Persee SA7/5 Series Hyphenated HPLC-AFS Speciation Analyzer
| Brand | Persee |
|---|---|
| Origin | Beijing, China |
| Manufacturer Type | Direct Manufacturer |
| Regional Classification | Domestic (China) |
| Model | SA7/5 |
| Instrument Type | Multi-channel Atomic Fluorescence Spectrometer |
| Sample Introduction | Pneumatic Flow Injection System (replaces peristaltic pump) |
| Waste Disposal | Gravity-driven dual-port drainage |
| Sample Introduction Mode | Vapor Generation / Hydride Generation |
| Gas Flow Control | Mass Flow Controller (MFC) |
| Detectable Elements | As, Hg, Se, Sb |
| RSD | ≤1.0% (at 0.01 ng/mL for As, Sb, Bi |
| Linear Dynamic Range | >3 orders of magnitude |
| Detection Limits | As/Sb/Bi ≤ 0.01 ng/mL |
| PMT Spectral Range | Hamamatsu R7154, 160–320 nm |
| Atomizer Temperature Control | Ambient to 100 °C, adjustable in 0.1 °C increments |
| UV Digestion Cell | Quartz plate-type flow-through reactor with high UV photon utilization |
| Dual-Channel Waste Separation | Organic and inorganic waste streams isolated via dedicated tubing paths |
| Certified Performance Data Source | Public Service Platform for Enterprise Standard Information (China) |
Overview
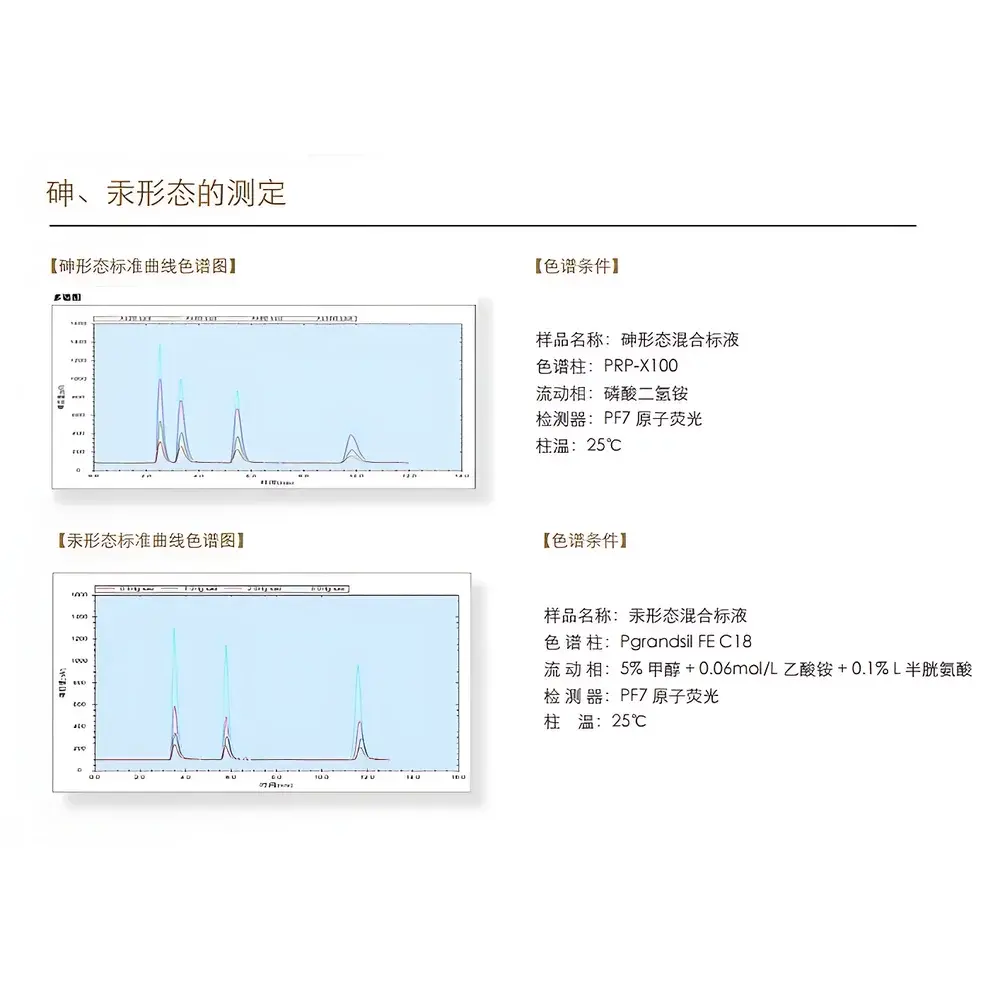
The Persee SA7/5 Series Hyphenated HPLC-AFS Speciation Analyzer is a purpose-built analytical platform engineered for quantitative elemental speciation analysis—specifically targeting arsenic (As), mercury (Hg), selenium (Se), and antimony (Sb) species in environmental, food safety, pharmaceutical, and clinical research laboratories. It integrates high-performance liquid chromatography (HPLC) with atomic fluorescence spectrometry (AFS) using a fully synchronized, hardware-coupled interface. Unlike conventional post-column AFS setups requiring manual reconfiguration, the SA7/5 features an embedded, factory-aligned speciation interface within the PF7-series AFS detector, enabling seamless transition between total-element quantification and species-resolved analysis without disassembly or recalibration. The system operates on hydride generation (HG) and cold vapor generation (CVG) principles, where target species are selectively converted into volatile hydrides (e.g., AsH₃, SbH₃) or atomic vapor (Hg⁰) prior to atomization in a quartz cell and fluorescence detection at characteristic wavelengths (193.7 nm for As, 253.7 nm for Hg, etc.). This hyphenated architecture ensures species integrity, minimizes interconversion artifacts, and delivers trace-level sensitivity with chromatographic resolution.
