Shimadzu LabSolutions LIRS SDK
| Brand | Shimadzu |
|---|---|
| Origin | Japan |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Product Category | Imported Software |
| Model | LabSolutions LIRS SDK |
| Software Type | Chromatography Data System (CDS) Integration Toolkit |
| Development Platforms | .NET Framework, Java SE |
| Communication Mode | Synchronous & Asynchronous |
| Integration Scope | Bidirectional and Unidirectional API-based Interfacing with LabSolutions CS |
| Compliance Support | 21 CFR Part 11 Ready (Audit Trail, Electronic Signature, User Authentication), GLP/GMP-Aligned Architecture |
Overview
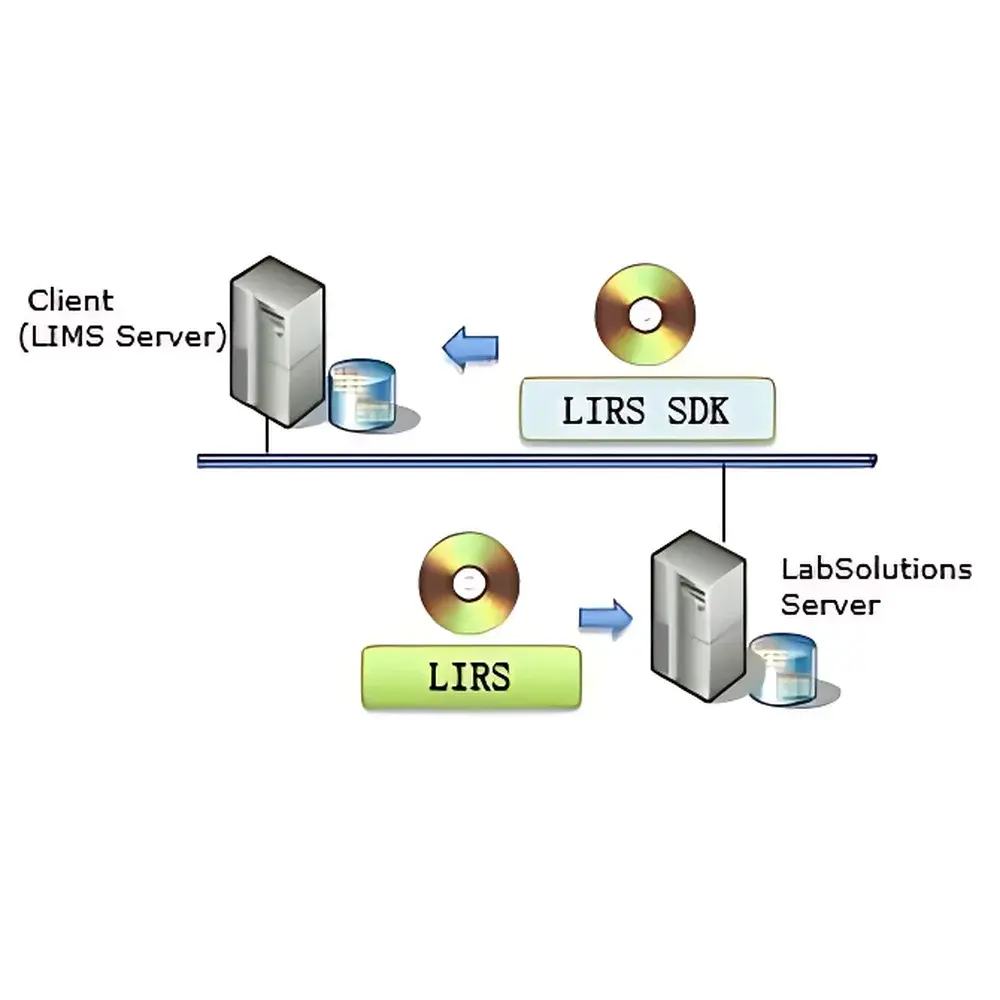
The Shimadzu LabSolutions LIRS SDK (LabSolutions Integration Remote Service Software Development Kit) is a production-grade integration toolkit engineered for seamless, standards-compliant interoperability between Shimadzu’s LabSolutions Chromatography Data System (CDS) and enterprise laboratory informatics platforms—including Laboratory Information Management Systems (LIMS), Electronic Lab Notebooks (ELN), and custom-built workflow orchestration applications. Built upon a service-oriented architecture, the LIRS SDK abstracts low-level CDS communication protocols into well-defined, domain-specific interfaces that mirror core analytical workflows: acquisition, processing, reporting, administration, and generic system interaction. It does not replace or modify LabSolutions CS; rather, it acts as a certified, vendor-supported bridge enabling programmatic access to batch management, method invocation, raw data retrieval, processed result ingestion, and report generation—without requiring direct database access or UI automation. This architecture ensures deterministic behavior, repeatable execution, and full traceability—critical prerequisites for regulated environments governed by FDA 21 CFR Part 11, ISO/IEC 17025, and ICH-GCP.
