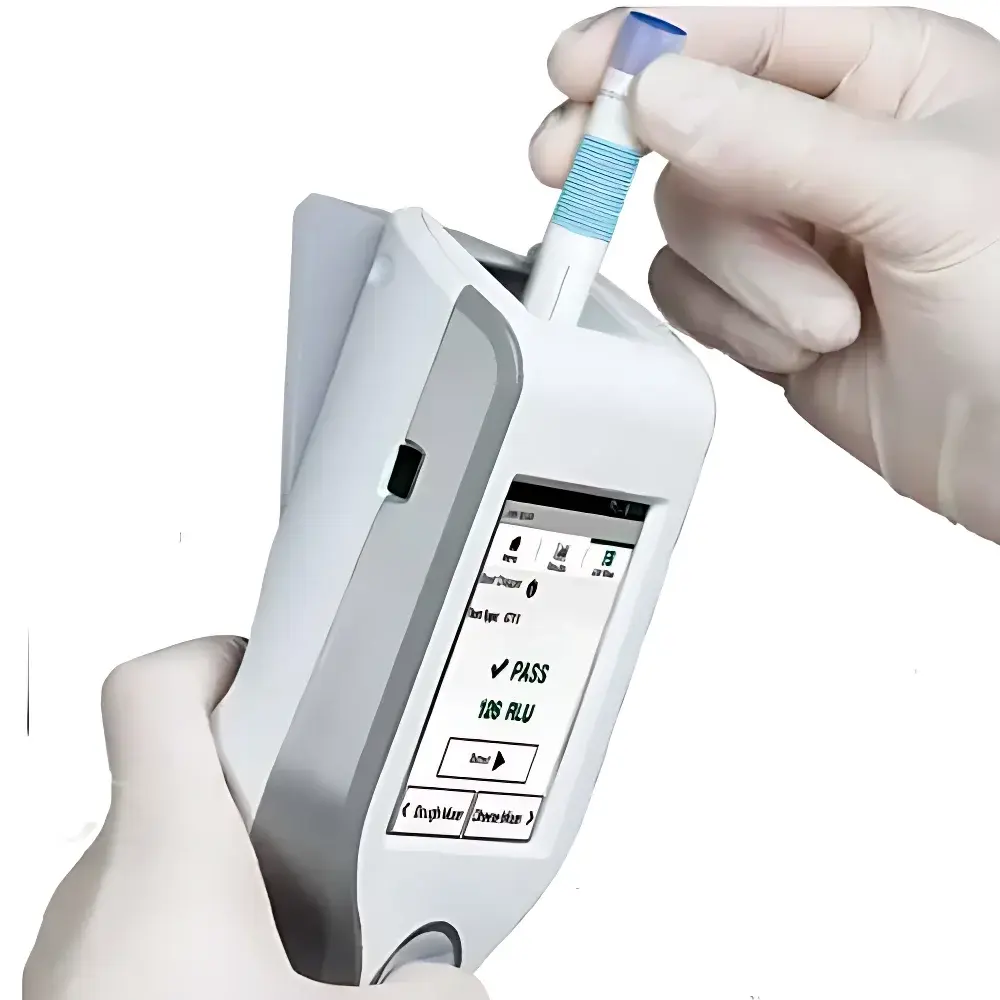
3M Clean-Trace ATP LM1 Handheld Bioluminescence Luminometer
| Brand | 3M |
|---|---|
| Origin | USA |
| Model | Clean-Trace ATP LM1 |
| Detection Principle | ATP-dependent bioluminescence assay |
| Sample Types | Surface swabs (UXL100), total aqueous ATP (AQT200), free aqueous ATP (AQF100) |
| Compliance | Designed for use in food safety hygiene monitoring per ISO 22000, HACCP, and FDA Food Code guidance |
| Software | 3M Clean-Trace Hygiene Management Software with wireless data sync, audit-trail-capable reporting, and 21 CFR Part 11–ready configuration options |
| Portability | Handheld, battery-operated, integrated cradle charging station |
Overview
The 3M Clean-Trace ATP LM1 Handheld Bioluminescence Luminometer is an engineered solution for rapid, quantitative assessment of surface and liquid hygiene status in food production, retail, healthcare, and institutional environments. It operates on the well-established principle of ATP-dependent bioluminescence: adenosine triphosphate (ATP), a universal energy metabolite present in all viable microbial cells (bacteria, yeasts, molds) and organic residues, reacts with recombinant firefly luciferase and luciferin to emit measurable photons. The intensity of emitted light—quantified in relative light units (RLUs)—is directly proportional to the amount of ATP extracted from a sample. This correlation enables objective, real-time evaluation of cleaning efficacy and microbial contamination burden without culture-based incubation delays. Unlike qualitative visual inspection or subjective scoring, the LM1 delivers reproducible, instrument-calibrated RLU values traceable to standardized ATP reference materials, supporting evidence-based hygiene verification across GMP-aligned facilities.
