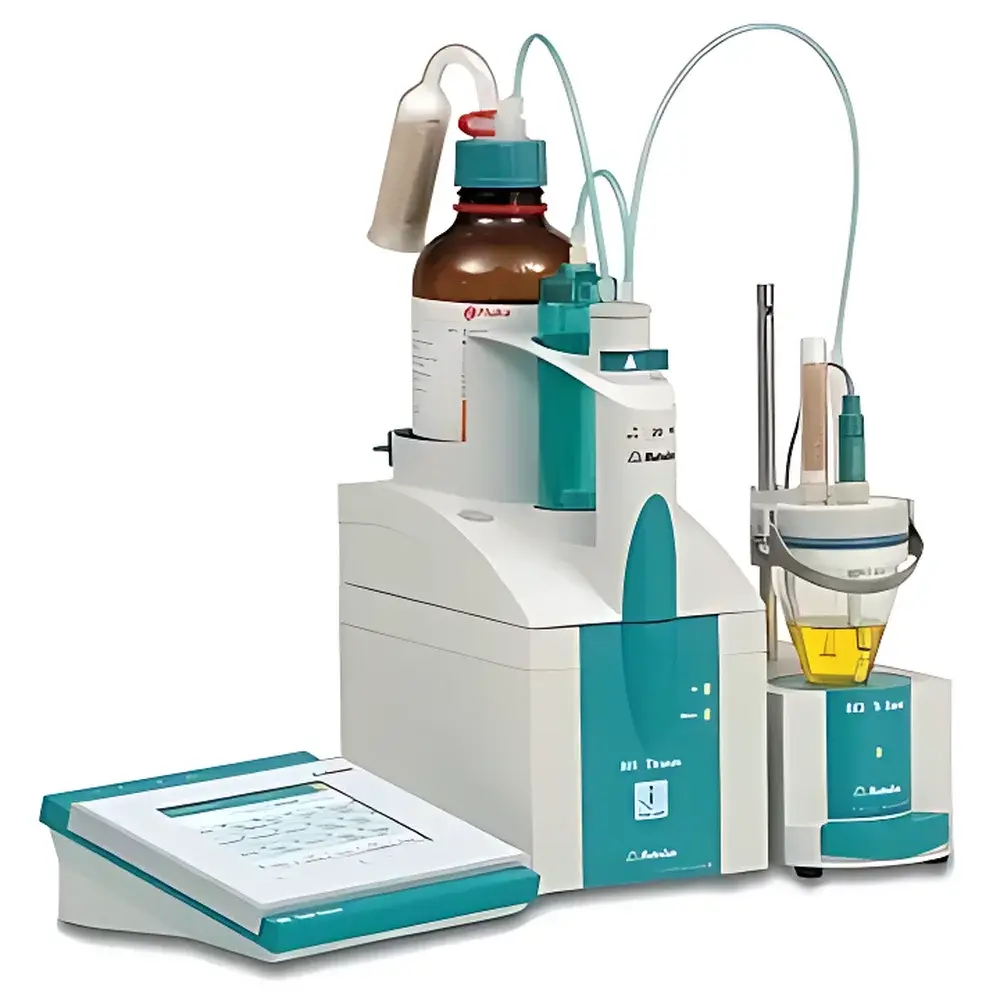
Metrohm 906 Titrando Automatic Potentiometric Titrator
| Brand | Metrohm |
|---|---|
| Origin | Switzerland |
| Manufacturer | Metrohm AG |
| Type | Universal Automatic Potentiometric Titrator |
| Measurement Range | ±1200 mV, ±120 µA |
| Electrode Reading Accuracy | 0.15% (±30 µL at 20 mL) |
| Resolution | 0.001 pH, 0.1 mV, 0.1 °C |
| Titration Accuracy | 0.15% (±30 µL at 20 mL) |
| Supported Titration Modes | DET (Dynamic Equivalence Point Detection), MET (Multi-Endpoint Titration), SET (Setpoint Titration), KF Coulometric & Volumetric Karl Fischer, STAT (Potentiostatic Titration) |
| Integrated Dosino Dosing Units | 1 × 806 Smart Exchange Unit (20,000-step resolution), expandable to up to 9 × 805 Dosing Units |
| Stirrer Support | Up to 4 × 801 Magnetic Stirrers or 4 × 804 Titration Stands with 802 Rod Stirrers or 803 KF Stands |
| Interface Ports | 4 × MSB (Metrohm Serial Bus), 1 × USB for Sample Processor/Peripherals, PCMCIA Slot for Method/Data Storage & User Authentication Cards, Auxiliary Module Slot (e.g., 867 pH Module or 856 Conductivity Module) |
| Temperature Sensors | Pt1000 or NTC |
| Optional Real-Time Curve Display | 90 mm × 120 mm Color Touchscreen or External PC Monitor |
| Data Compliance | FDA 21 CFR Part 11, GLP/GMP Audit Trail, Electronic Signatures, Password Protection |
Overview
The Metrohm 906 Titrando is a fully automated, modular potentiometric titrator engineered for precision, traceability, and interoperability in regulated analytical laboratories. It operates on the fundamental principle of potentiometric endpoint detection—measuring the potential difference between a reference electrode and an indicator electrode as titrant is incrementally added to the sample. This electrochemical signal is processed in real time to identify equivalence points using algorithmic evaluation methods including derivative-based DET (Dynamic Equivalence Point Tracking), multi-point MET (Multi-Endpoint Titration), and user-defined SET (Setpoint Titration). The system integrates seamlessly with volumetric Karl Fischer water determination (KF), potentiostatic (STAT) titration, and ion-selective electrode (ISE) measurements—enabling quantitative analysis of acidity, redox activity, halides, sulfides, cyanides, and other electroactive species across pharmaceutical, food & beverage, environmental, and chemical manufacturing applications.