Key Features
- Pneumatic flow injection system with zero-wear components—eliminates peristaltic pump tubing fatigue, calibration drift, and maintenance downtime.
- Dual independent hydride generation systems: one optimized for total-element analysis (PF7 base configuration), another dedicated to speciation workflows—ensuring reaction kinetics, reagent stoichiometry, and gas–liquid separation parameters are precisely tuned for individual species (e.g., As(III) vs. As(V)).
- Integrated quartz plate-type UV digestion module operating inline between HPLC and AFS; provides uniform 254 nm irradiation across the full flow path, achieving >92% oxidative conversion efficiency for organoarsenicals (DMA, MMA) and methylmercury without carbon deposition or channel clogging.
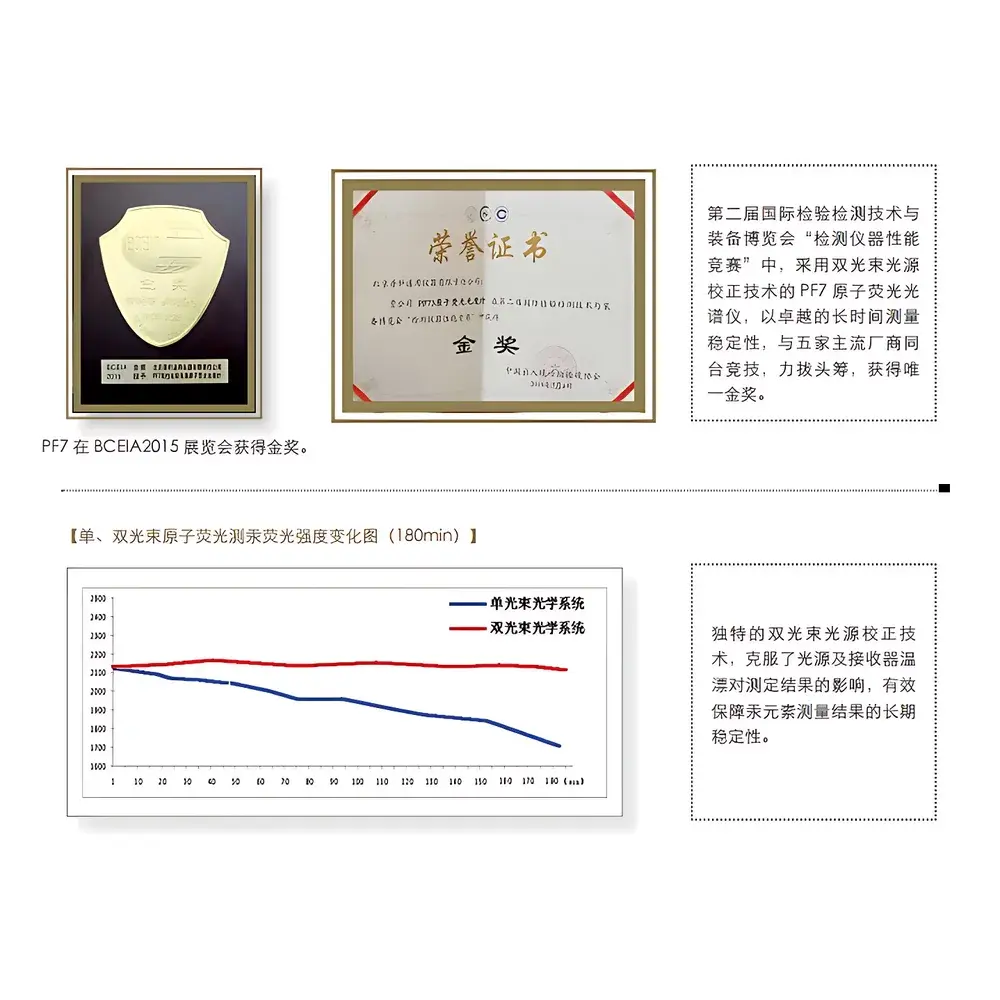
- Cryogenic gas–liquid separator with active cooling (−5 °C) and mechanical agitation—reduces water vapor load on the atomizer by >85%, suppresses baseline noise, and improves signal-to-noise ratio by up to 3.2× compared to ambient-phase separators.
