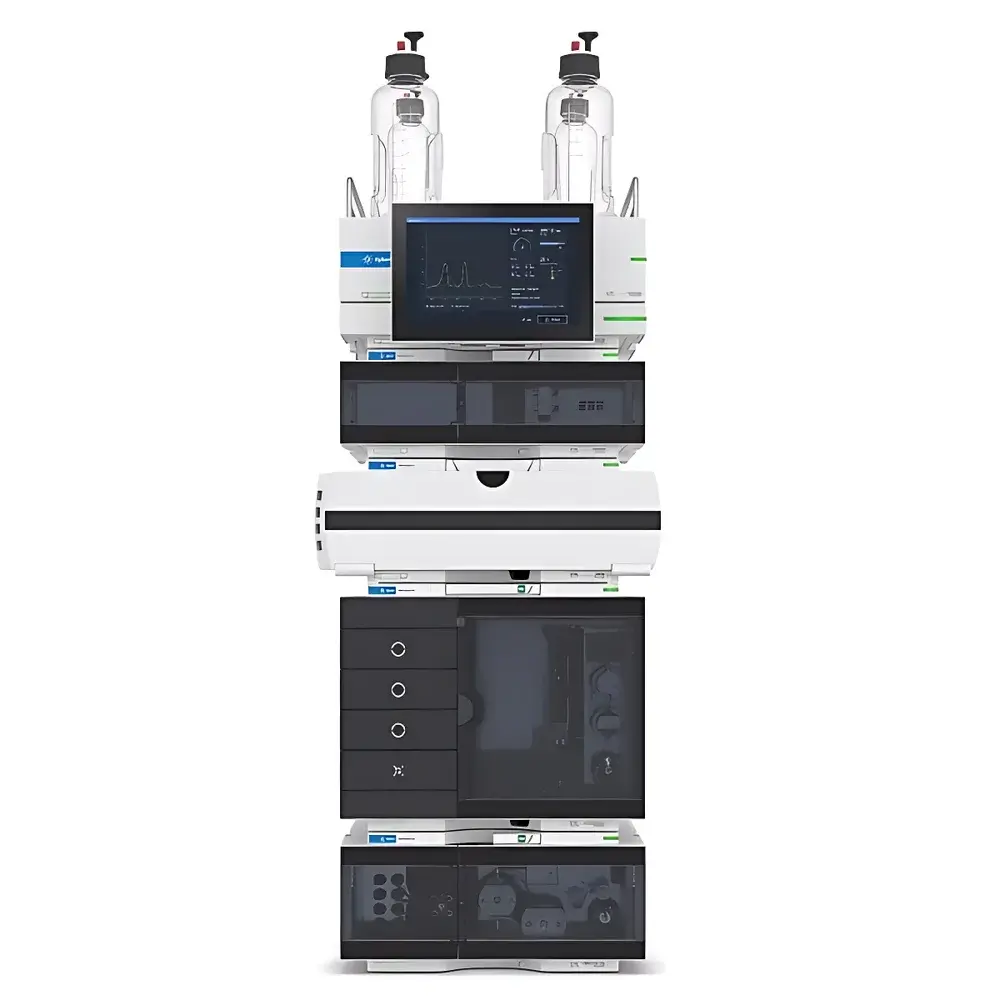
Agilent 1260 Infinity III Prime Bio LC System
| Brand | Agilent Technologies |
|---|---|
| Origin | Germany |
| Manufacturer | Agilent Technologies |
| Origin Category | Imported |
| Model | 1260 Infinity III Prime Bio LC |
| Instrument Type | Ultra-High Performance Liquid Chromatograph (UHPLC) |
| Flow Rate Range | Up to 5 mL/min |
| Maximum Operating Pressure | Up to 800 bar |
| Injection Volume Range | 0.1–100 µL |
| Column ID Reader | Optional |
| Solvent Channels | 4 (integrated quaternary gradient valve) |
| Temperature Zones | 2 |
| Column Capacity | 4 |
| Bio-Compatibility | Yes |
| Power Supply | 100–240 VAC |
| ACT Certified | Yes (My Green Lab ACT Label) |
Overview
The Agilent 1260 Infinity III Prime Bio LC System is a purpose-engineered high-pressure liquid chromatography platform designed specifically for the robust, reproducible, and biocompatible separation of large biomolecules—including monoclonal antibodies (mAbs), antibody-drug conjugates (ADCs), peptides, oligonucleotides, and viral vectors. Built upon the proven architecture of the 1290 Infinity III platform, this system operates under true UHPLC conditions—delivering up to 800 bar maximum pressure and flow rates up to 5 mL/min—while maintaining full compatibility with standard HPLC methods and legacy workflows. Its fluidic path is constructed entirely from inert, bio-inert materials (e.g., titanium, PEEK, and ceramic components), minimizing nonspecific adsorption, metal-catalyzed degradation, and pH-induced denaturation. This ensures structural integrity of sensitive analytes across extreme mobile phase conditions—from pH 1 to pH 13 and high-salt buffers (e.g., up to 2 M ammonium sulfate)—making it suitable for critical quality attribute (CQA) assessment in biopharmaceutical development and QC laboratories operating under ICH Q5, Q6B, and USP guidelines.
