Clinical Laboratory Instruments
Filter
Showing 61–87 of 87 results
| Origin | Imported |
|---|---|
| Manufacturer Type | OEM Supplier |
| Country of Origin | Lithuania |
| Model | MicrUs Pro |
| Spatial Resolution | 30 µm |
| Temporal Resolution | 7.5 ns |
| Gray Scale Levels | 16–256 |
| Time Gain Compensation (TGC) | Adjustable |
| Measurement Capabilities | Distance, Area, Perimeter, Angle, Volume, Stenosis Ratio, Time, Velocity, Heart Rate |
| Dynamic Range | 12 dB to 54 dB |
| Display Modes | B, B+B, 4B, B+M, M |
| Maximum Penetration Depth | 50 mm |
| Brand | Agela |
|---|---|
| Origin | Tianjin, China |
| Manufacturer Type | Authorized Distributor |
| Country of Origin | China |
| Model | MiracleBox |
| Pricing | Available Upon Request |
| Temperature Range | 0°C to Ambient |
| Temperature Accuracy | ±1°C |
| Thawing Time (Bovine Serum) | ≤20 min |
| Cooling-to-Setpoint Time (Bovine Serum) | ≤12 min |
| Sample Compatibility | Standard Racks, Tube Holders, 96-Well Collection Plates |
| Control Method | Dry-Programmed Heating/Cooling |
| Interface | Touchscreen with Real-Time Sample Temperature Display |
| Multi-Zone Temperature Control | Yes |
| Modular Integration Capability | Yes |
| Brand | Meiko Rikka |
|---|---|
| Origin | Japan |
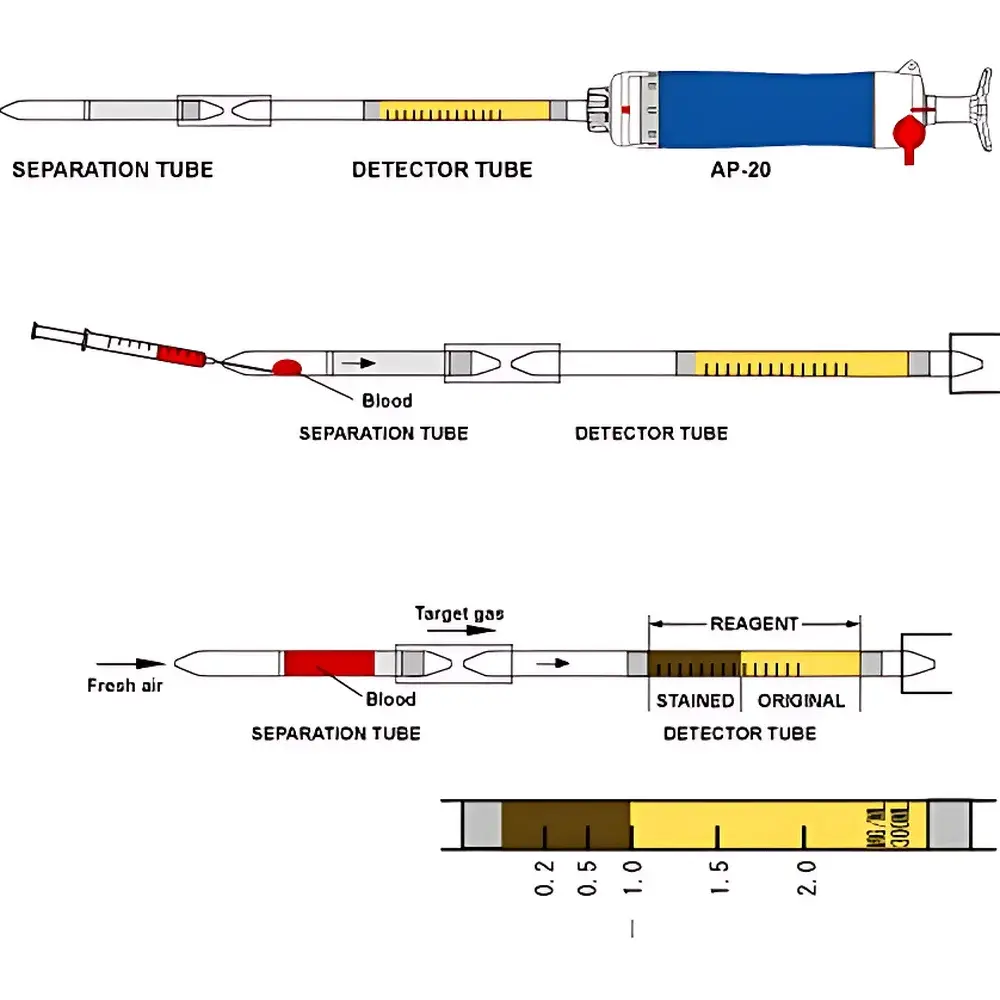
| Model Numbers | 290CN, 290EA, 290CO, 290H2S, 290PQ, 290OP, 290PA |
| Detection Time | ≤60 s per test |
| Sample Volume | 10 mL whole blood |
| Sample Type | Human peripheral venous or capillary blood |
| Loading Mode | Semi-automatic manual insertion |
| Detection Limits | Malathion ≥0.004% (w/v), Dichlorvos ≥0.008% (w/v), Glufosinate ≥0.02% (w/v), Glyphosate ≥0.02% (w/v) |
| Regulatory Compliance | Designed per Japanese National Police Agency Forensic Guidelines (NPA-FG-2018), ISO 15197:2013 principles for qualitative point-of-care toxicant screening |
| Brand | Meiko Rikka |
|---|---|
| Origin | Japan |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | 290CO |
| Pricing | Upon Request |
| Brand | Octadem |
|---|---|
| Origin | Jiangsu, China |
| Manufacturer | Octadem Biotech Co., Ltd. (Wuxi) |
| Model | ACR-2000 |
| Detection Principle | Transmission Turbidimetry & Photometric Quantification |
| Sample Throughput | 4 samples/12 min |
| Data Storage | Internal high-capacity memory with USB export |
| Calibration | Onboard QC calibration with traceable standards |
| Regulatory Compliance | Meets GB/T 26124–2010 (Clinical Chemistry Analyzers), aligned with CLSI EP15-A3 and ISO 15197:2013 for precision requirements |
| Intended Use | Quantitative determination of urinary albumin and creatinine to calculate UACR for early diabetic kidney disease (DKD) screening in primary care settings |
| Brand | Olympus |
|---|---|
| Country of Origin | Japan |
| Model | BF-1TQ290 |
| Insertion Tube Outer Diameter | 6.0 mm |
| Distal Tip Outer Diameter | 5.9 mm |
| Working Length | 600 mm |
| Field of View | 120° |
| Direction of View | Forward |