Key Features
- Smart Hardware Architecture: All core components—including the 906 main unit, 806 Smart Exchange Unit, intelligent electrodes, and digital measurement cables—feature embedded microchips enabling automatic identification, calibration history logging, and firmware-aware communication via Metrohm’s proprietary MSB (Metrohm Serial Bus) protocol.
- High-Precision Liquid Handling: The integrated 806 exchange unit delivers dosing resolution of 1 part in 20,000, achieving ±30 µL absolute accuracy over a 20 mL titrant volume—validated per ISO 8655-3 for piston burettes.
- Modular Expansion Capability: Supports up to nine additional 805 dosing units, four magnetic stirrers (801), or four titration stands (804/803) with configurable stirring modes and temperature control interfaces.
- Dual-Display Operation: Real-time titration curves and method navigation are rendered on the built-in 90 × 120 mm color touchscreen or mirrored to a remote PC via tiamo™ software—ensuring ergonomic workflow continuity during method development and routine analysis.
- Regulatory-Ready Control System: Implements full audit trail functionality, electronic signatures, role-based access control, and password-protected method editing—all compliant with FDA 21 CFR Part 11, EU Annex 11, and ISO/IEC 17025 requirements.
Sample Compatibility & Compliance
The 906 Titrando accommodates aqueous and non-aqueous matrices, suspended solids (with optional filtration modules), viscous oils, and heterogeneous suspensions—provided appropriate electrode selection (e.g., double-junction Ag/AgCl, glass pH, or solid-contact ISEs) and stirring configuration. It supports standardized test procedures aligned with ASTM E203 (KF moisture), ISO 8587 (acid–base titration), EN 14104 (iodine value), and USP (potentiometric assay). All hardware and firmware are certified under CE, UL, and RoHS directives. Electrode performance verification—including slope, offset, and response time—is automated via GLP-compliant electrode testing routines embedded in tiamo™.
Software & Data Management
Control and data handling are centralized through tiamo™ v3.x (or later), a validated laboratory software platform supporting database-driven sample registration, parallel titration scheduling, statistical result reporting (mean, RSD, confidence intervals), and export to LIMS via ODBC or CSV. Methods are stored with version control, user attribution, and timestamped modification logs. Raw data—including mV/pH/T vs. volume traces, derivative plots, and equivalence point assignments—are archived in encrypted binary format (.TIA) with SHA-256 checksum integrity validation. Backup and restore operations utilize PCMCIA memory cards containing method templates, calibration certificates, and user authentication tokens.
Applications
- Pharmaceutical QC: Assay of active ingredients (e.g., amine hydrochlorides, organic acids) per USP monographs; residual solvent quantification via KF titration.
- Food & Agriculture: Total acidity (TA) in wines and juices; salt content (chloride) in dairy products; peroxide value in edible oils.
- Environmental Analysis: Alkalinity and hardness profiling in wastewater; cyanide and fluoride determination in drinking water per EPA Method 325 and ISO 10304-1.
- Chemical Manufacturing: Catalyst activity assessment (e.g., acid number in polyesters); purity verification of inorganic salts and surfactants.
- Research & Development: pKa determination via Gran plot analysis; redox stoichiometry studies using controlled-potential coulometric titration (STAT mode).
FAQ
Does the 906 Titrando support remote operation and network integration?
Yes—it features native LabLink connectivity for secure LAN/WAN deployment, enabling centralized instrument monitoring, method push-down, and data aggregation across multi-site laboratories.
Can the system validate electrode performance automatically?
Yes—GLP-compliant electrode diagnostics are executed prior to each batch run, logging slope (%), asymmetry potential (mV), and response time (s) with pass/fail criteria configurable per SOP.
Is tiamo™ software qualified for GxP environments?
Yes—tiamo™ is delivered with IQ/OQ documentation packages, 21 CFR Part 11 configuration files, and vendor-supplied validation support services for installation in FDA- and EMA-regulated facilities.
What types of external devices can be interfaced via USB or MSB?
Sample processors (e.g., 863 Compact Auto-sampler), analytical balances (via RS232-to-USB adapters), barcode scanners, and thermal printers (including Bluetooth-enabled models) are natively supported.
How is data integrity ensured during power loss or unexpected shutdown?
All titration sequences employ atomic transaction logging: incomplete runs are flagged, raw data buffers are preserved in non-volatile flash memory, and resume-on-restart functionality restores measurement state without data truncation.