LUM GmbH LUMiFlector Multi-Reflection Spectroscopy (MRS) Analyzer for Milk Fat and Protein Quantification
| Brand | LUM GmbH |
|---|---|
| Origin | Germany |
| Model | LUMiFlector |
| Application Principle | Near-Infrared and UV-Visible Multi-Reflection Spectroscopy (MRS®) |
| Measurement Time | <20 s (lab), <8 s (inline) |
| Sample Handling | Non-contact, no sample preparation, no reagents |
| Output Parameters | Up to 4 simultaneous quantitative attributes (e.g., fat, protein, dry matter, total solids) |
| Calibration | Product-specific multivariate calibration models |
| Interface | 4× 4–20 mA analog I/O, 4× 24 V digital outputs, VARINLINE® process housing (inline version) |
| Compliance Context | Designed for GLP/GMP-aligned QA/QC environments |
Overview
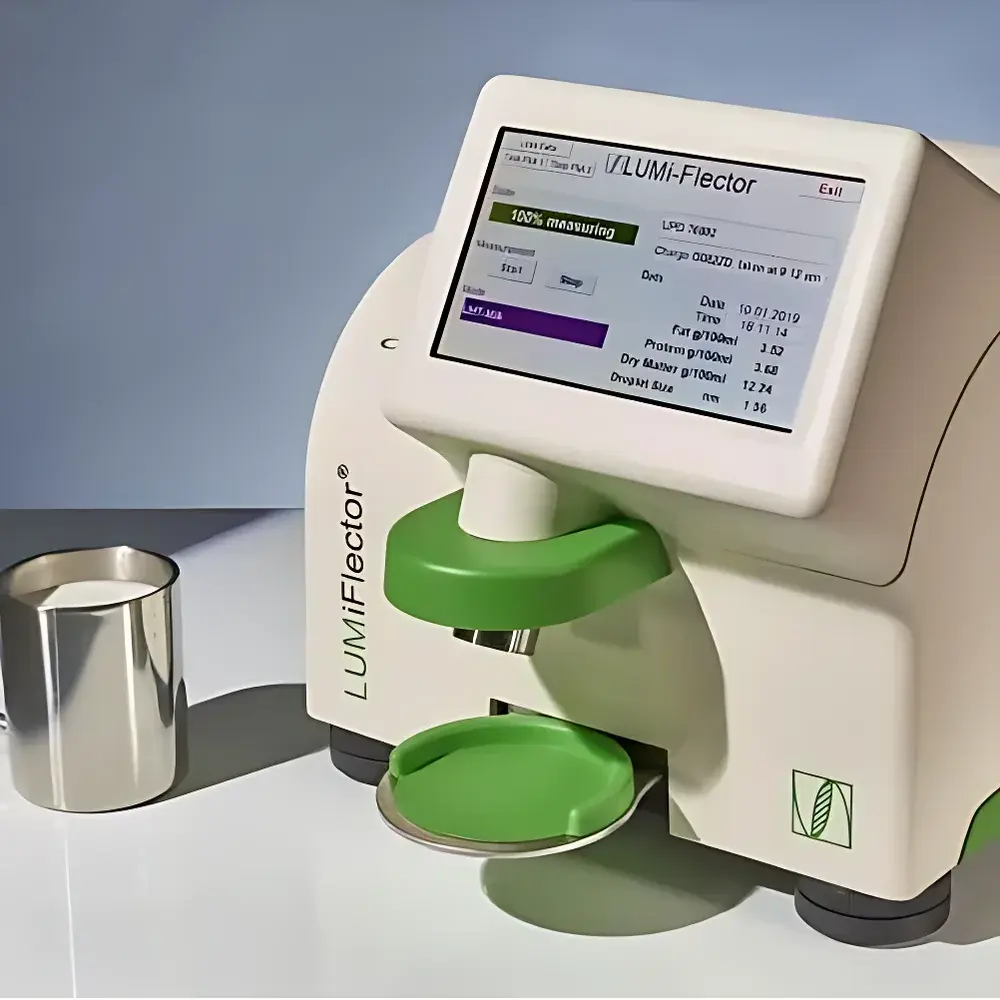
The LUM GmbH LUMiFlector is a benchtop and inline multi-reflection spectroscopy (MRS®) analyzer engineered for rapid, non-invasive quantification of compositional parameters in liquid and semi-liquid food matrices—particularly milk, dairy formulations, enteral nutrition products, feed suspensions, and biotechnological dispersions. Unlike conventional wet-chemistry or chromatographic methods, the LUMiFlector operates on the physical principle of multi-angle, broadband optical interrogation (spanning UV to NIR wavelengths), capturing ~100 independent intensity signals generated by scattering, reflection, and fluorescence interactions within the sample. These spectral responses are translated into quantitative values—including fat content, protein concentration, dry matter, total solids, and, in select calibrated applications, droplet size distribution—via robust, product-specific partial least squares (PLS) or principal component regression (PCR) models. The instrument delivers laboratory-grade precision without consumables, solvents, or sample dilution, making it suitable for both routine QC screening and real-time process monitoring in regulated food manufacturing environments.
