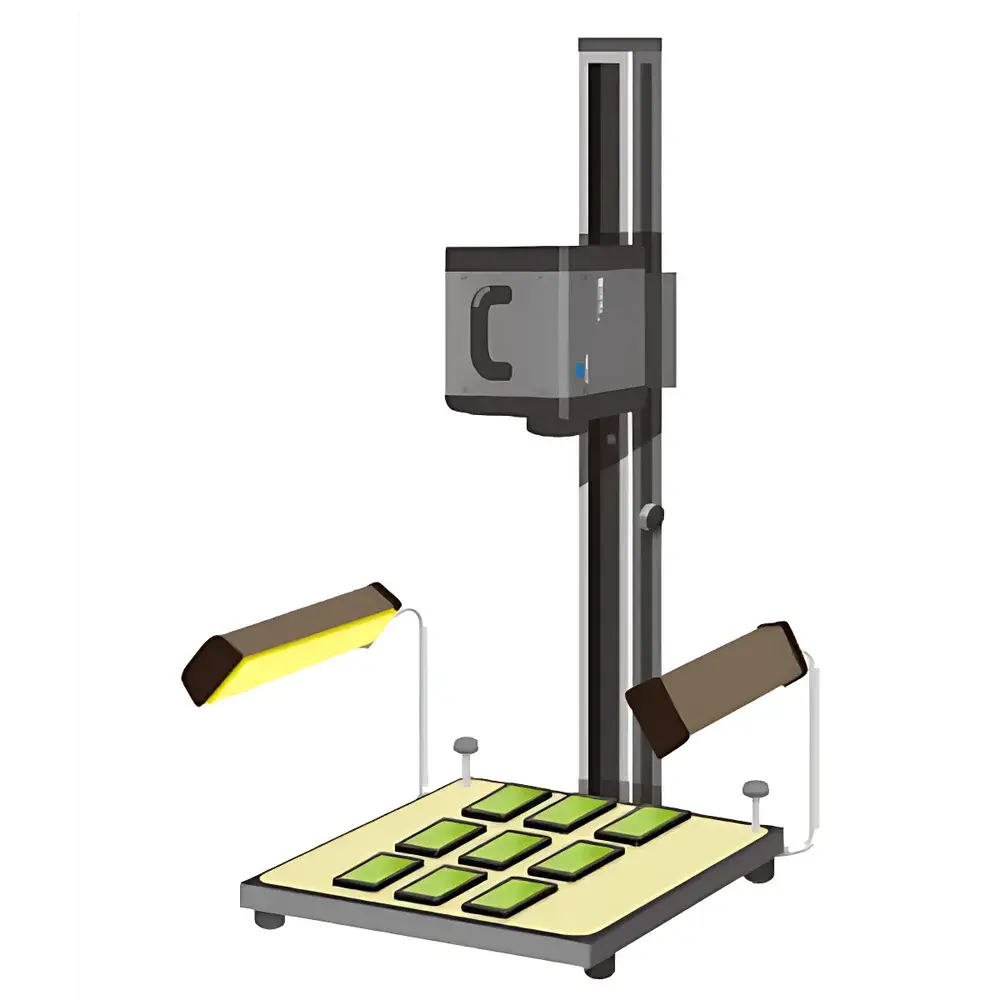
WD-A Pharmaceutical Stability Testing Chamber
| Origin | Tianjin, China |
|---|---|
| Manufacturer Type | Distributor |
| Origin Category | Domestic |
| Model | WD-A |
| Pricing | Upon Request |
| Temperature Range | 6–70 °C (±2 °C accuracy) |
| Humidity Range | 10–95 %RH (±4 %RH accuracy) |
| Illuminance Range | 0–7000 lx (adjustable, natural light reference included) |
| Chamber Dimensions | 510 × 400 × 380 mm (dual-tier stainless steel interior with removable partitions) |
| Construction | AISI 304 stainless steel chamber and shelves |
Overview
The WD-A Pharmaceutical Stability Testing Chamber is a purpose-built environmental simulation system engineered in strict alignment with the Guiding Principles for Stability Testing of Drugs outlined in the Chinese Pharmacopoeia (ChP). It enables controlled, time-resolved assessment of active pharmaceutical ingredients (APIs) and finished dosage forms under defined combinations of temperature, relative humidity, and photostress—key stressors specified in ICH Q1A(R2), Q1B, and Q5C guidelines. Unlike generic climate chambers, the WD-A integrates calibrated, independently monitored thermal, hygric, and photometric subsystems within a single compact footprint, supporting long-term stability studies (e.g., 25 °C/60 %RH, 30 °C/65 %RH), accelerated testing (40 °C/75 %RH), and photostability protocols per ICH Q1B Option 1 (near-UV and visible light exposure). Its design prioritizes spatial uniformity, sensor traceability, and operational repeatability—critical attributes for generating data suitable for regulatory submissions to NMPA, FDA, EMA, and PMDA.
