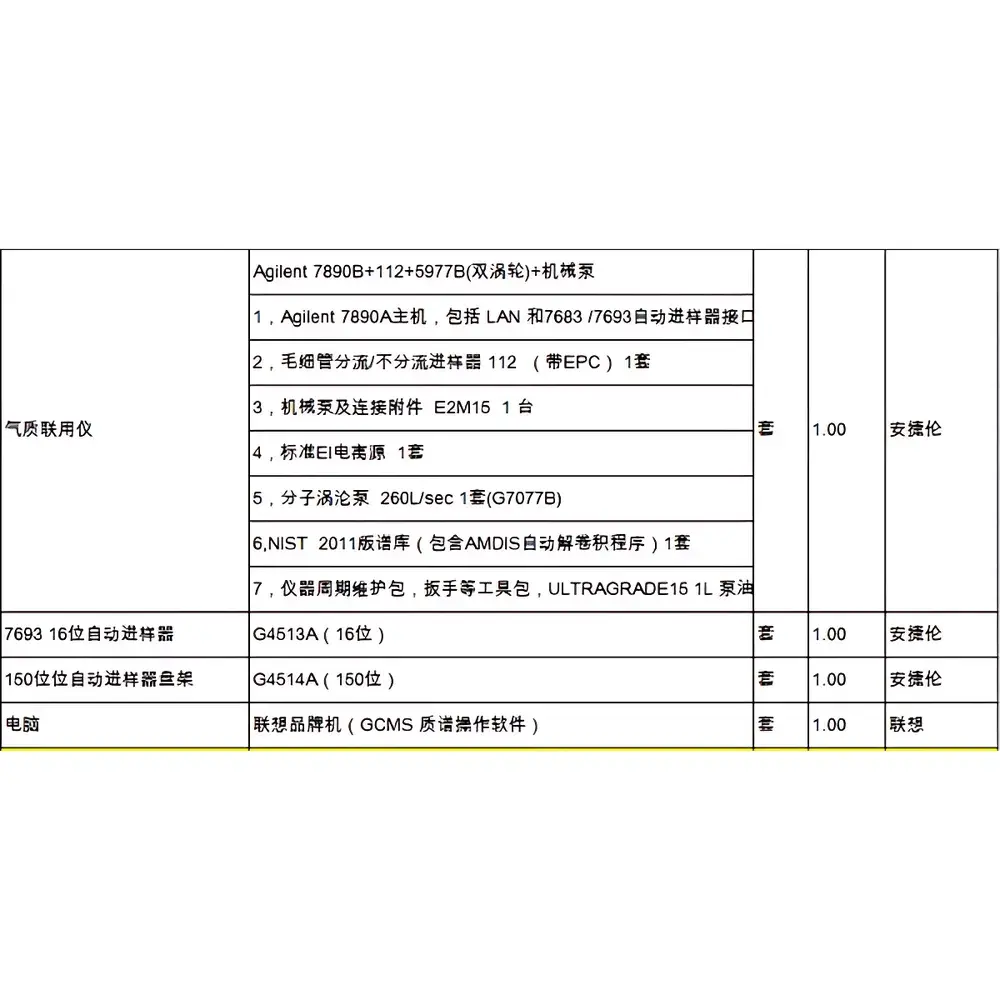
Agilent 7890B GC System Coupled with 5977B Mass Selective Detector (Used)
| Brand | Agilent Technologies |
|---|---|
| Origin | USA |
| Model | Agilent 7890B + 5977B |
| Configuration | Gas Chromatography–Mass Spectrometry (GC-MS) System |
| Vacuum System | High-Performance Turbomolecular Pump |
| Ion Source | Dual-Filament EI Source, Inert (Non-Coated, Non-Stainless Steel) |
| Mass Analyzer | Gold-Plated Quartz Quadrupole with Turner-Kruge Lenses |
| Mass Range | 1.6–1050 u (0.1 u increments) |
| Resolution | Unit Mass Resolution |
| Mass Stability | ≤0.10 u / 48 h |
| Full-Scan Sensitivity (EI) | ≥400:1 S/N for 1 pg OFN (m/z 272, 50–300 u scan) |
| SIM Sensitivity (EI) | ≥10:1 S/N for 20 fg OFN |
| Max Scan Rate | 12,500 u/s (1 ms dwell in SIM) |
| Dynamic Range | 10⁶ |
| SIM Capacity | 100 groups, up to 60 ions/group |
| GC Oven Temp Range | 4 °C above ambient to 450 °C |
| Max Ramp Rate | 120 °C/min |
| Temperature Stability | <0.01 °C per 1 °C ambient change |
| Programmable Temperature Zones | ≥6 heated zones (including inlet, detector, transfer line), all independently controlled up to 400 °C |
| EPC Pressure Control Accuracy | ±0.001 psi |
| Split/Splitless Inlet | Up to 7500:1 split ratio, 0–1250 mL/min H₂/He flow range |
| Auto-sampler | 16-position (standard), optional 150-position tray |
| Detectors Included | FID (detection limit <1.8 pg C/s), micro-ECD (<6 fg/mL lindane), NPD (<0.4 pg N/s, <0.2 pg P/s) |
| Compliance | Fully compatible with EPA Methods (e.g., 502.2, 524.4, 8260, 8270), USP <621>, ASTM D5845, and GLP/GMP data integrity requirements per FDA 21 CFR Part 11 |
Overview
The Agilent 7890B Gas Chromatograph coupled with the 5977B Mass Selective Detector (MSD) represents a mature, field-proven GC-MS platform engineered for high-throughput qualitative and quantitative analysis of volatile and semi-volatile organic compounds. This used instrument configuration operates on electron ionization (EI) principles, utilizing a gold-plated quartz quadrupole mass analyzer and a high-efficiency turbomolecular vacuum system to deliver robust performance across environmental, pharmaceutical, food safety, and forensic laboratories. The system integrates precise gas chromatographic separation—enabled by Agilent’s advanced electronic pressure control (EPC)—with sensitive, reproducible mass spectral detection. Its design supports full-scan, selected ion monitoring (SIM), and trace-level analysis under regulated workflows, including those aligned with U.S. EPA, ISO, and pharmacopeial methods.