Key Features
- Non-contact optical measurement: No sensor immersion, no cross-contamination, no wear-related drift
- Zero-reagent operation: Eliminates chemical procurement, hazardous waste disposal, and associated regulatory documentation
- Sub-20-second analysis cycle (lab mode); sub-8-second response (inline configuration)
- Simultaneous output of up to four independent compositional parameters per measurement
- Pre-loaded method library supporting up to eight distinct product types with auto-switching calibration
- Modular design: Identical core optics and algorithms across lab and inline versions—ensuring method transferability
- VARINLINE®-compliant process interface (inline model): Standardized sanitary housing with tri-clamp, flange, or threaded connections; compatible with 15–150 mm pipe diameters
- Industrial-grade I/O: Four isolated 4–20 mA analog inputs/outputs and four 24 V digital outputs for PLC integration, SCADA interfacing, or automated feedback control
Sample Compatibility & Compliance
The LUMiFlector is validated for use with opaque, turbid, and highly scattering media—including raw and pasteurized milk, whey protein concentrates, soy-based infant formulas, yeast suspensions, starch slurries, tomato paste, hot sauce emulsions, and pharmaceutical nutraceutical suspensions. Its optical architecture tolerates particulate loadings up to 60% w/w and viscosities exceeding 10,000 mPa·s without signal saturation or flow dependency (in inline mode). From a regulatory standpoint, the system supports quality systems compliant with ISO 22000, IFS Food, and BRCGS standards. When deployed with validated firmware and electronic record-keeping software, it meets foundational requirements for data integrity under FDA 21 CFR Part 11—including user authentication, audit trails, and electronic signature capability. While the hardware itself does not carry CE marking for medical devices, its application in nutritional product release testing aligns with EU Regulation (EU) No 609/2013 on foods for specific groups.
Software & Data Management
Instrument control, calibration management, and result visualization are handled via LUM’s proprietary LUMiSoft platform—a Windows-based application supporting method creation, spectral preprocessing (baseline correction, scatter normalization), PLS model development, and performance validation (RMSEP, bias, R²). All calibrations are stored as encrypted binary files with embedded metadata (date, operator ID, reference method traceability). Raw spectra and processed results are exportable in CSV, XML, or ASTM E1382-compliant formats. For enterprise integration, OPC UA and Modbus TCP drivers enable bidirectional communication with MES and LIMS platforms. Audit trail logs capture every parameter change, calibration update, or user login event—retained for ≥36 months unless manually purged under documented SOPs. No cloud dependency: all processing occurs locally, satisfying data sovereignty requirements in EU and APAC jurisdictions.
Applications
- Milk standardization: Real-time adjustment of fat/protein ratios during blending prior to UHT treatment
- QC release testing: Verification of nutritional label claims for protein, fat, and solids in ready-to-drink clinical nutrition products
- Feed formulation control: Monitoring consistency of soybean meal suspensions or molasses-based premixes
- Process optimization: Detecting emulsion breakdown or phase separation onset in salad dressings or dairy alternatives
- R&D screening: High-throughput evaluation of homogenization efficiency across shear rate gradients
- Stability assessment: Tracking time-dependent changes in backscattering profiles indicative of creaming or sedimentation kinetics
- Regulatory compliance: Supporting USP <1210> and ISO 17025 method validation protocols for NIR-based fat/protein assays
FAQ
Does the LUMiFlector require daily recalibration?
No. Once a product-specific calibration is validated per ASTM E1655 or ISO 12099, routine verification is performed using control samples at defined intervals (e.g., per shift or batch). Drift compensation is embedded in the MRS algorithm.
Can it measure particle size distribution?
Yes—when calibrated against orthogonal techniques (e.g., laser diffraction or centrifugal sedimentation), the LUMiFlector can report mean droplet diameter (d₄,₃) and polydispersity index for oil-in-water emulsions with R² > 0.92 versus reference methods.
Is method transfer between lab and inline units straightforward?
Yes. Identical optical pathlength, detector geometry, and spectral preprocessing ensure <±0.2% absolute deviation in fat measurements between configurations—provided identical calibration models and temperature stabilization are applied.
What maintenance is required?
Annual verification of optical alignment and LED source intensity; quarterly cleaning of quartz window with deionized water and lint-free wipes. No consumables, lasers, or moving parts require replacement.
How is compliance with food safety standards documented?
LUM provides a comprehensive Validation Support Package—including IQ/OQ templates, traceable reference material certificates, and a risk-based verification protocol aligned with ICH Q2(R2) and AOAC guidelines.