Key Features
- Five Modular Interface Components: Structured around chromatographic workflow phases—Generic (server/user configuration, authentication, system status), Acquisition (batch definition, sequence upload, instrument control initiation), Processing (data file download/upload, peak integration parameter application, reprocessing triggers), Reporting (batch summary extraction, sample-level report generation in PDF/CSV/XML), and Administration (role-based permission queries, audit log access, license validation).
- Dual-Platform Native Support: Full API surface available for both .NET Framework (C#/.NET Core-compatible) and Java SE (JDK 8+), enabling integration with heterogeneous IT infrastructures without middleware abstraction layers.
- Synchronous & Asynchronous Operation Modes: Supports blocking calls for deterministic batch submission and status polling, as well as event-driven callback registration for real-time notification of processing completion or error conditions—optimizing resource utilization in high-throughput labs.
- Built-in Data Integrity Safeguards: Enforces user context propagation, session-bound credential validation, and immutable transaction logging. All data transfers occur via encrypted LIRS service endpoints—not direct filesystem or database access—ensuring ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate) are preserved end-to-end.
- Zero-Touch Background Execution: Enables scheduled or event-triggered automation (e.g., post-run data export to LIMS, auto-report distribution) without manual intervention, reducing human error and supporting unattended operation during off-hours.
Sample Compatibility & Compliance
The LIRS SDK operates exclusively with Shimadzu LabSolutions CS v5.95 or later, deployed on Windows Server 2016/2019/2022 or Windows 10/11 (64-bit). It supports all Shimadzu GC, LC, and GC-MS systems controlled through LabSolutions CS. No hardware drivers or firmware modifications are required. From a regulatory standpoint, the SDK is designed to support compliance with FDA 21 CFR Part 11 (electronic records/signatures), EU Annex 11 (computerized systems), and ISO/IEC 17025:2017 Clause 7.7 (result reporting integrity). Audit trail data—including timestamped API call metadata, user identity, input parameters, and response codes—is persistently stored within the LabSolutions server environment and accessible via administrative interfaces for internal audit or regulatory inspection.
Software & Data Management
Integration projects using the LIRS SDK follow a documented development lifecycle: SDK installation (via Shimadzu-provided installer), LIRS Windows service configuration, certificate-based mutual TLS authentication setup, and interface binding to LabSolutions CS instances. All SDK methods return structured JSON or XML responses with standardized error codes (e.g., LIRS_ERR_BATCH_NOT_FOUND, LIRS_ERR_PERMISSION_DENIED), enabling robust exception handling in client applications. Versioned API contracts ensure backward compatibility across minor releases; breaking changes are communicated in Shimadzu’s official release notes and require explicit migration planning. Documentation includes comprehensive Javadoc and XMLDoc annotations, REST-like endpoint mappings, and sample code repositories for common use cases—such as automated QC release workflows or multi-instrument batch consolidation.
Applications
- Automated transfer of validated chromatographic batches from LabSolutions CS into LIMS for sample tracking, stability study management, and release decision support.
- Triggering reprocessing of legacy datasets under updated SOPs directly from ELN entries, with version-controlled parameter inheritance.
- Populating corporate analytics dashboards with real-time metrics—peak area RSD, retention time drift, system suitability pass/fail status—extracted programmatically per batch.
- Enabling centralized user provisioning and role synchronization between Active Directory and LabSolutions CS administrative permissions via periodic sync jobs.
- Supporting hybrid cloud-hybrid deployments where LabSolutions CS resides on-premises while LIMS or data lakes operate in Azure or AWS environments—leveraging secure outbound-only LIRS service connectivity.
FAQ
Does the LIRS SDK require LabSolutions CS to be installed on the same machine as the client application?
No. The SDK communicates over TCP/IP with the LIRS Windows service, which must reside on the same host as LabSolutions CS. Client applications may run remotely, provided network policies permit outbound connections to the LIRS service port.
Can the SDK initiate instrument hardware actions (e.g., start injection, open valve)?
No. Instrument control remains exclusively within LabSolutions CS. The SDK only manages software-level workflow orchestration—batch submission, data retrieval, and report generation.
Is source code provided with the SDK?
No. Shimadzu distributes compiled libraries (.dll for .NET, .jar for Java) along with header files, documentation, and sample projects. The underlying LIRS service binaries are proprietary and closed-source.
How is user authentication handled?
Authentication uses Windows Integrated Authentication (Kerberos/NTLM) or Shimadzu-defined username/password credentials configured in LabSolutions CS. All sessions inherit the security context of the authenticated user, enforcing granular access control at the API level.
What happens if LabSolutions CS is restarted during an active SDK session?
The LIRS service detects the CDS restart and automatically re-establishes its internal connection. Client applications receive appropriate disconnect/reconnect notifications and may resume operations after verifying session validity via the Generic interface’s health-check method.