Key Features
- High-Fidelity Mass Detection: The 5977B MSD features an inert, non-coated EI ion source with dual long-life filaments and software-selectable ionization energy (5–241.5 eV). Its temperature-controlled source (150–350 °C) and gold-plated quadrupole (operable up to ≥200 °C) ensure consistent sensitivity and reduced memory effects.
- Precision Chromatographic Control: The 7890B GC delivers exceptional retention time stability (<0.008% RSD or <0.0008 min) and peak area reproducibility (<1% RSD). Its 20-stage, 21-platform oven programming supports rapid thermal cycling (120 °C/min ramp; sub-4-minute cool-down from 400 °C to 50 °C), while six independently heated zones—including inlet, detector, and transfer line—minimize condensation and degradation of thermally labile analytes.
- Advanced EPC Architecture: All pneumatic modules employ Agilent’s second-generation EPC, offering ±0.001 psi pressure resolution, automatic ambient compensation, and flexible flow/pressure programs (up to three levels). This enables precise method transfer, robust method development, and seamless integration with multi-detector configurations (FID, micro-ECD, NPD).
- Multidetector Capability: The system includes factory-integrated FID (detection limit <1.8 pg C/s), micro-ECD (<6 fg/mL lindane), and NPD (<0.4 pg N/s), each with dedicated EPC control and optimized dynamic ranges (up to 10⁷ for FID, >5×10⁴ for ECD). These detectors support orthogonal confirmation and compound-class-specific screening without hardware reconfiguration.
- Regulatory-Ready Software Stack: Operates under Agilent MassHunter GC/MS Acquisition and Data Analysis software on Windows OS. Features include retention time locking (RTL), automated calibration, audit-trail-enabled data handling, and full compliance with FDA 21 CFR Part 11, GLP, and GMP documentation standards.
Sample Compatibility & Compliance
This GC-MS system accommodates liquid, solid, and gaseous samples via multiple introduction techniques—including split/splitless injection, purge-and-trap (with optional 80-position autosampler), and headspace analysis. It is validated for use with U.S. EPA Methods 502.2 (VOCs in drinking water), 524.4 (purgeable organics in groundwater), 8260D (VOCs in soil/waste), and 8270E (semivolatiles). The inert ion source and high-temperature transfer line (>300 °C) prevent adsorption or decomposition of polar or reactive analytes. All hardware and firmware revisions meet ISO/IEC 17025 technical competence requirements for accredited testing laboratories. Instrument qualification documentation (IQ/OQ/PQ protocols) is available upon request and aligns with ASTM E2655 and USP <1058> guidelines.
Software & Data Management
Data acquisition, processing, and reporting are managed through Agilent MassHunter software, which provides integrated instrument control, real-time spectral deconvolution, library search (NIST/EPA/WHOLIS), and customizable report templates. The software enforces role-based access control, electronic signatures, and immutable audit trails compliant with FDA 21 CFR Part 11 Subpart B. Raw data files (.D format) are stored in a secure, timestamped hierarchy with automatic backup options. Retention time locking allows cross-instrument method portability without re-optimization. Optional ChemStation legacy compatibility mode supports migration from older Agilent platforms. All software licenses are transferable and include post-purchase remote support and patch updates for the duration of the warranty period.
Applications
- Environmental analysis: Quantification of VOCs, SVOCs, PAHs, PCBs, pesticides, and dioxins in water, soil, air, and biota.
- Pharmaceutical QC/QA: Residual solvent testing per ICH Q3C, extractables/leachables profiling, and impurity identification in APIs and finished dosage forms.
- Food and flavor chemistry: Detection of contaminants (e.g., mycotoxins, plasticizers), aroma profiling, and authenticity verification.
- Forensic toxicology: Screening and confirmation of drugs of abuse, metabolites, and novel psychoactive substances in biological matrices.
- Polymer and materials science: Additive identification, degradation product analysis, and monomer purity assessment.
FAQ
Is this a refurbished or certified pre-owned instrument?
This is a professionally inspected, functionally verified used system. It undergoes full operational validation—including vacuum integrity tests, mass calibration, sensitivity verification using octafluoronaphthalene (OFN), and GC retention time reproducibility checks—prior to shipment.
Does the system include original Agilent software licenses?
Yes. A valid, transferable MassHunter software license (with current maintenance eligibility) is included. Installation media, activation keys, and license transfer documentation are provided.
What level of technical support is available post-purchase?
Buyers receive 12 months of Agilent-authorized remote diagnostics and application support, plus access to Agilent’s global knowledge base and method library. On-site service contracts can be arranged separately.
Can the system be upgraded with newer firmware or hardware modules?
All major subsystems (7890B GC, 5977B MSD, MassHunter v10.x) remain fully supported by Agilent. Optional upgrades include 7693A autosamplers, inert flow path kits, and high-sensitivity tuning standards.
Are IQ/OQ documents and calibration records included?
A complete set of as-tested performance reports—including sensitivity, mass accuracy, retention time stability, and EPC linearity—is delivered with the instrument. Custom IQ/OQ protocol execution is available at additional cost.