- Mass flow controller (MFC)-regulated carrier gas delivery—maintains ±0.3% flow stability over 72 h, ensuring retention time reproducibility <0.2% RSD and peak area precision <1.0% RSD (n = 10).
- Dual-waste routing architecture: separate stainless-steel and PTFE-lined discharge lines segregate organic mobile phases (e.g., methanol/water gradients) from acidic/buffered aqueous waste—supporting compliance with ISO 14001 laboratory waste management protocols.
Sample Compatibility & Compliance
The SA7/5 supports aqueous extracts from solid matrices (soil, sediment, biological tissue), filtered drinking water, seafood homogenates, and pharmaceutical excipients. It accommodates standard C18, anion-exchange (SAX), and cation-exchange (SCX) columns (2–4.6 mm ID, 50–250 mm length) and is compatible with mobile phases containing up to 40% organic modifier (acetonitrile or methanol) and pH 2–9 buffers (e.g., ammonium acetate, phosphate). Method validation adheres to ICH Q2(R2) guidelines for specificity, linearity, accuracy, and precision. Instrument firmware and control software comply with FDA 21 CFR Part 11 requirements—including electronic signatures, audit trails, role-based access control, and immutable raw data archiving. All performance claims (DLs, RSDs, linear range) are verified per GB/T 27417–2017 (Guidelines for Validation of Analytical Methods in Chemical Testing) and referenced in registered enterprise standard Q/PRY 001–2023.
Software & Data Management
Persee SpeciationSuite v3.2 is a dedicated, single-interface application for method development, instrument control, peak integration, species identification (retention time matching + spike recovery), and quantitation (external calibration, standard addition, or isotope dilution where applicable). It supports ASTM D6724–22 (Standard Test Method for Determination of Arsenic Species in Water by HPLC-ICP-MS or HPLC-AFS) and USP / elemental impurities workflows. Raw chromatograms and fluorescence transients are stored in vendor-neutral .cdf format (NetCDF 4.8 compliant); processed reports export to PDF, CSV, or XML for LIMS integration. Audit trail logs record all parameter changes, user logins, calibration events, and data modifications with timestamps and operator IDs—fully traceable for GLP/GMP audits.
Applications
- Regulatory monitoring of inorganic arsenic (As(III)/As(V)) and methylated species (MMA, DMA) in rice, infant formula, and seaweed per EU Commission Regulation (EU) No 2023/915.
- Speciation of mercury in fish tissue—quantifying methylmercury (MeHg) vs. inorganic Hg(II) using reversed-phase HPLC coupled with CVG-AFS, meeting EPA Method 1630 requirements.
- Quality control of selenium-enriched supplements—differentiating selenomethionine, selenocysteine, and selenite via anion-exchange chromatography and HG-AFS detection.
- Environmental speciation in groundwater and wastewater—tracking redox-driven transformations of antimony species (Sb(III)/Sb(V)) under varying Eh/pH conditions.
- Method transfer support for laboratories transitioning from ICP-MS–based speciation to cost-optimized AFS platforms without sacrificing detection capability for priority elements.
FAQ
Does the SA7/5 require column oven integration for temperature-sensitive separations?
Yes—the system supports external column ovens (ambient to 60 °C) via analog voltage input; optional integrated oven modules (±0.1 °C stability) are available for methods requiring strict thermal control (e.g., AsB separation).
Can the instrument perform simultaneous multi-element speciation (e.g., As and Hg in one run)?
No—due to divergent optimal HG/CVG reaction conditions (acid concentration, KBH₄ strength, reaction coil temperature), sequential analysis is recommended. However, automated method-switching via SpeciationSuite allows unattended overnight runs across multiple elements.
Is the quartz UV digestion cell resistant to halogenated solvents?
The cell is rated for continuous exposure to ≤10% methanol, acetonitrile, or THF in aqueous buffer; higher organic loads require post-digestion dilution or solvent exchange via membrane interface to prevent quartz etching.
What documentation is provided for 21 CFR Part 11 compliance verification?
A complete validation package includes IQ/OQ/PQ protocols, electronic signature configuration guide, audit trail verification report, and raw data integrity test results—all pre-certified by Persee’s Quality Assurance Unit and aligned with Annex 11 (EU GMP) expectations.
How frequently must the PMT be recalibrated?
The Hamamatsu R7154 PMT is factory-calibrated and requires no routine recalibration; gain stability is verified during daily startup diagnostics. Full spectral response validation is recommended annually or after detector service.