Key Features
- Triple-parameter independent control: Precision PID-regulated heating/cooling, ultrasonic humidification/dehumidification, and digitally dimmable LED-based illumination array (0–7000 lx, spectral output compliant with ISO 10977:1993 for near-UV and visible bands)
- High-fidelity environmental monitoring: Dual-channel Pt100 RTD sensors for temperature (±0.3 °C probe accuracy), capacitive RH sensors (traceable to NIST standards, ±2.5 %RH typical), and cosine-corrected silicon photodiode lux meters (±3 % linearity)
- Stainless steel chamber architecture: Entire test volume—including walls, floor, ceiling, and two removable adjustable shelves—constructed from electropolished AISI 304 stainless steel (Ra ≤ 0.8 µm) to ensure corrosion resistance, cleanability, and compliance with GMP Annex 1 surface requirements
- Thermal and humidity uniformity: Validated chamber performance demonstrates ≤ ±1.5 °C temperature deviation and ≤ ±3.5 %RH variation across all shelf positions at steady state (per ISO 16770:2017 test protocol)
- Real-time data logging: Internal SD card storage (16 GB) with timestamped records at user-defined intervals (1–60 min); optional Ethernet/RS485 interface for integration into facility SCADA or LIMS
Sample Compatibility & Compliance
The WD-A accommodates standard stability sample configurations—including open trays, amber glass vials, blister packs, and secondary packaging—across its dual-tier internal volume (510 × 400 × 380 mm). Shelf spacing is fully adjustable to optimize airflow and irradiance distribution. All materials in contact with samples comply with USP Plastic Packaging Systems for Pharmaceutical Use (USP , ) and EU Ph. Eur. 3.1.11. The instrument supports full qualification per ASTM E2500-13 and IQ/OQ/PQ documentation templates. Environmental profiles are programmable to meet ICH-aligned storage conditions (e.g., “Long-Term”, “Intermediate”, “Accelerated”, “Photostability”), and audit trails—including parameter changes, door openings, and alarm events—are retained for ≥12 months to satisfy FDA 21 CFR Part 11 and EU Annex 11 requirements.
Software & Data Management
The embedded controller features a 7-inch resistive touchscreen with multilingual UI (English, Chinese, Spanish). It supports up to 16 independent test programs, each with up to 8 segmented ramp/soak steps. Data export is available via USB flash drive in CSV format, compatible with statistical analysis tools (e.g., JMP, Minitab) and stability software platforms (e.g., Tessella Stability Manager, LabVantage). Optional PC-based software provides remote monitoring, automated report generation (including trend charts, deviation alerts, and summary tables per ICH Q5C), and electronic signature capability aligned with ALCOA+ principles. All firmware updates are digitally signed and version-controlled to ensure integrity during GxP operations.
Applications
- ICH-compliant stability protocol execution for registration batches and commercial release
- Excipient compatibility screening under combined thermal-hygrometric stress
- Photodegradation kinetics studies for light-sensitive compounds (e.g., nitrofurantoin, riboflavin, chlorpromazine)
- Shelf-life estimation using Arrhenius modeling and real-time degradation curve fitting
- Package integrity evaluation under cyclic humidity conditions (e.g., 25 °C ↔ 40 °C / 60 %RH ↔ 75 %RH)
- Supporting quality-by-design (QbD) initiatives through DoE-based stability mapping
FAQ
Is the WD-A validated for GMP environments?
Yes—the system includes factory IQ/OQ documentation and supports site-specific PQ execution per Annex 15. Sensor calibration certificates are provided with NIST-traceable references.
Can the illuminance profile be customized to match ICH Q1B Option 2 (cool white fluorescent + near-UV)?
The built-in LED array allows independent intensity control of visible (400–700 nm) and near-UV (320–400 nm) channels; spectral power distribution reports are supplied upon request.
Does the chamber support automated data backup to network drives?
Via optional Ethernet module, it supports SFTP upload to designated servers at configurable intervals, with TLS 1.2 encryption enabled.
What is the maximum allowable load per shelf without compromising uniformity?
Maximum recommended load is 15 kg per shelf (distributed evenly); exceeding this may affect airflow dynamics and require requalification.
Are replacement sensors and calibration kits available directly from the manufacturer?
Yes—temperature, humidity, and lux sensors are field-replaceable; annual calibration kits (including certified reference standards) are stocked and shipped globally.