| Depth of Field | 2–100 mm |
| Instrument Channel ID | 3.0 mm |
| Minimum Focusing Distance | 3.0 mm from distal tip |
| Insertion Tube Rotation | ±120° |
| Compatible Light Source | Olympus CLV-290SL / CLV-290 |
| Compatible Video Processor | Olympus CV-290 |
| NBI (Narrow Band Imaging) | Yes |
| Range ID Function | Yes |
| Waterproof One-Touch Connector | Yes |
| High-Frequency Electrocautery Compatible | Yes |
| Laser Compatibility | Nd:YAG and 810 nm Diode Lasers |
| EBUS-Compatible | Yes |
| Reprocessing Compatibility | Fully compatible with Olympus ETD series cleaners/disinfectors and drying cabinets |
| Sterilization Compatibility | Ethylene Oxide (ETO) and Plasma Sterilization supported |
| Brand | Olympus |
|---|---|
| Country of Origin | Japan |
| Model | CV-170 + GIF-H170 + CF-H170L/i |
| Regulatory Clearance | China NMPA Import Registration No. 20152221796 (CV-170), 20143222972 (GIF-H170), 20143222973 (CF-H170L), 20143222945 (CF-H170i) |
| Power Input | 100–240 V AC, 50/60 Hz ±1 Hz |
| Dimensions (W×H×D) | 295 × 145 × 425 mm |
| Weight | 11.0 kg |
| Light Source | Integrated Long-Life LED |
| Video Output | HD-SDI, SD-SDI, DVI-D, RGB/YPbPr (1080i), Composite/Y/C (480i/576i) |
| Image Storage | MAJ-1925 Portable Memory (2 GB, TIFF/JPEG compression) |
| Compatibility | EVIS 100/130/140, OLYMPUS 150, EXERA 160, EXERA II 180, GI/BF/VISERA Series Endoscopes |
| Brand | OPTI |
|---|---|
| Origin | USA |
| Model | OPTI LION |
| Type | Dry-chemistry, Single-use Cartridge-based Electrolyte Analyzer |
| Measurement Parameters | pH, Na⁺, K⁺, Cl⁻, Ca²⁺ (including ionized calcium and standardized corrected calcium at pH 7.4) |
| Interface | Color Touchscreen with Multilingual Support (including English) |
| Barcode Scanner | Integrated |
| Quality Control | On-board QC with Statistical Analysis (Mean, SD, CV, Levey-Jennings charts) |
| Software | FDA 21 CFR Part 11–compliant firmware with Lifetime Free Updates |
| Regulatory Status | CE-IVD, FDA 510(k)-cleared |
| Compliance | Meets CLIA, ISO 15197, ISO 22870, and ASTM E2573 standards for point-of-care and core laboratory use |
| Brand | OPTI |
|---|---|
| Origin | USA |
| Model | OPTI R |
| Measurement Principles | Optical Fluorescence (pO₂), Severinghaus-type CO₂ Sensor (pCO₂), Potentiometric Ion-Selective Electrodes (pH, Na⁺, K⁺, Ca²⁺), Spectrophotometric Hemoglobin Detection (tHb, SO₂) |
| Sample Type | Whole blood (arterial, venous, capillary), heparinized |
| Test Time | ≤ 60 seconds per sample |
| Cartridge Capacity | 50 tests per single-use disposable cartridge |
| Onboard QC | Three-level automated quality control with integrated calibrators |
| Data Storage | 300 patient results + 60 days of QC records (3 levels) |
| Interface | Built-in barcode scanner, RS232/USB, LIS/HIS-compatible software |
| Display | 7-inch color touchscreen with intuitive graphical UI |
| Regulatory Compliance | FDA 510(k) cleared, CE IVD, ISO 13485 certified |
| Origin | Jiangsu, China |
|---|---|