Key Features
- Bio-inert fluidic path: Titanium pump heads, ceramic check valves, and PEEK-lined tubing eliminate metal leaching and surface-mediated aggregation or fragmentation.
- High-pressure UHPLC capability: Sustained operation at ≤800 bar enables use of sub-2-µm particle columns and shallow-gradient separations for enhanced resolution of charge variants and glycoforms.
- Quaternary solvent delivery with integrated gradient valve: Enables precise, low-dead-volume mixing of up to four solvents—including volatile and non-volatile buffers—for complex salt/pH gradient elution schemes.
- Expandable sample capacity: Optional deep-well plate drawer accommodates up to 6,144 samples (96- or 384-well format), supporting high-throughput stability-indicating assays and forced degradation studies.
- Agilent Buffer Advisor software: A validated, rule-based tool that automates buffer selection, pH calculation, and gradient generation—reducing manual error and accelerating method development for ion-exchange, hydrophobic interaction, and mixed-mode chromatography.
- Modular detector compatibility: Supports variable wavelength (VWD), diode array (DAD), fluorescence (FLD), multi-wavelength detection (MWD), and direct LC/MS coupling via low-diffusion interfaces.
Sample Compatibility & Compliance
The 1260 Infinity III Prime Bio LC is validated for use with native and denatured proteins, glycans, plasmid DNA, mRNA, and lipid nanoparticles (LNPs). Its biocompatibility extends to aggressive mobile phases used in cation-exchange (CEX), anion-exchange (AEX), hydrophobic interaction (HIC), and size-exclusion (SEC) chromatography. All wetted parts meet USP Class VI and ISO 10993-5 cytotoxicity standards. The system supports 21 CFR Part 11-compliant audit trails when operated with OpenLab CDS or MassHunter software, and includes electronic signatures, user access controls, and instrument qualification (IQ/OQ/PQ) documentation packages aligned with GMP/GLP environments. It conforms to ASTM E2500-13 for analytical instrument lifecycle management and carries the My Green Lab ACT label—verifying its environmental impact across manufacturing, energy use, and end-of-life recyclability.
Software & Data Management
Method development, acquisition, and reporting are managed through Agilent OpenLab CDS (ChemStation Edition or EZChrom Edition), offering full traceability, electronic lab notebook (ELN) integration, and automated report generation compliant with FDA 21 CFR Part 11 Annex 11 requirements. The Buffer Advisor module integrates directly into method templates, allowing real-time buffer formulation validation and automatic export of preparation instructions to lab personnel. Sequence scheduling supports conditional logic (e.g., pause-on-failure, auto-reinjection), while peak tracking algorithms ensure consistent integration of co-eluting species across multi-day stability runs. Raw data files (.D directories) are stored in vendor-neutral formats compatible with third-party reprocessing tools (e.g., Chromeleon, Simplicity).
Applications
- Monoclonal antibody purity profiling (main peak, aggregates, fragments) via SEC and CEX
- Peptide mapping and deamidation/oxidation monitoring under native and denaturing conditions
- Oligonucleotide impurity profiling (n-1, n+1, phosphorothioate diastereomers)
- mRNA cap structure analysis and poly(A) tail length distribution
- Viral vector titer and empty/full capsid ratio quantification by AEX or SEC-MALS
- Host cell protein (HCP) clearance assessment using 2D-LC workflows
FAQ
Is the 1260 Infinity III Prime Bio LC compatible with existing 1200/1260 series methods?
Yes—backward compatibility is engineered into the hardware and firmware. Gradient delay volumes, dwell time compensation, and dwell volume calibration routines enable seamless method transfer from legacy Agilent systems without re-optimization.
Can the system perform pH gradients without manual buffer preparation?
Yes—the integrated Buffer Advisor software calculates exact mixing ratios for two-buffer systems (e.g., sodium acetate/phosphoric acid) and generates executable gradient methods, including pH ramping profiles validated per ICH Q5E.
What column temperature control options are available?
Two independent thermostatted zones support column oven (up to 80 °C) and autosampler tray (4–40 °C), each with ±0.1 °C stability and NIST-traceable calibration certificates.
Does the system support column identification and tracking?
An optional RFID-enabled column ID reader logs serial number, usage history, and maintenance events directly into OpenLab CDS, supporting ALCOA+ data integrity principles.
How is system suitability verified for biopharmaceutical QC applications?
Pre-configured system suitability tests (SSTs) include precision (n=6 injections), resolution (USP tailing factor, plate count), and carryover (<0.05%)—all reportable in IQ/OQ-compliant PDF format with digital signature fields.