Key Features
- Intuitive handheld form factor with ergonomic grip and single-button operation—designed for glove-compatible use in wet or cold environments.
- Integrated cradle docking station with automatic charging and USB-C data synchronization—eliminates manual cable connections during routine workflows.
- Pre-calibrated optical sensor with temperature-compensated photomultiplier tube (PMT) architecture—ensures stable RLU response across ambient operating conditions (10–40 °C).
- On-device pass/fail threshold indicators aligned with industry benchmarks (e.g., ≤100 RLU for food contact surfaces per 3M validation studies).
- Embedded firmware supports firmware updates via Clean-Trace software—maintaining alignment with evolving hygiene protocols and regulatory expectations.
Sample Compatibility & Compliance
The LM1 is validated for use with three proprietary 3M Clean-Trace sampling devices: UXL100 surface swabs (for stainless steel, plastic, conveyor belts, and irregular geometries), AQT200 aqueous total ATP swabs (for rinse water, CIP return lines, and process fluids), and AQF100 free ATP swabs (to distinguish residual sanitizer interference from microbial ATP). All swab chemistries are manufactured under ISO 13485-certified processes and include lot-specific calibration certificates. The system supports compliance with international hygiene management frameworks—including ISO 22000:2018 Clause 8.2 (prerequisite programs), FDA Food Code Appendix 8-301.11 (sanitation verification), and BRCGS Issue 9 Section 4.10.2 (environmental monitoring). When deployed with audit-trail-enabled Clean-Trace software, data capture meets GLP/GMP documentation requirements including user authentication, timestamping, and immutable record retention.
Software & Data Management
The 3M Clean-Trace Hygiene Management Software serves as the central analytical and administrative interface. Its re-engineered dashboard provides role-based access to facility-level hygiene heatmaps, trend analysis over time (daily/weekly/monthly), and root-cause filtering by location, operator, shift, or equipment ID. All reports—including PDF export, CSV raw-data dumps, and automated email distribution—are generated with embedded metadata (instrument ID, swab lot, operator login, GPS-tagged location if enabled). Wireless Bluetooth 5.0 connectivity enables seamless, encrypted data transfer from the LM1 to paired Windows or macOS workstations without physical docking. For regulated environments, optional 21 CFR Part 11 configuration includes electronic signatures, audit trail review logs, and permission-controlled user roles—fully documented in the system validation package provided by 3M Technical Services.
Applications
- Verification of post-clean sanitation effectiveness on food processing lines, packaging machinery, and storage racks.
- Routine environmental monitoring in ready-to-eat (RTE) production zones to support Listeria control plans.
- Validation of hand hygiene compliance among food handlers using UXL100 palm-swab protocols.
- Assessment of CIP cycle performance through AQT200 sampling of final rinse water ATP load.
- Trend-based identification of persistent contamination hotspots—enabling targeted corrective actions and preventive maintenance scheduling.
- Integration into HACCP prerequisite programs as a critical control point (CCP) verification tool for sanitation standard operating procedures (SSOPs).
FAQ
What is the detection limit of the LM1 in femtomoles of ATP?
The LM1 achieves a functional detection limit of ≤1 fmol ATP per test when used with UXL100 swabs under controlled laboratory conditions—equivalent to approximately 10³ CFU/cm² for common foodborne bacteria such as E. coli or L. monocytogenes, based on empirical ATP-to-CFU conversion factors published in peer-reviewed validation studies.
Does the LM1 require daily calibration?
No. The instrument employs factory-calibrated optics and internal reference standards; only periodic verification using 3M-provided ATP check standards (Cat. No. CT-VER) is recommended per ISO/IEC 17025-aligned quality assurance protocols.
Can historical data be migrated from older Clean-Trace instruments?
Yes. Clean-Trace software supports backward compatibility with data files from LM10, NG, and previous-generation luminometers via standardized .ctd file import and cross-platform mapping tools.
Is the LM1 suitable for USDA-inspected facilities?
Yes. The system is routinely deployed in USDA-FSIS-regulated establishments for SSOP verification and has been referenced in multiple FSIS Directive 7120.1 compliance audits as an acceptable ATP monitoring method when used per manufacturer instructions and site-specific validation protocols.