| Manufacturer Type | Distributor |
| Origin Category | Domestic |
| Model | Paragon XHD |
| Price Range | USD 0–140,000 (FOB) |
| Spatial Resolution | 80–100 µm |
| Temporal Resolution | 30 fps |
| Gray Scale | 256 levels |
| Time-Gain Compensation (TGC) | 8 adjustable segments |
| Measurement Functions | Distance, Circumference, Area, Volume, Depth, Angle |
| Dynamic Range | 30–150 dB |
| Display Modes | B-mode + Color Doppler + Pulsed-Wave Doppler (B+C+PW) |
| Maximum Penetration Depth | 50 mm |
| Brand | Raymetech |
|---|---|
| Origin | Jiangsu, China |
| Model | MMS100 |
| Type | Fully Manual Mechanical Micromanipulator |
| Positioning Resolution | Sub-micrometer (typical manual fine-adjustment capability) |
| Mounting | Standard 36 mm dovetail or M4/M6 threaded base interface |
| Material | Anodized aluminum alloy with stainless steel precision screws |
| Load Capacity | ≤ 200 g (static, tip-mounted) |
| Degrees of Freedom | 3-axis orthogonal translation (X/Y/Z) with coarse + fine dual-scale control |
| Repeatability | ≤ ±1 µm (under stable environmental conditions) |
| Brand | SCChengyi |
|---|---|
| Origin | Sichuan, China |
| Model | RM6240T |
| Manufacturer Type | Authorized Distributor |
| Regional Classification | Domestic (China) |
| Pricing | Available Upon Request |
| Brand | Roche |
|---|---|
| Origin | Switzerland |
| Model | Cedex Bio / Cedex Bio HT |
| Principle | Continuous Flow (Channel-Based) Analysis |
| Reagents | Roche-Approved Only |
| Sample Type | Cell Culture Supernatant |
| Analyte Scope | Substrates, Metabolites, Products |
| Throughput | High (Cedex Bio HT), Standard (Cedex Bio) |
| Regulatory Context | Designed for GMP-aligned bioprocess monitoring and QC release testing |
| Brand | RWD |
|---|---|
| Origin | Guangdong, China |
| Manufacturer Type | Direct Manufacturer |
| Region Category | Domestic (China) |
| Model | MP-500 |
| Pricing | Upon Request |
| Brand | Selecta |
|---|---|
| Origin | Spain |
| Model | PhotometerS-2000 |
| Measurement Principle | Continuous Flow (Flow-Through Cuvette System) |
| Detection Reagents | Selecta-branded clinical reagent kits |
| Optical System | Dual-wavelength solid-state photodiode detector with 7 fixed interference filters (340, 405, 500, 546, 578, 620, 670 nm) |
| Bandwidth | 10 nm |
| Cuvette Volume | 30 µL |
| Absorbance Range | 0.000–2.500 ABS |
| Display Resolution | 0.001 ABS |
| Computational Resolution | 0.0001 ABS |
| Temperature Control | Peltier-heated sample holder (setpoints: 25 °C, 30 °C, 37 °C |
| Light Source | 6 V / 10 W tungsten-halogen lamp |
| Lamp Life-Saving Mode | Yes |
| Display | 640 × 240 pixel LCD, 256-color graphics |
| Data Interface | RS-232 serial port |
| Ambient Operating Conditions | 15–32 °C, ≤85% RH (non-condensing) |
| Internal Storage | Up to 134 assay methods, 1000 patient results |
| Preloaded Methods | 47 most common clinical assays (e.g., cholesterol, glucose, serum iron, LDH, AST, urea) |
| Curve Fitting Algorithms | Endpoint, Kinetic, Linear Regression, Multi-Point Calibration, Best-Fit Polynomial, Bichromatic Correction |
| Control Interface | Scroll wheel or external USB mouse |
| Dimensions (H×W×D) | 41.2 × 45.3 × 33 cm |
| Power Supply | 100–220 V AC, 50–60 Hz |
| Weight | 15 kg |
| Optional Accessory | BIO-BATH metal dry-block incubator (37 °C, 8-position, for 75 × 13 mm test tubes) |
| Origin | USA |
|---|---|
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | SN-01 |
| Pricing | Upon Request |
| Brand | SOOHOW INSTRUMENT |
|---|---|
| Origin | Jiangsu, China |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Product Category | Domestic |
| Model | STS-A200 |
| Pricing Range | USD 14,000–21,000 |
| Architecture | Discrete Analytical System |
| Instrument Class | Low-Speed Fully Automated (0–400 rpm equivalent throughput logic) |
| Sample Types | Whole Blood, Serum, Plasma, Urine, Cerebrospinal Fluid (CSF) |
| Analyte Class | Specific Proteins (e.g., CRP, IgG, IgA, IgM, C3, C4, ASO, RF, HbA1c, etc.) |
| Throughput | 180 Tests per Hour |
| Optical Principle | Rate Nephelometry and/or Turbidimetry |
| Light Source | 12 V Long-Life Halogen Lamp |
| Wavelength Range | 340–700 nm (8 Fixed Wavelengths) |
| Reaction Temperature Control | 37.0 ± 0.1 °C via Solid-State Direct-Heating with Full-Circumference Thermal Sensors |
| Reaction Vessels | 40 UV-Transparent Cuvettes |
| Sample Capacity | 25 Tubes on 5-Position Rack-Based Continuous Feed |
| Dispensing Accuracy | ±5% at 3–30 µL (Sample), ±5% at 20–200 µL (Reagent) |
| Precision (CV) | ≤5% |
| Carryover | <1% |
| Stability (4h/8h) | Deviation (B) ≤ ±5% vs. Calibrator Assigned Values |
| Compliance | GB/T 14710–2009 (Environmental & Mechanical Robustness), GB 4793.1–2007 / GB 4793.9–2013 / YY 0648–2008 (Electrical Safety), GB/T 18268.1–2010 / GB/T 18268.26–2010 (EMC) |
| Brand | Sutter |
|---|---|
| Origin | USA |
| Model | MM-33 |
| Control Type | Manual |
| Coarse Adjustment Range (X/Y/Z) | 37 mm / 20 mm / 20 mm |
| Coarse Resolution | 0.1 mm per axis |
| Fine Adjustment Range (X) | 10 mm |
| Fine Resolution | 0.01 mm |
| Rotational Base | Lockable, Dual-Axis (Horizontal & Vertical) |
| Mounting Compatibility | Magnetic base, standard dovetail or clamp-style stage adapters |
| Intended Use | Electrophysiology, patch-clamp, microinjection, and extracellular stimulation |
| Brand | TECAN |
|---|---|
| Origin | Switzerland |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | ADME |
| Pricing | Available Upon Request |
| Brand | Terumo BCT |
|---|---|
| Origin | USA |
| Model | Spectra Optia® |
| Device Type | Automated Blood Separation and Cell Processing Platform |
| Regulatory Classification | FDA-cleared Class II Medical Device (510(k) K122936, K151487, etc.) |
| Weight | 90 kg |
| Extracorporeal Volume | ≥110 mL |
| Patient Weight Range | 2–227 kg |
| Data Storage Capacity | 100 patient sessions |
| Integrated Tube Sealer | Yes |
| Power Failure Memory | Yes |
| User Interface | Color Touchscreen GUI with Multilingual Support (including Simplified Chinese) |
| Key | Brand: Thermal Technology |
|---|---|
| Model | NTE-2A |
| Temperature Range | 0–50 °C |
| Control Accuracy | ±0.1 °C |
| Resolution | 0.1 °C |
| Probe Tip Diameter | 13 mm |
| Probe Length | 16.5 cm |
| Response Time (Heating/Cooling) | <4 s |
| Base Temperature Adjustment Range | 20–30 °C |
| Input Power | 200–240 V AC, 50 Hz, 100 W |
| Dimensions | 20.3 × 17.8 × 38.1 cm |
| Weight | 12.7 kg |
| Compliance | ASTM F2696 (Standard Guide for Quantitative Sensory Testing), ISO 13485 (Medical Devices), FDA 21 CFR Part 11 (Software Validation Ready) |
| Brand | URIT |
|---|---|
| Model | UC-A580Vet |
| Origin | Guangxi, China |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Regional Classification | Domestic (China) |
| Pricing | Available Upon Request |
| Brand | URIT |
|---|---|
| Origin | Guangxi, China |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Country of Origin | China |
| Model | URIT-1280 + URIT-1600 |
| Pricing | Upon Request |
| Brand | Wasatch Photonics |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Product Origin | Imported |
| Model | Cobra OCT Spectrometer |
| Pricing | Available Upon Request |
| Brand | YUYANBIO |
|---|---|
| Origin | USA |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Product Category | Imported Medical Device |
| Model | e-Celsius® Human Core Temperature Telemetry System |
| Pricing | Available Upon Request |
| Brand | YUYANBIO |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Direct Manufacturer |
| Country of Origin | China |
| Model | YAN-105A |
| Price Range | USD 4,200 – 12,600 (FOB) |
| Throughput | 100 samples/hour |
| Sample Volume | 17 µL (whole blood mode) |
| Sample Type | Whole blood (native or pre-diluted) |
| Loading Mode | Fully automated aspiration and processing |
| Detection Principle | Coulter impedance + non-cyanide colorimetric hemoglobin assay |
| Output | 3-parameter histogram (WBC, RBC, PLT), integrated thermal printer, 50,000-sample onboard storage |
| Brand | YUYANBIO |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Direct Manufacturer |
| Country of Origin | China |
| Model | YAN-505A |
| Testing Speed | 100 samples/hour |
| Sample Volume | 10 µL (whole blood, pre-diluted mode) |
| Sample Type | Whole Blood |
| Loading Mode | Automatic |
| White Blood Cell Differential Method | Multi-angle semiconductor laser scattering flow cytometry |
| Output Graphics | 3 histograms + 4 scatter plots (including dedicated BASO scatter plot) |
| Internal Storage Capacity | Up to 50,000 complete test records with graphical data |
| Display | 10.4-inch full-color capacitive touchscreen |
| LIS Connectivity | Bidirectional wireless LIS interface compliant with ASTM E1384 and HL7 v2.x |
| Regulatory Compliance | Designed for GLP-compliant preclinical research environments |
| Optional Module | Reticulocyte detection (YAN-505A variant only) |